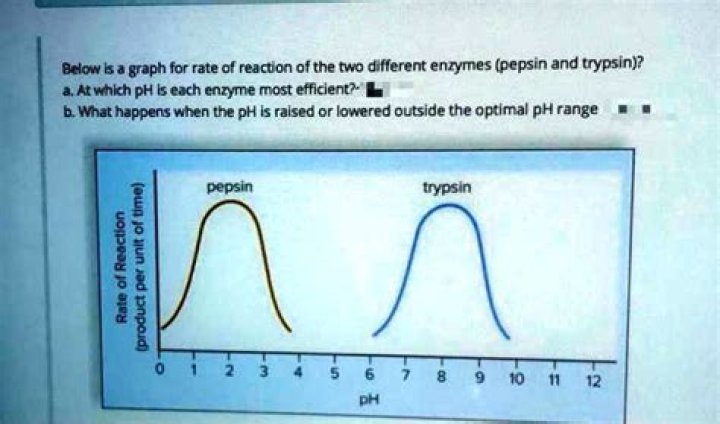
At what pH value is pepsin likely to denature
Increasing the pH to >7.2 (as expected in the normal small intestine) or the temperature to >65°C irreversibly denatures pepsin, whereas pepsinogen is stable to pH 10 and 100°C. The molecular structure of human pepsin (Fig.
At what pH will pepsin likely denature?
neutral pH range (> pH 6.0) pepsin is rapidly denatured.
Is the enzyme pepsin likely to be denatured by low pH?
At what pH values is pepsin likely to be denatured? Justify your answer. pH 5-14 At these pH values, pepsin has no activity – shape has likely been altered.
Does pepsin denature in high pH?
Pepsin Inactive at Higher pH Pepsin becomes inactive in this environment because the concentration of hydrogen atoms is lower. The hydrogen on pepsin’s carboxylic acid in the enzyme active site is then removed, and the enzyme becomes inactive.At what pH do enzymes denature?
EnzymeOptimum pHPancreatic protease (trypsin)7.5–8.0
At what pH should the reaction occur optimally?
A substrate is no longer able to fit into the active site, and the reaction does not occur. A pH of about 7 is optimal and every level away from it, whether its more acidic or basic, slows the reaction. Every single enzyme shows highest activity, called the optimal temperature.
What pH value is lipase likely to denature?
At pH values above 7, lipase denatures and aggregates when heated at temperatures above 45 degrees C. However, at pH below 6 lipase denatures upon heating but the activity and its native structure is completely recovered upon cooling.
What is the optimal pH level for pepsin activity quizlet?
For the enzyme pepsin, which works in the stomach, optimal activity occurs around pH 2.Does the effectiveness of pepsin depend on pH?
Does the effectiveness of pepsin depend on pH? Yes, pepsin is so acidic that it can only work in a very acidic atmosphere, only affective if has the proper pH.
How extreme pH denature enzymes?These are the ionic and hydrogen bonds. Extreme pHs can therefore cause these bonds to break. When the bonds holding the complementary active site of an enzyme break, it cannot bind to its substrate. The enzyme is thus denatured, as no enzyme-substrate or enzyme-product complexes can form.
Article first time published onWhy does pH denature?
Changes in pH affect the chemistry of amino acid residues and can lead to denaturation. … Protonation of the amino acid residues (when an acidic proton H + attaches to a lone pair of electrons on a nitrogen) changes whether or not they participate in hydrogen bonding, so a change in the pH can denature a protein.
Do all enzymes work best at pH 7?
Enzymes in the stomach, such as pepsin ( which digests protein ), work best in very acid conditions ( pH 1 – 2 ), but most enzymes in the body work best close to pH 7.
At which pH does the lipase catalyzed reaction proceed the fastest?
What would happen if you used a human polymerase in a series of PCR reactions? Denending on fining, å new human polymerase would be used each The rate of an enzyme-catalyzed reaction can also be affected by the presence of other molecules that can time. 22.
At what pH is trypsin working at its maximum rate?
Trypsin is a serine protease which is secreted by the pancreas and is most active in the pH range between 7 and 9 at 37°C. It reacts with peptide bonds between the carboxylic acid group of lysine or arginine and the amino group of the adjacent amino acid residue.
