How do you find which isotope is more abundant
To determine the most abundant isotopic form of an element, compare given isotopes to the weighted average on the periodic table. For example, the three hydrogen isotopes (shown above) are H-1, H-2, and H-3. The atomic mass or weighted average of hydrogen is around 1.008 amu ( look again at the periodic table).
How do you find the abundance of an isotope?
The equation can be set up as a percent or as a decimal. As a percent, the equation would be: (x) + (100-x) = 100, where the 100 designates the total percent in nature. If you set the equation as a decimal, this means the abundance would be equal to 1. The equation would then become: x + (1 – x) = 1.
Is the heavier isotope more abundant?
The heavier isotope is a more abundant one, so always relate that average mass to the two isotopes. If the mass is closer to the heavier isotope, it means that one is more abundant.
What makes an isotope abundant?
Atoms that have the same number of protons but different numbers of neutrons are known as isotopes. … The relative abundance of an isotope is the percentage of atoms with a specific atomic mass found in a naturally occurring sample of an element.Why is one isotope more abundant than another?
Isotopes are atoms of the same element that have a different number of neutrons. … Certain isotopes are more abundant in some materials than others since some physical and chemical processes “prefer” one isotope over another.
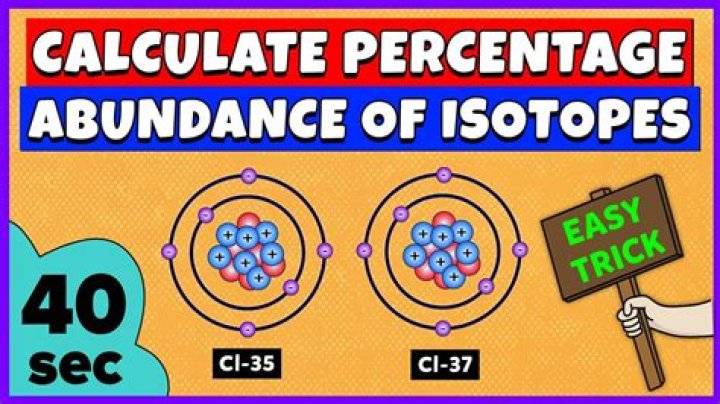
Which isotope of chlorine is more abundant?
This is because the chlorine-35 isotope is much more abundant than the chlorine-37 isotope. The table shows the mass numbers and abundances of naturally-occurring copper isotopes.
Which isotope is more abundant in nitrogen?
Nitrogen has two stable isotopes, 14N and 15N (atomic masses of 14 and 15, respectively). 14N is the more abundant of the two, comprising 99.63% of the nitrogen found in nature.
Which isotope of lead is likely to be the most abundant?
The relative abundances of the four stable isotopes are approximately 1.5%, 24%, 22%, and 52.5%, combining to give a standard atomic weight (abundance-weighted average of the stable isotopes) of 207.2(1). Lead is the element with the heaviest stable isotope, 208Pb.Which lithium isotope is more abundant?
Lithium-7 is by far the most abundant isotope of lithium, making up between 92.2% and 98.1% of all terrestrial lithium. A lithium-7 atom contains three protons, four neutrons, and three electrons.
How is relative abundance calculated?It is usually measured as the number of individuals found per sample. … Relative species abundance is calculated by dividing the number of species from one group by the total number of species from all groups.
Article first time published onWhat is the most abundant isotope of manganese?
The isotopes of manganese range in atomic weight from 44 u (44Mn) to 69 u (69Mn). The primary decay mode before the most abundant stable isotope, 55Mn, is electron capture and the primary mode after is beta decay.
Which isotope is more abundant n14 or n15?
N-14 is more abundant in nature because it is closest to the average atomic mass of 14.007.
Which silver isotope is more abundant?
Naturally occurring silver (47Ag) is composed of the two stable isotopes 107Ag and 109Ag in almost equal proportions, with 107Ag being slightly more abundant (51.839% natural abundance).
What does abundant mean in chemistry?
In a chemical reaction, a reactant is considered to be in abundance if the quantity of that substance is high and virtually unchanged by the reaction. … In this case, should the reaction go to completion, the quantity of the reactant in excess will have halved.
