How do R groups affect protein structure
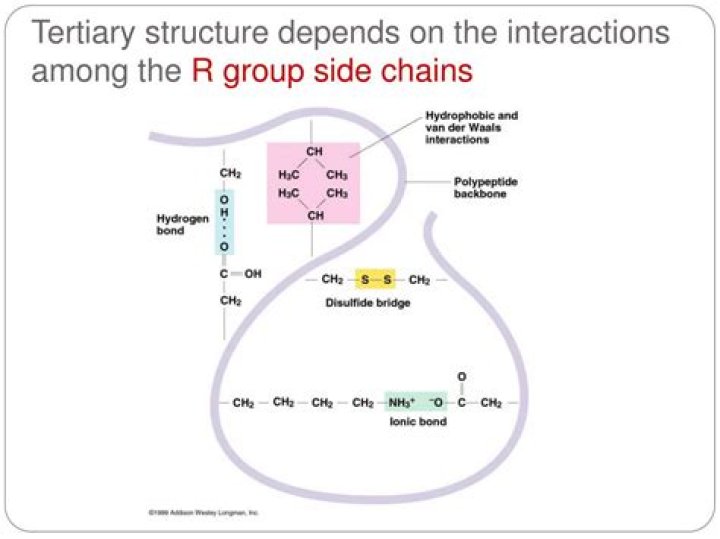
The R group determines the characteristics (size, polarity, and pH) for each type of amino acid. Peptide bonds form between the carboxyl group of one amino acid and the amino group of another through dehydration synthesis. A chain of amino acids is a polypeptide.
How do R groups affect the tertiary structure of a protein?
R group interactions that contribute to tertiary structure include hydrogen bonding, ionic bonding, dipole-dipole interactions, and London dispersion forces – basically, the whole gamut of non-covalent bonds.
How do R groups affect protein folding?
Most proteins fold into complex, three dimensional, globular shapes. Hydrophilic R-groups interact positively with the surrounding water. The entire chain twists until the maximum number of these groups are in full contact with the surrounding water.
How do R groups affect protein structure quizlet?
*The structure of the R-group affects the chemical reactivity and solubility of the amino acid. *In proteins, amino acids are joined bu a peptide bond between the carboxyl group of one amino acid and the amino group of another amino acid. … the combination of polypeptides represents the protein’s quarternary structure.What is the function of the R group attached to the protein?
Each of the 20 amino acids has a specific side chain, known as an R group, that is also attached to the α carbon. The R groups have a variety of shapes, sizes, charges, and reactivities. This allows amino acids to be grouped according to the chemical properties of their side chains.
What is the R in protein?
The amino acids present in proteins differ from each other in the structure of their side (R) chains. The simplest amino acid is glycine, in which R is a hydrogen atom. In a number of amino acids, R represents straight or branched carbon chains.
What is the effect of temperature on protein shape?
Protein is very sensitive to temperature. Temperature change denature protein and changes its shape. The amino acid sequence in protein structure does not change due to temperature but the folding of the three-dimension polypeptide chain changes.
How does the primary protein structure affect the function of protein enzymes?
The structure of a protein determines its activity. A protein’s functioning will be altered or lost if its structure changes. Enzyme proteins are usually globular in shape. Changes in temperature and pH impair the intra- and intermolecular interactions that bind proteins in their secondary and tertiary structures.Which of the following can impact the structure of a protein?
The main forces that affect structure are electrostatic forces, hydrogen bonding forces, hydrophobic forces, and disulfide bonds. Each of these affect protein structure in different ways. The electrostatic forces are when two like charges are repelled or two opposite charges are attracted.
How are proteins built up into a functional structure?Proteins are built as chains of amino acids, which then fold into unique three-dimensional shapes. Bonding within protein molecules helps stabilize their structure, and the final folded forms of proteins are well-adapted for their functions.
Article first time published onWhy do channel proteins often fold with nonpolar R groups?
Why do channel protein often fold with non-polar R groups to the channel’s exterior and polar R groups to its interior? The interior of a plasma membrane is hydrophobic. … The loss of shape disrupts the protein’s active site and thus its ability to carry out its biological functions.
What makes an R group hydrophobic?
There are about 10 nonpolar amino acids with R-groups that are not stable when in contact with water. They are hydrophobic. About 5 amino acids have polar side chains, R-groups which do not ionize or become positively or negatively charged. These R-groups are neither strongly hydrophilic nor hydrophobic.
