Does bromine have high or low electronegativity
The first scale of electronegativity was developed by Linus Pauling and on his scale bromine has a value of 2.96 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).
Does bromine have high or low electronegativity? - Google Search
Electronegativity is the tendency for an atom to attract electrons. … As such, it has a lower tendency to attract electrons , and thus has a lower electronegativity. In the case of carbon, bromine and fluorine, they all can gain electrons to achieve “full outer shells” (fluorine is in fact the most electronegative atom).
What is a high or low electronegativity?
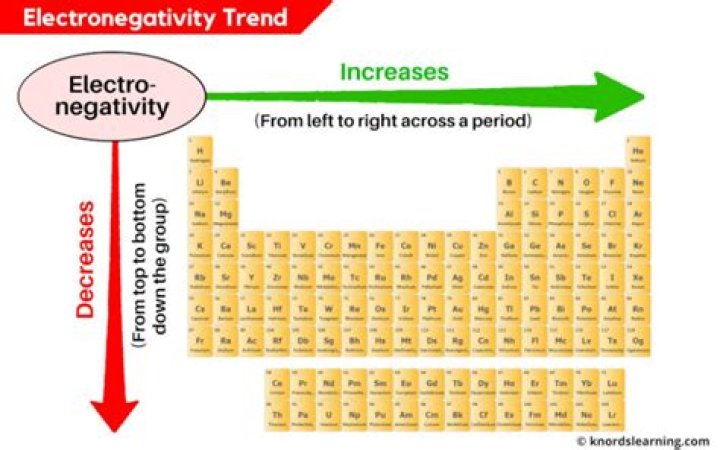
Electronegativity refers to the ability of an atom to attract shared electrons in a covalent bond. The higher the value of the electronegativity, the more strongly that element attracts the shared electrons. … Electronegativity increases from bottom to top in groups, and increases from left to right across periods.
Does bromine or chlorine have a high electronegativity?
HalogenElectronegativityFluorine4.0Chlorine3.0Bromine2.8Iodine2.5Why does bromine have a lower electronegativity than chlorine?
The atomic radius of the atom. This makes the radius (the distance from the nucleus to the outer shell) of the bromine atom larger than that of the chlorine atom. The bromine nucleus therefore has to attract an electron from further away, and this greater distance means the electron is attracted less strongly. 3.
Why does chlorine have a high electronegativity?
Electronegativity is the ability an atom has to attract other electrons. Chlorine has higher electronegativity value than iodine because chlorine has small size than iodine. So, which atoms do you think will more easily pull in electrons?
Which of the following has the lowest electronegativity?
Of the main group elements, fluorine has the highest electronegativity (EN = 4.0) and cesium the lowest (EN = 0.79). This indicates that fluorine has a high tendency to gain electrons from other elements with lower electronegativities. We can use these values to predict what happens when certain elements combine.
What is low electronegativity?
Electronegativity is the tendency of an atom to attract an approaching bonding pair of electrons from another atom. … So a low electronegativity element is one that is low atomic number, large atomic radius, and with a greater number of screening outer electrons.How do you determine electronegativity?
Subtract the smaller electronegativity from the larger one to find the difference. For example, if we’re looking at the molecule HF, we would subtract the electronegativity of hydrogen (2.1) from fluorine (4.0). 4.0 – 2.1 = 1.9.
What is the electronegativity of nacl?Electronegativity DifferenceType of Bond Formed> 1.5ionic
Article first time published onWhere is the highest electronegativity found?
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
What causes low electronegativity?
If an atoms is larger with more electron shells it will have a lower electronegativity, If the nucleus has fewer protons in the same period it will have a lower electronegativity.
Is bromine less reactive than chlorine?
Because chlorine is more reactive than bromine, it displaces bromine from sodium bromide.
Why bromine is less reactive but more selective than chlorine?
Bromination of alkanes occurs by a similar mechanism, but is slower and more selective because a bromine atom is a less reactive hydrogen abstraction agent than a chlorine atom, as reflected by the higher bond energy of H-Cl than H-Br.
Which one of the following has highest electronegativity?
Answer: Thus, fluorine is the most electronegative element, while francium is one of the least electronegative.
Which of the following elements has the highest electronegativity answers?
The correct answer is option 1, i.e. Fluorine. Fluorine is the most electronegative element. As we move right in a period, the electronegativity keeps increasing up to the halogens.
Which element has the lowest electronegativity in period 2?
> Hence, in the second period of the periodic table, the least electronegative element is lithium. In fact, lithium is the most electropositive element in the second period of the periodic table. Hence, the option A ) lithium is the correct answer.
Why do nonmetals have high electronegativity?
Electronegativity differs from electron affinity because electron affinity is the actual energy released when an atom gains an electron. … Nonmetals have more valence electrons and increase their stability by gaining electrons to become anions. The electronegativities of nonmetals are generally high.
Does chlorine have a high electronegativity?
ElementchlorineSymbolClAtomic number17Electronegativity3.16
Which has higher electronegativity metals or nonmetals?
Nonmetals have much higher electronegativities than metals; of the nonmetals, fluorine is the most electronegative, followed by oxygen, nitrogen, and chlorine. The larger the difference in electronegativity between two atoms, the more polar the bond between them.
What is the electronegativity of so4?
ElectronegativityValue in Pauling unitsPauling electronegativity2.58Sanderson electronegativity2.96Allred Rochow electronegativity2.44Mulliken-Jaffe electronegativity2.69 (16.7% s orbital)
What are some examples of electronegativity?
- The Concept of Electronegativity. Unlike many other periodic trends, electronegativity does not have actual units. …
- Sodium Chloride, an Ionic Bond. Sodium has an electronegativity of 0.9, while chlorine has an electronegativity of 3.0. …
- Water, a Polar Bond. …
- Hydrogen Gas, a Covalent Bond.
How do you tell if a bond is ionic or covalent using electronegativity?
1. If the electronegativity difference (usually called ΔEN) is less than 0.5, then the bond is nonpolar covalent. 3. If the ΔEN is greater than 2.0, then the bond is ionic.
Is ch4 polar compound?
Methane (CH4) is a non-polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms. Methane is non-polar as the difference in electronegativities between carbon and hydrogen is not great enough to form a polarized chemical bond.
Why do alkali metals have low electronegativity?
As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly attracted towards it. This corresponds with a decrease in electronegativity down Group 1.
What is the electronegativity difference of bromine?
ElectronegativityValue in Pauling unitsPauling electronegativity2.96Sanderson electronegativity3.22Allred Rochow electronegativity2.74Mulliken-Jaffe electronegativity2.95 (14.3% s orbital)
What is the electronegativity difference of sodium and bromine?
ElementElectronegativityHydrogen (H)2.1Sodium (Na)0.9Magnesium (Mg)1.2Calcium (Ca)1.0
Do ionic compounds have high electronegativity?
Type of BondDifference in ElectronegativityIonicgreater than 2.1
Does fluorine or bromine have a larger value for electronegativity group of answer choices?
(Noble Gases do not tend to form chemical bonds and so escape consideration). Down a Group, the nuclear charge tends to be shielded by inner core electrons, and thus fluorine is more electronegative than chlorine, which is more electronegative than bromine etc.
What factor affect electronegativity?
An atom’s electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons.
Which element is more reactive chlorine or bromine?
Chlorine is more reactive than both bromine and iodine. But fluorine is the most reactive of the halogens.



