How do you find the degrees of unsaturation in nitrogen
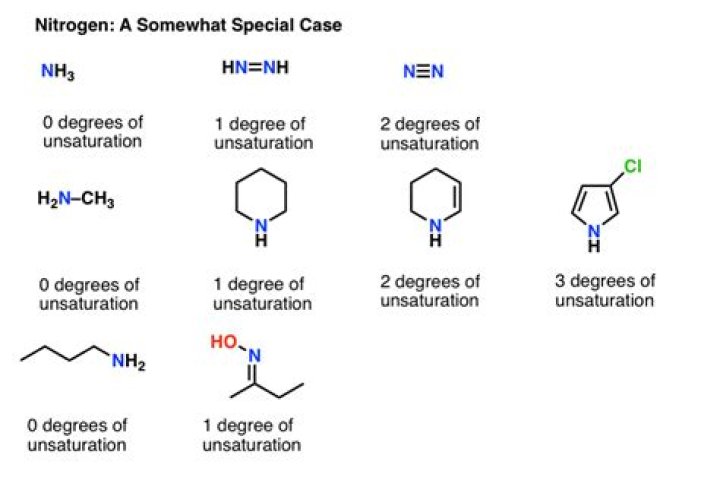
Although, nuclear magnetic resonance (NMR) and infrared radiation (IR) are the primary ways of determining molecular structures, calculating the degrees of unsaturation is useful information since knowing the degrees of unsaturation make it easier for one to figure out the molecular structure; it helps one double-check …
Why do we calculate degree of unsaturation?
Although, nuclear magnetic resonance (NMR) and infrared radiation (IR) are the primary ways of determining molecular structures, calculating the degrees of unsaturation is useful information since knowing the degrees of unsaturation make it easier for one to figure out the molecular structure; it helps one double-check …
How many degrees of unsaturation do molecules have?
The degrees of unsaturation in a molecule are additive — a molecule with one double bond has one degree of unsaturation, a molecule with two double bonds has two degrees of unsaturation, and so forth.
What is the formula for unsaturated?
Alkenes have the general formula CnH2n, and are examples of unsaturated hydrocarbons. The presence of a double bond compound’s structure causes there to be two fewer hydrogen atoms in the molecule than there are in an alkane with the same number of carbon atoms.What is meant by degree of unsaturation?
In the analysis of the molecular formula of organic molecules, the degree of unsaturation (also known as the index of hydrogen deficiency (IHD), double bond equivalents, or unsaturation index) is a calculation that determines the total number of rings and π bonds. …
What does degree of unsaturation of 3 mean?
0. 1. Remember, the degrees of unsaturation only gives the sum of double bonds, triple bonds and/or rings. For instance, a degree of unsaturation of 3 can contain 3 rings, 2 rings+1 double bond, 1 ring+2 double bonds, 1 ring+1 triple bond, 1 double bond+1 triple bond, or 3 double bonds. Example: Benzene.
What is the degree of unsaturation present in DDT?
Answer: Interpretation: The degree of unsaturation in DDT, C14H9Cl5 is to be calculated and to draw five possible structures with this formula. Concept introduction: The degree of unsaturation is equal to the number of rings and/or multiple bonds present in the molecule.
Which formula represents unsaturated hydrocarbons?
Unsaturated hydrocarbons have double and/or triple bonds between carbon atoms. Those with double bond are called alkenes and have the general formula CnH2n (assuming non-cyclic structures). Those containing triple bonds are called alkynes and have general formula CnH2n-2.What is degree of unsaturation of cube?
The degree of unsaturation determines the double bond equivalent. … DBE or double bond equivalent LU or level of unsaturation is the number of unsaturation’s present in an organic molecule. The term unsaturated means a double bond or a ring system.
How many degrees of unsaturation are present in a molecule with formula c10h16?Here the given molecular formula contain 10 carbon atom i.e has six fewer hydrogens. The degree of unsaturation is found to be 3.
Article first time published onWhat is the degree of unsaturation in benzene?
Benzene has 6 carbon atoms and 4 degrees of unsaturation (1 ring and 3 double bonds).
Do isomers have same degree of unsaturation?
Identifying Degrees of Unsaturation Every ring or pi bond in a compound is said to represent one degree of unsaturation. … Each of the following compounds are isomers of C5H7 and contain two degrees of unsaturation.