At what pH is enzyme B working at its maximum rate?
Enzyme B is working at its maximum rate at a pH of 8.
What is the optimal pH that this enzyme functions at?
Most other enzymes function within a working pH range of about pH 5-9 with neutral pH 7 being the optimum.
At what temperature do enzymes begin to denature?
Optimum Temperature While higher temperatures do increase the activity of enzymes and the rate of reactions, enzymes are still proteins, and as with all proteins, temperatures above 104 degrees Fahrenheit, 40 degrees Celsius, will start to break them down.
What could denature pepsin?
Increasing the pH to >7.2 (as expected in the normal small intestine) or the temperature to >65°C irreversibly denatures pepsin, whereas pepsinogen is stable to pH 10 and 100°C. … The central hydrophobic core of pepsin (catalytic aspartic acid residues at position 32 and 215) comprises the active site of the enzyme.
What happens to the activity of pepsin when it enters the small intestine where the pH is 8?
Pepsin works best at a pH of 2. And as the stomach has a very acidic condition, pepsin works well. But in the small intestine, the pH is about 8, which is too high for pepsin to function. Therefore, pepsin gets denatured in the small intestine.
What is the optimum temperature for pepsin?
When testing the effect of temperature on pepsin enzyme activity, the results showed that pepsin worked best at the temperature 30 °C.
What occurred when pepsin was boiled quizlet?
What occurred when pepsin was boiled? It was inactivated. The enzyme didn’t work because the pH was too low.
Why natural selection might have resulted in the optimal pH for pepsin?
Using the figure to guide your response, select the best explanation for why natural selection might have resulted in the optimal pH for pepsin. Natural selection favors enzymatic activity of pepsin at a pH of 2.0 because 2.0 is the pH inside the stomach.
What is the optimal pH for Ptyalin?
The optimum conditions for ptyalin activity are a pH range of 5.6–6.9, a temperature of 378C, and the presence of certain anions and activators, such as chloride, bromide, and iodide.
Which enzyme has an optimum pH that is alkaline basic pH above 7?
EnzymepH OptimumPepsin1.5 – 1.6Trypsin7.8 – 8.7Urease7.0Invertase4.5
How do enzymes denature?
As the temperature rises, reacting molecules have more and more kinetic energy. … Above this temperature the enzyme structure begins to break down (denature) since at higher temperatures intra- and intermolecular bonds are broken as the enzyme molecules gain even more kinetic energy.
How does pH affect the rate of reaction?
Optimal pH increases enzyme rate of reaction while less than optimal pH decreases it. Increasing temperature also increases enzyme rate of reaction, until things get too hot, then the enzyme denatures and ceases to function.
How pH causes protein denaturation?
Protein denaturation due to pH The ionizable groups in amino acids are able to become ionized when changes in pH occur. A pH change to more acidic or more basic conditions can induce unfolding. Acid-induced unfolding often occurs between pH 2 and 5, base-induced unfolding usually requires pH 10 or higher.
How does decreasing pH affect proteins?
Decreasing the pH by adding an acid converts the –COO- ion to a neutral -COOH group. In each case the ionic attraction disappears, and the protein shape unfolds. Various amino acid side chains can hydrogen bond to each other. … Changing the pH disrupts the hydrogen bonds, and this changes the shape of the protein.
Which statement accurately describes how pH is related to denaturation?
Which statement accurately describes how pH is related to denaturation? Either a large increase or a large decrease in pH can lead to denaturation. The higher the concentration of H+ is, the lower the pH is. polar and therefore increase a biomolecule’s solubility in water.
Why does pepsin work best at 37 degrees?
Most enzyme functions are performed at 37∘C in humans because the enzymes are able to retain its structure at that temperature, allowing it to break down complex molecules efficiently.
What is the effect of pH on pepsin action on albumen?
Pepsin, which is a digestive enzyme, has been observed to have an optimum pH of approximately 2-3. This means that Pepsin works best, and maintains the active site most at a pH of 2-3. … A pH which is varied too much from this optimum, will cause the shape of the enzyme to distort and the enzyme will no longer be active.