Does sulfur form double bonds
Because sulfur does not form strong S=S double bonds, elemental sulfur usually consists of cyclic S8 molecules in which each atom completes its octet by forming single bonds to two neighboring atoms, as shown in the figure below. S8 molecules can pack to form more than one crystal.
What bonds does sulfur form?
Covalent Bonding : Example Question #2 Sulfur is a nonmetal in group 6A , and therefore has 6 valence electrons. In order to obey the octet rule, it needs to gain 2 electrons . It can do this by forming 2 single covalent bonds.
Can sulfur form three bonds?
It is in the same column of the periodic table of elements as oxygen is and oxygen will form two bonds. … Of course sulfur is not oxygen, so it does not behave exactly the same way that oxygen behaves. For example, sulfur can form up to six bonds, which is what is the case in something like sulfuric acid — (H2)SO4.
What can form double bonds?
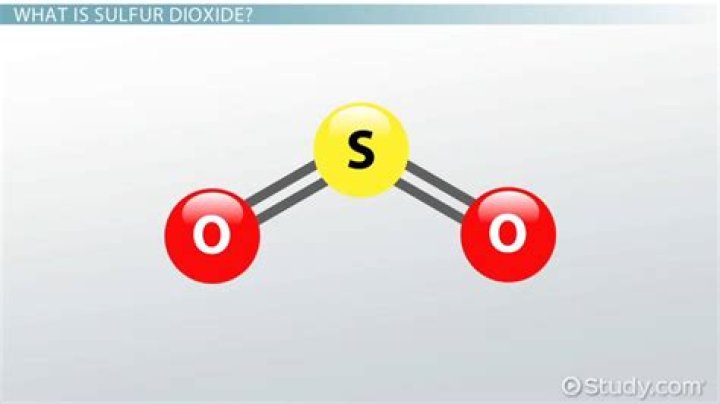
Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O).Does sulfur dioxide have two double bonds?
In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur. To minimise repulsions, the double bonds and the lone pair get as far apart as possible, and so the molecule is bent.
Can sulfur form ionic bonds?
Sulfur, which only needs two electrons to complete it octet, will pick up the two electrons coming from magnesium, becoming the sulfide anion, S2− , in the process. The electrostatic force of attraction will then bring the magnesium cations and the sulfur anions together → an ionic bond is formed.
Can sulfur Form 8 bonds?
Sulfur can make use of its 2 unpaired electrons to form 2 covalent bonds plus the 4 electrons from its 2 lone pairs to give a total of 8 electrons. Hence sulfur obeys octet rule in this case. … So in addition to being octet, sulfur can expand octet to have 10 or 12 electrons.
Which atoms Cannot form double bonds?
The double bond rule states that chemical elements with a principal quantum number greater than 2 for their valence electrons (period 3 elements and lower) tend not to form multiple bonds (e.g. double bonds and triple bonds).What elements can form double or triple bonds?
The formation of double and triple bonds is not as widespread among the atoms of the periodic table as one might expect. At least one of the atoms involved in a multiple bond is almost always C, N, or O, and in most cases both atoms are members of this trio.
How do you find double bonds?DBE = UN = PBoR = C – (H/2) + (N/2) +1, where: C = number of carbon atoms, H = number of hydrogen and halogen atoms, and N = number of nitrogen atoms. One DBE = one ring or one double bond.
Article first time published onCan sulfur make 6 bonds?
Sulfur is capable of forming 6 bonds because it can have an expanded valence shell; sulfur is in period 3 of the Periodic Table.
How many bonds and lone pairs does sulfur have?
The sulfur atom shares a bonding pair and three lone pairs. In total, it has six valence electrons.
How many bonds does neutral sulfur have?
In summary, Sulfur is neutral in SO4, and has six valence electrons. The sulfur atom shares four valence electrons with two oxygen atoms as two covalent double bonds, and shares its remaining two electrons with two oxygen atoms, forming two covalent single bonds.