Which isotope is more abundant on earth boron 10 or boron 11?
The atomic mass of boron is 10.81 u. And 10.81 u is a lot closer to 11u than it is to 10u, so there must be more of boron-11. Where u is the unit for atomic mass and x is the proportion of boron-10 out of the total boron abundance which is 100%. And thus the abundance of boron-11 is roughly 81%.
How do you calculate percent abundance of chlorine?
- 34.96 × x + 36.95 × y = 35.43.
- 34.96 × x + 36.95 × (1 – x) = 35.43 (since, y = 1 – x)
- Percentage abundance of Cl-35 = 0.7595 × 100 = 75%
- Percentage abundance of Cl-37 = 0.2405 × 100 = 24.05%
Are lighter isotopes more abundant?
Isotopes have the same number of protons but differ in their number of neutrons, resulting in different masses. The lighter form is generally the more common one (Hobson & Wassenaar 2008).
What does percent abundance mean?
Percent abundance is defined as the percent value of the number of isotopes available in nature for a given element.
How do you find the fractional abundance of two isotopes?
Divide the relative abundance of any one isotope by the total number of isotopes to calculate the fractional abundance in decimal form. In the example, the isotope measurement of 200 would be divided by 300, which results in a fractional abundance of 0.667.
What is the abundance of nitrogen?
Nitrogen is pronounced as NYE-treh-gen. History and Uses: Nitrogen was discovered by the Scottish physician Daniel Rutherford in 1772. It is the fifth most abundant element in the universe and makes up about 78% of the earth’s atmosphere, which contains an estimated 4,000 trillion tons of the gas.
How do u find average atomic mass?
To calculate the average atomic mass, multiply the fraction by the mass number for each isotope, then add them together. Whenever we do mass calculations involving elements or compounds (combinations of elements), we always use average atomic masses.
Do most elements exist as a mixture of two or more isotopes?
REMEMBER: Atoms of the same chemical element do not always have the same mass because, although the number of protons in the nucleus is the same for all atoms of the same element, the number of neutrons is not. Most elements as they occur naturally on earth are mixtures of several isotopes.
Which sodium isotope is most abundant?
IsotopeDecayabundancemode22Natraceβ+23Na100%stable24Natraceβ−
Is 37cl or 35Cl more abundant?
35Cl is more abundant The average atomic mass of Cl is 35.45 amu.
What is the most abundant form of chlorine?
Sodium chloride (salt) is the most common compound of chlorine and occurs in large quantities in the ocean.
Why is Li 7 more abundant?
Lithium-7. Lithium-7 is the second stable lithium isotope and the most abundant. … Because of lithium’s nuclear properties, it is less abundant in the universe than helium, beryllium, carbon, nitrogen, or oxygen.
How do you determine isotopes?
Subtract the atomic number (the number of protons) from the rounded atomic weight. This gives you the number of neutrons in the most common isotope. Use the interactive periodic table at The Berkeley Laboratory Isotopes Project to find what other isotopes of that element exist.
What is the natural abundance of each boron isotope?
Boron has two natural stable isotopes, 10B and 11B, with an average abundance of approximately 19.9% and 80.1% respectively.
How can you predict the mass of the most common isotope?
To determine the most abundant isotopic form of an element, compare given isotopes to the weighted average on the periodic table. For example, the three hydrogen isotopes (shown above) are H-1, H-2, and H-3. The atomic mass or weighted average of hydrogen is around 1.008 amu ( look again at the periodic table).
What makes an isotope more stable?
The neutron/proton ratio and the total number of nucleons determine isotope stability. … This attractive force comes from the neutrons. More protons in the nucleus need more neutrons to bind the nucleus together. The graph below is a plot of the number of neutrons versus the number of protons in various stable isotopes.
Why is PB 208 the most stable?
Some consider lead to be the most stable element of all because of its high atomic number. Lead has three stable isotopes and one unstable isotope. All of them are natural. … The stable isotopes Pb-206, 207 and 208 are generated from isotopes of the elements thorium and uranium.



