Are hydrogen bonds Van der Waals
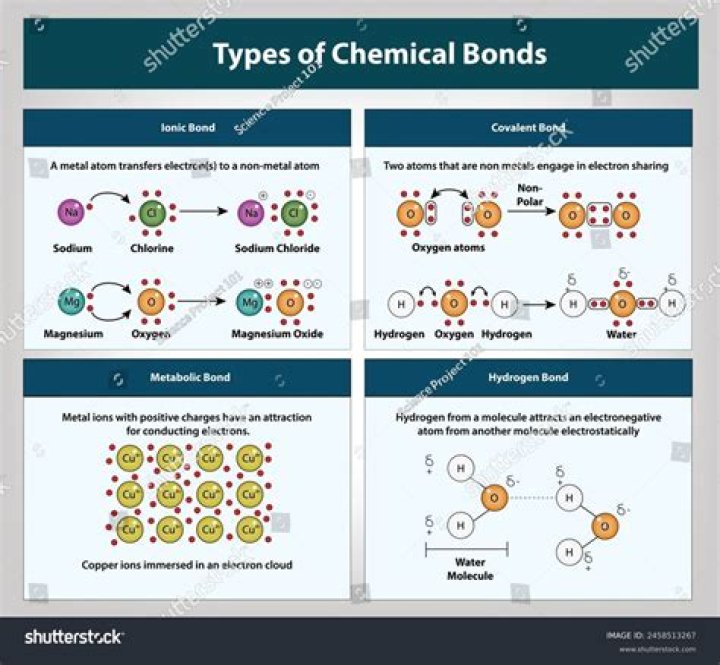
Hydrogen bonding is the third type of van der Waals’ forces. It is exactly the same as dipole-dipole interaction, it just gets a special name. A hydrogen bond is a dipole dipole interaction that occurs between any molecule with a bond between a hydrogen atom and any of oxygen/fluorine/nitrogen.
What is the difference between Van der Waals bond and hydrogen bond?
Hydrogen bonds occur in inorganic molecules, such as water, and organic molecules, such as DNA and proteins. Van der Waals attractions can occur between any two or more molecules and are dependent on slight fluctuations of the electron densities.
Are hydrogen bonds or van der Waals interactions stronger?
2.2 Hydrogen Bonding. A hydrogen bond is the interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen, or fluorine from another molecule. … The hydrogen bond that was generally from 5 to 30 kJ /mol is stronger than a van der Waals interaction, but weaker than covalent or ionic bonds.
What type of bond is van der Waals?
Van der Waals bond: A weak attractive force between atoms or nonpolar molecules caused by a temporary change in dipole moment arising from a brief shift of orbital electrons to one side of one atom or molecule, creating a similar shift in adjacent atoms or molecules.Are covalent bonds Van der Waals?
Covalent compounds exhibit van der Waals intermolecular forces that form bonds of various strengths with other covalent compounds. The three types of van der Waals forces include: 1) dispersion (weak), 2) dipole-dipole (medium), and 3) hydrogen (strong).
What is the difference between van der Waals and dipole dipole?
Summary. Van der Waals forces are weak interactions between molecules that involve dipoles. Polar molecules have permanent dipole-dipole interactions. Non-polar molecules can interact by way of London dispersion forces.
Why are hydrogen bonds not van der Waals?
Hydrogen bonding is a type of dipole-dipole interaction, so it would fit the definition of a van der Waals force. The way I think of it is: van der Waals forces are anything that make a gas non-ideal, since that’s how they were originally discovered and defined.
What kind of bonds are hydrogen bonds?
Hydrogen bonding is a special type of dipole-dipole attraction between molecules, not a covalent bond to a hydrogen atom. It results from the attractive force between a hydrogen atom covalently bonded to a very electronegative atom such as a N, O, or F atom and another very electronegative atom.Does hydrogen fluoride have hydrogen bonding?
Although a diatomic molecule, HF forms relatively strong intermolecular hydrogen bonds. Solid HF consists of zig-zag chains of HF molecules. The HF molecules, with a short H–F bond of 95 pm, are linked to neighboring molecules by intermolecular H–F distances of 155 pm.
Which is weaker hydrogen bonds or van der Waals?Hydrogen bonds are typically stronger than Van der Waals forces.
Article first time published onWhy is hydrogen bonding only possible with hydrogen?
Hydrogen bonds are only possible with hydrogen because hydrogen is small. When hydrogen is bonded to an electronegative atom like nitrogen or oxygen…
Are hydrogen bonds stronger than hydrophobic interactions?
Strength of Hydrophobic Interactions Hydrophobic interactions are relatively stronger than other weak intermolecular forces (i.e., Van der Waals interactions or Hydrogen bonds). … However, at an extreme temperature, hydrophobic interactions will denature.
