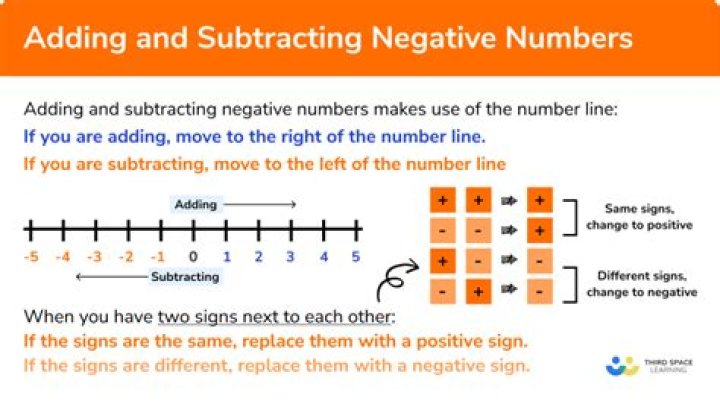
Does Iron react with dilute acid

Dilute acids react with relatively reactive metals such as magnesium, aluminium, zinc and iron. The products of the reaction are a salt plus hydrogen gas. … In general, the more reactive the metal, the faster the reaction.
What happens when you put acid on iron?
Oxidizing acids like nitric acid react with iron to form a passivating layer on the surface of the iron; this passivating layer protects the iron underneath from further attack by the acid, although the brittle oxides of the layer can flake off and leave the interior metal exposed.
What happen when iron react with dilute sulphuric acid?
When iron reacts with dilute sulphuric acid, it forms iron sulphate and hydrogen gas. When hydrogen is ignited with a burning matchstick it burns with a pop sound.
Do iron reacts with hydrochloric acid?
Iron reacts with hydrochloric acid to produce iron(II) chloride and hydrogen gas.What is produced when iron reacts with dilute hydrochloric acid?
Iron reacts with dilute hydrochloric acid to produce iron chloride solution and one other product.
Which of the following metals will not react with dilute acid?
– Therefore the metals which do not react with dilute hydrochloric acid are copper and mercury.
What happens when metal reacts with dilute acid?
Metals react with dilute hydrochloric acid to form the corresponding chloride salt and hydrogen which is evolved as bubbles.
Does iron react with bases?
With bases iron does not usually react unless the base is weak, in which case a displacement reaction will occur and the cation of the base will become free.Which test tube contains iron and dilute hydrochloric acid?
Test-tube that contains iron and dilute hydrochloric acid is the one where greenish FeCl2 is formed. Green coloured Iron(II) chloride and hydrogen gas are created when dilute hydrochloric acid are introduced to iron filings.
Which chemical reaction correctly represents the reaction between iron and dilute?2Fe+3H2SO4→Fe2(SO4)+3H2.
Article first time published onWhat happens on adding dilute HCl to a mixture of iron filings and Sulphur?
As part ‘A’ is heated, a compound FeS is formed by the reaction between iron filings and sulphur. When dilute HCI is added to part A, FeS will react with dil HCI to form H2S gas which has smell of rotten eggs and will turn lead acetate paper black.
Which one of the following is observed when dilute HCl is added to iron filings in a conical flask?
when dilute HCl is added to Fe then Ferrous chloride and hydrogen gas will be form.
Does silver react with acid?
Reaction of silver with acids Silver metal dissolves in hot concentrated sulphuric acid. Silver metal also dissolves in dilute or concentrated nitric acid, HNO3.
What metals react with dilute acids?
Dilute acids react with relatively reactive metals such as magnesium, aluminium, zinc and iron. The products of the reaction are a salt plus hydrogen gas.
Which metal can displace iron from iron sulphate solution?
Copper can displace iron from iron sulphate.
Does copper react with acid?
Copper has a lower reduction potential than hydrogen, it does not react with non – oxidising acids like HCl. C. Copper is a less reactive metal, hence does not react with any acid.
Which element has no reaction with dilute acid?
A metal below hydrogen in the reactivity series will not react with dilute acids. Platinum is placed below gold in the reactivity series.
Which metals Cannot react with acid?
We know gold is a noble metal and it lies below hydrogen in the reactivity series and thus it is unreactive towards acids.
Will zinc react with dilute acid?
Zinc is more reactive than hydrogen and it displaces hydrogen from dilute acids. Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved.
Which metals will react with iron III chloride?
Barium is higher than iron on the reactivity series, and so, barium will react with iron(III) chloride.
What happens when you mix iron and copper?
There are times when iron will be added into copper alloys to provide positive benefits. Some of the main benefits to adding iron into copper alloys is to provide increased tensile strength and corrosion resistance without impacting the conductivity that the copper alloys already possess.
What happens when you mix iron with copper chloride?
Solid Iron reacts with an aqueous solution of copper(II) chloride to produce aqueous iron (II) chloride and solid copper. … This reaction has a 1:1 mole ratio between iron used and copper produced. The iron will be provided by two iron nails. The copper will initially be in the form of Cu2+ ions in solution.
Which ion causes the acidity in dilute hydrochloric acid?
Acid nameFormulaIonic FormulaHydrochloric acidHClH +(aq) Cl –(aq)Sulfuric acidH 2SO 42H +(aq) SO 4 2-(aq)Nitric acidHNO 3H +(aq) NO 3 -(aq)
Which of the following oxide S of Iron would be obtained on prolonged reaction of iron with steam?
Answer: (c) Fe3O4 The formation of iron(II, III) oxide and hydrogen when red hot iron interacts with steam.
Why acid is added to water along the sides of the test tube?
The sulfuric acid is very dense and it will pass through your test solution and go to the bottom of your test tube.
Does iron corrode with acid?
It is observed that iron corrodes in different concentration of sulphuric acid because there is considerable decrease in original weight of the specimen. The corrosion is due to water, air and hydrogen ion, which enhance the corrosion process. The corrosion takes place at the surface of iron, which is a reaction site.
Is iron acidic or basic?
Materials: Metals and Non-Metals | Exercise Iron metal reacts slowly with the oxygen and moisture of damp air to form rust. State, whether the rust formed, is acidic, basic, or neutral. Iron metal reacts slowly with the oxygen and moisture of damp air to form rust. Rust is a metal oxide hence, it is basic in nature.
Which gas is given out when iron reacts with dilute sulphuric acid?
Hydrogen disulphate gas liberated when dilute sulphuric acid reacts with iron sulphide.
Does iron react with concentrated sulphuric acid?
Iron react with sulfuric acid to produce iron sulfate (III), oxide sulfur (IV) and water. Sulfuric acid – concentrated solution. This reaction takes place heating the reaction mixture.
What gas can be obtained when diluted HCl is added to a mixture of iron and sulfur?
When dilute HCL is added to setup B Hydrogen gas is evolved and sulphur does not take part in the reaction. When a burning match stick is brought near the evolved gas, matchstick burns with a pop. This is the indication of the production of Hydrogen gas.
When dilute hydrochloric acid is added to a mixture of iron and sulphur the gas given out is ?
Hydrogen gas and iron chloride are produced.