How do R groups differ?
Each of the 20 amino acids commonly found in proteins has an R (reactive) group with its own distinctive chemistry. R groups differ in their size, polarity, charge and bonding potentials.
What is R in amino acid structure?
Every amino acid also has another atom or group of atoms bonded to the central atom known as the R group. This R group, or side chain, gives each amino acid proteins specific characteristics, including size, polarity, and pH.
What does R group stand for in amino acids?
R groups are the radical, meaning it is the variable part of amino acids. Amino acids have three main groups, the carboxyl groups, the amino group and a radical which can change. They are important because their properties determine protein structure and function.
Are R groups the same as functional groups What does having a different R group do to each amino acid?
There are 20 common amino acids found in proteins, each with a different R group (variant group) that determines its chemical nature. … The chemical properties associated with the whole collection of individual functional groups gives each amino acid R group unique chemical potential.
How does low temperature affect protein structure?
Proteins undergo both cold and heat denaturation, but often cold denaturation cannot be detected because it occurs at temperatures below water freezing. Proteins undergoing detectable cold as well as heat denaturation yield a reliable curve of protein stability.
How does high temperature affect protein structure?
The denatured protein has the same primary structure as the original, or native, protein. The weak forces between charged groups and the weaker forces of mutual attraction of nonpolar groups are disrupted at elevated temperatures, however; as a result, the tertiary structure of the protein is lost.
What levels of protein structure are affected by chemically straightening hair?
What level(s) of protein structure are affected by chemically straightening hair? Tertiary and quaternary structures. Why doesn’t hair stay straight forever after being chemically straightened?
Why are some R groups nonpolar?
They all have basically similar charges they have there and what other bond is the Carbon. Since they all have the Hydrogens there, they all have the same charge, so they’re non-polar.
What characteristic would the R groups of amino acids have if they are located?
What characteristic would the R groups of amino acids have if they are located within the interior of biological membranes? The R groups would hydrophobic.
What are the different classifications of R groups of amino acids?
Based on type of functional group (R group) present amino acids are classified as: Aliphatic, aromatic, acidic, basic, acid amide, sulfur and cyclic amino acids. Based on characteristic of functional group amino acids are classified as: polar and non-polar amino acids.
What factors affect protein stability?
Many factors affect the process of protein folding, including conformational and compositional stability, cellular environment including temperature and pH, primary and secondary structure, solvation, hydrogen bonding, salt bridges, hydrophobic effects, van der Waals (vdW) forces, ligand binding, cofactor binding, ion …
How does the primary structure of a protein affect the other structural levels?
The primary structure of a protein is defined by the sequence of amino acid residues. It is this sequence that lays the foundation for all other higher levels of structures in a protein. Secondary structure is defined by the hydrogen bonding between the carboxyl and amino backbone of the amino acids.
What causes changes in protein structure claim and evidence?
The unique sequence for every protein is ultimately determined by the gene encoding the protein. A change in nucleotide sequence of the gene’s coding region may lead to a different amino acid being added to the growing polypeptide chain, causing a change in protein structure and function.
What is the relationship between protein structure and function?
Protein structure sets the foundation for its interaction with other molecules, which ultimately determines its function. Proteins fold up into specific shapes according to the sequence of amino acids in the polypeptide chain.
How do amino acids affect protein structure?
The sequence of amino acids of a protein determines protein shape, since the chemical properties of each amino acid are forces that give rise to intermolecular interactions to begin to create secondary structures, such as α-helices and β-strands.
What causes the changes in protein structure through the three or four levels of structure?
What causes the changes in protein structure through the three or four levels of structure? … The primary structure is based on the bonds between individual amino acids while the secondary structure is based on the formation of alpha and beta pleated sheets.
How is protein structure determined?
Currently, the main techniques used to determine protein 3D structure are X-ray crystallography and nuclear magnetic resonance (NMR). In X-ray crystallography the protein is crystallized and then using X-ray diffraction the structure of protein is determined.
Why is protein structure important?
Having a protein structure provides a greater level of understanding of how a protein works, which can allow us to create hypotheses about how to affect it, control it, or modify it. For example, knowing a protein’s structure could allow you to design site-directed mutations with the intent of changing function.
What is protein structure?
Protein structure is the three-dimensional arrangement of atoms in an amino acid-chain molecule. Proteins are polymers – specifically polypeptides – formed from sequences of amino acids, the monomers of the polymer. … Very large protein complexes can be formed from protein subunits.