Are hydrogen bonds stronger than dipole dipole?
Hydrogen bonds are typically stronger than other dipole-dipole forces.
Are hydrogen bonds covalent bonds?
The hydrogen molecule is the simplest substance having a covalent bond. It forms from two hydrogen atoms, each with one electron in a 1s orbital. Both hydrogen atoms share the two electrons in the covalent bond, and each acquires a helium-like electron configuration. A similar bond forms in Cl2.
Are hydrogen bonds polar or nonpolar?
These bonds are extremely polar because of the high electronegativity difference between the atoms. This strong polarity causes very strong dipole-dipole interactions between molecules, called hydrogen bonding. Hydrogen bonds are weaker than chemical bonds.
Are hydrogen bonds intermolecular forces?
A hydrogen bond is an intermolecular force (IMF) that forms a special type of dipole-dipole attraction when a hydrogen atom bonded to a strongly electronegative atom exists in the vicinity of another electronegative atom with a lone pair of electrons. Intermolecular forces (IMFs) occur between molecules.
Are dipole dipole forces van der Waals?
Dipole-dipole force are a type of Van Der Waals force. When two polar molecules interact, opposite partial charges attract, similarly to ionic bonding, but generally weaker, because of the smaller charge magnitude.
Why are hydrogen bonds and van der Waals interactions necessary for cells quizlet?
Why are hydrogen bonds and van der Waals interactions necessary for cells? … They provide the structure and shape necessary for proteins and DNA within cells so that they function properly.
What's the difference between hydrogen bonds and covalent?
A hydrogen bond is formed due to the attraction between two atoms of two different molecules. The main difference between covalent and hydrogen bonds is that covalent bonds are intramolecular attractions whereas hydrogen bonds are intermolecular attractions.
What is difference between dipole-dipole forces and hydrogen bonding?
An ion-dipole force is a force between an ion and a polar molecule. A hydrogen bond is a dipole-dipole force and is an attraction between a slightly positive hydrogen on one molecule and a slightly negative atom on another molecule.
Are van der Waals forces intermolecular or intramolecular?
Van der Waals forces are a category of intermolecular forces that includes London dispersion and dipole-dipole interactions. Some sources also consider hydrogen bonding to be a Van der Waals force.
What intermolecular forces are present in gaseous hydrogen?
Hydrogen Bonds: When a hydrogen atom is covalently bonded to nitrogen, oxygen or fluorine a very strong dipole is formed. The dipole-dipole interactions that result from these dipoles is known as hydrogen bonding.
How do hydrogen bonds form between hydrogen fluoride?
Hydrogen fluoride is formed through covalent bonding (electron sharing) between a hydrogen atom and a fluorine atom. Hydrogen contains one electron, and fluorine requires one electron to become stable, so the bond forms readily when the two elements interact.
What intermolecular forces does hydrogen fluoride have?
Hydrogen fluoride, HF, has extensive INTERMOLECULAR hydrogen bonding, because the individual molecules comprise a molecular dipole, i.e. “”^(delta+)H-F^(delta-), and this intermolecular force is responsible for the elevated normal boiling point of HF at 19.5 “”^@C.
Does N Chloromethylamine have hydrogen bonding?
We are provided the molecule with the name N-chloromethylamine. … Due to large size of chlorine it doesn’t show hydrogen bonding and due to smaller size of nitrogen it shows hydrogen bonding.
Are hydrogen bonds covalent or ionic?
Hydrogen bonds and London dispersion forces are both examples of van der Waals forces, a general term for intermolecular interactions that do not involve covalent bonds or ions.
What kind of bonds are hydrogen bonds apex?
Hydrogen bonding is a kind of ‘dipole-dipole interaction’, where two molecules with a dipole moment – some charge separation – are attracted to each other. Molecules have charge separation due to electronegativity differences among the molecules that make them up.
Where does hydrogen bonding occur?
Hydrogen bonding occurs only in molecules where hydrogen is covalently bonded to one of three elements: fluorine, oxygen, or nitrogen. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the H atom very electron-deficient.
Are hydrogen bonds weak or strong?
hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; such a bond is weaker than an ionic bond or covalent bond but stronger than van der Waals forces.
Is hydrogen bond stronger or weaker than van der Waals forces explain giving reason?
Hydrogen bonds are stronger than the van der Waals forces because H-bonds are considered as an extreme form of dipole-dipole interaction.
Why are hydrogen bonds the weakest bonds?
Because the hydrogen is slightly positive, it will be attracted to neighboring negative charges. When this happens, a weak interaction occurs between the δ+ of the hydrogen from one molecule and the δ– charge on the more electronegative atoms of another molecule, usually oxygen or nitrogen, or within the same molecule.