What is the bond order of SO3 2?
In SO3-2 ion, there is a total of four sigma bonds and π bonds to the three O atoms. Therefore, the bond order of S-O bond becomes 4/3= 1.33.
Which substance would form a double bond when bonded covalently?
Two oxygen atoms can both achieve stable structures by sharing two pairs of electrons as in the diagram. The double bond is shown conventionally by two lines joining the atoms. Each line represents one pair of shared electrons. Ethene has a double bond between the two carbon atoms.
Does sulfur follow octet rule?
Sulfur can follow the octet rule as in the molecule SF2. Each atom is surrounded by eight electrons. It is possible to excite the sulfur atom sufficiently to push valence atoms into the d orbital to allow molecules such as SF4 and SF6.
What bond is formed between Sulphur and oxygen?
NaHSO4=sodium hydrogen sulfateNa2SO4=sodium sulfate
What type of bond is formed between Sulphur and oxygen?
Sulfur and oxygen are both nonmetals due to their position on the periodic table, and so sulfur monoxide or SO would just be held by covalent bonding.
Is Sulphur ionic or covalent?
Each sulfur atom is bonded to each of its two neighbors in the ring by covalent S-S single bonds.
How do ions form ionic bonds?
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.
What does barium and Sulfur form?
Barium and sulfur form several sulfides, of which the most studied is barium sulfide (BaS) (81.07% Ba), which melts at 2200°C. Other sulfides of barium are Ba2S, BaS2, and BaS3. The sulfide BaS crystallizes in a cubic lattice of the type NaCl (a = 0.6381 nm), which has a density of 4.252 g/cm3.
What two elements can form single double and triple bonds?
Carbon-Carbon Bonds Carbon can form single, double, or even triple bonds with other carbon atoms. In a single bond, two carbon atoms share one pair of electrons. In a double bond, they share two pairs of electrons, and in a triple bond they share three pairs of electrons.
How do you know when to add a double bond?
When figuring out whether to place a double or triple bond you should always look at the number of valence electrons present as well as the number of bonds a central atom is likely to form. Another good way to know whether to use double or single bonds is to calculate the formal charge on each atom in the molecule.
Which elements can triple bond?
Boron joins carbon and nitrogen as one of the few elements in the periodic table known to form stable compounds featuring triple bonds.
Why do atoms make double bonds?
Double and triple covalent bonds occur when four or six electrons are shared between two atoms, and they are indicated in Lewis structures by drawing two or three lines connecting one atom to another.
Can bromine form double bonds?
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The double bond breaks, and a bromine atom becomes attached to each carbon.
Does boron form double bonds?
Boron is an unusual molecule because it does not follow the octet rule by having eight valence electrons around the boron atom. BF3 has single bonds between the boron atom and the fluorine atoms and contains no double bonds and an empty p orbital (figure 3).
What is double bond equivalent with example?
As in simple words, the double bond equivalent is the number of double bonds and number of triple bonds present in organic molecules. For example, in the case of benzene, there are three double bonds and one ring so its double bond equivalent is four.
What is alkenes general formula?
Alkenes are defined as either branched or unbranched hydrocarbons that possess at least one carbon–carbon double bond (CC) and have a general formula of CnH2n [1].
Can sulfur form 5 bonds?
Sulfur usually forms 2 bonds, e.g. H2S, -S-S- compounds… This is because of its 3p4 orbital. p-orbitals allow for 6 places to be filled, hence sulfur tends to form 2 bonds. It can “expand the octet” as it has 6 valence electrons, hence allowing the formation of 6 bonds.
How many covalent bonds can sulfur form with atomic number 16?
Sulfur makes use of its 2 unpaired electrons in the 3p orbital to form covalent bonds. Sulfur has got 2 unpaired electrons and 2 lone pairs giving a total of 8 electrons. Thus, obeying the octet rule. Now sulfur has got 4 unpaired electrons which can form 4 covalent bonds.