What is the degree of saturation?
The degree of saturation is defined as the ratio of actual humidity ratio to the humidity ratio of a saturated mixture at the same temperature and pressure.
What is degree of unsaturation Class 11?
Unsaturation means the presence of double bond or triple bond. It is used to calculate the number of rings and pi bonds present in a molecule. Complete step by step answer: … Total degree of unsaturation is $3+6+2=11$ .
How do you know if a molecule is unsaturated?
A molecule belonging to either the alkene or alkyne homologous series is an unsaturated hydrocarbon. The carbon atoms can be arranged in straight chains, branched chains, or in rings (cyclic compounds), as long as there is at least 1 double bond (C=C) and/or 1 triple bond (C≡C) the molecule will be unsaturated.
What does degree of unsaturation of 4 mean?
Example: Benzene (4 Degrees Of Unsaturation) For instance, thus the molecular formula C6H6 (4 degrees of unsaturation) is satisfied by molecules with. 4 pi bonds. 3 pi bonds and a ring (benzene) two pi bonds and two rings (the very unstable Dewar Benzene, synthesized in 1963)
How many degrees of unsaturation is a triple bond?
Every ring and multiple bond in a compound is a ‘degree of unsaturation’. Each ring and double bond counts as one degree of unsaturation. Triple bonds count as two degrees of unsaturation.
What does a degree of unsaturation of 0 mean?
Each degree of unsaturation refers to a decrease in two hydrogens in the molecule, as a result of the presence of a pi bond or a ring. A degree of unsaturation of 0 means that the molecule follows the formula for an acyclic alkane (CnH2n+2). … Degrees of unsaturation can help us determine how an alkane molecule will act.
What is the chemical formula of 4 nonene?
4-Nonene | C9H18 – PubChem.
How do you find the degree of a carbon atom?
Identify the carbon in question and place your pencil on that carbon atom. Then examine how many lines emanate from the carbon atom that attach to another carbon—meaning the line does not lead to a hydrogen, oxygen or halogen. The number of lines that stem from that carbon represents the degree of substitution.
What is 1 degree 2 degree and 3 degree alcohol?
In a secondary (2°) alcohol, the carbon atom with the -OH group attached is joined directly to two alkyl groups. In a tertiary (3°) alcohol, the carbon atom holding the -OH group is attached directly to three alkyl groups.
What is a 1 degree hydrogen?
A primary (1°) hydrogen is a hydrogen atom residing on a primary carbon in an organic species.
What is DBE of cubane?
One DBE = one ring or one double bond. Complete step by step answer: Cubane (C8H8) is a synthetic hydrocarbon molecule and it consists of eight carbon atoms. These carbon atoms are arranged at the corners of a cube and one hydrogen atom is attached to each carbon atom.
What is DU of cubane?
Degree of unsaturation= 210=5.
Can degree of unsaturation be in fraction?
The answer is at hand. For example, if the saturation or unsaturation ratio were 1/2 or 3/4, you could express fractions as decimals and get the correct answer. 1/2 can be expressed to the nearest decimal point.
What is a saturated and unsaturated hydrocarbon?
Saturated Hydrocarbons — contain only carbon- carbon single bonds. • Unsaturated Hydrocarbons — contain carbon- carbon double or triple bonds (more hydrogens can be added).
Which is the unsaturated hydrocarbon?
Unsaturated hydrocarbons are hydrocarbons that have double or triple covalent bonds between adjacent carbon atoms. … The configuration of an unsaturated carbons include straight chain, such as alkenes and alkynes, as well as branched chains and aromatic compounds.
Are aromatics saturated or unsaturated?
Aromatic hydrocarbons are a special class of unsaturated hydrocarbon based on a six carbon ring moiety called benzene. The saturated hydrocarbon cyclohexane is transformed into the aromatic hydrocarbon benzene by adding three alternating carbon–carbon double bonds, as shown in Fig.
How do you calculate degrees of freedom?
The most commonly encountered equation to determine degrees of freedom in statistics is df = N-1. Use this number to look up the critical values for an equation using a critical value table, which in turn determines the statistical significance of the results.
What is the degree of unsaturation for c5h9no2?
Thus, C5H9N has two degrees of unsaturation and could contain one of the following: a) a triple bond; b) two double bonds or c) one double bond and a ring.