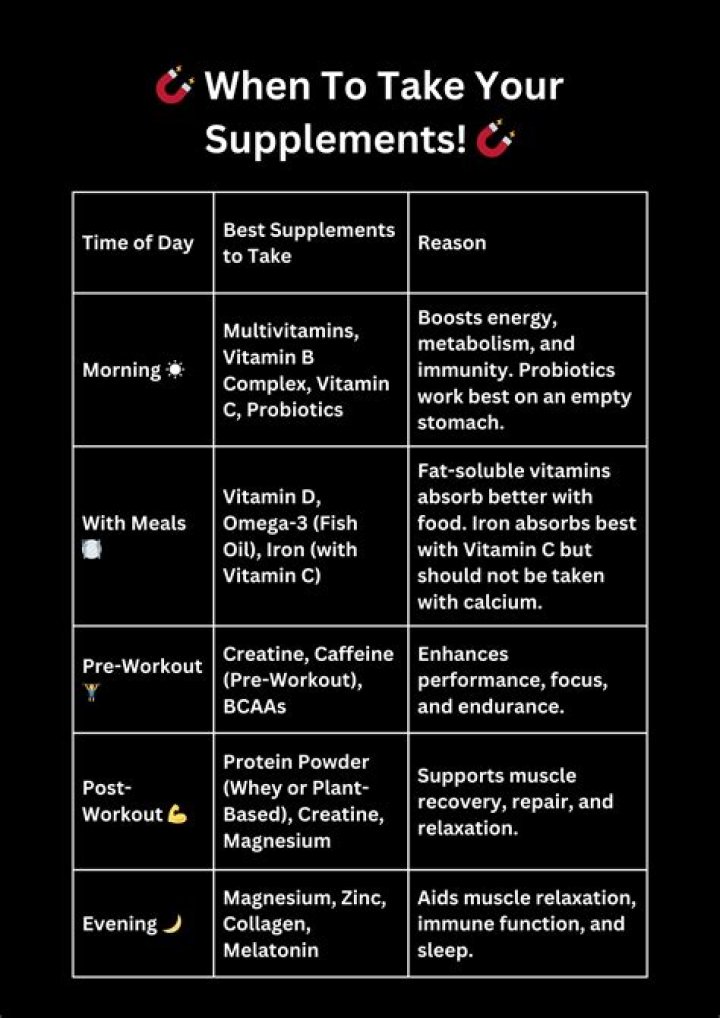
How does a noncompetitive inhibitor reduce an enzymes activity
How does a noncompetitive inhibitor reduce an enzyme’s activity? The inhibitor binds to the enzyme in a location other than the active site, changing the shape of the active site. … No change in enzyme activity would be observed.
How does a noncompetitive inhibitor reduce an enzyme's activity? - Google Search
How do non-competitive inhibitors work? –The inhibitor changes the conformation of the enzyme. The substrate can no longer bind, or it may be able to bind but the active site cannot catalyse the reaction, or catalyses it at a slower rate.
How does a non-competitive inhibitor inhibit binding of a substrate to an enzyme quizlet?
A noncompetitive inhibitor binds to the enzyme away from the active site, altering the shape of the enzyme so that even if the substrate can bind, the active site functions less effectively.
How do non-competitive inhibitors affect the rate of reaction?
The noncompetitive inhibitor slows down the reaction rate, i.e. the rate of the product formation is less with inhibitor present than with inhibitor absent. This means that the active site is modified, but not disabled, by the presence of the inhibitor.Where does a noncompetitive inhibitor bind quizlet?
A non-competitive inhibitor binds in two places: either on the enzyme or on the enzyme-substrate complex. It’s important to note that it does not bind to the active site.
How does an inhibitor decrease a reaction rate?
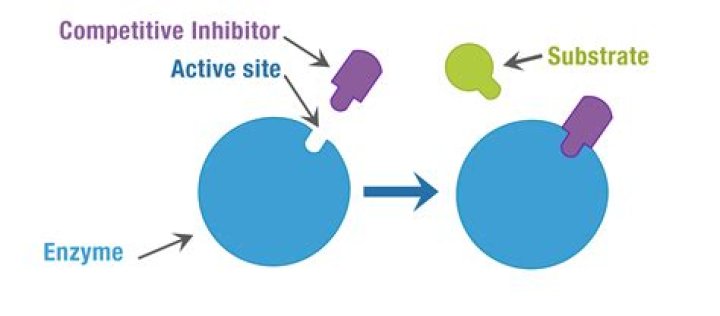
Enzyme Inhibitors reduce the rate of an enzyme catalysed reaction by interfering with the enzyme in some way. … Therefore less substrate molecules can bind to the enzymes so the reaction rate is decreased. Competitive Inhibition is usually temporary, and the Inhibitor eventually leaves the enzyme.
Why does Vmax decrease in noncompetitive inhibition?
A third type of enzymatic inhibition is that of uncompetitive inhibition, which has the odd property of a reduced Vmax as well as a reduced Km. The explanation for these seemingly odd results is due to the fact that the uncompetitive inhibitor binds only to the enzyme-substrate (ES) complex.
How does a noncompetitive inhibitor inhibit binding of a substrate to an enzyme *?
In noncompetitive inhibition, the inhibitor binds at an allosteric site separate from the active site of substrate binding. … Binding of the inhibitor to the enzyme or enzyme-substrate complex inactivates the enzyme, disallowing the production of its end product.Does a noncompetitive inhibitor decrease the rate of an enzyme catalyzed reaction?
A noncompetitive inhibitor is a type of regulator that binds to an enzyme at a place other than the active site. Upon binding to the noncompetitive inhibitor, the enzyme changes shape so that normal substrate binding cannot occur. Noncompetitive inhibitors decrease the rates of enzyme-catalyzed reactions.
Which of the following occurs in noncompetitive inhibition?Noncompetitive inhibition occurs when an inhibitor binds to the enzyme at a location other than the active site. In some cases of noncompetitive inhibition, the inhibitor is thought to bind to the enzyme in such a way as to physically block the normal active site.…
Article first time published onHow does a non-competitive inhibitor work quizlet?
How do non-competitive inhibitors work? Inhibitors bond to enzyme at a location other than the active site, the allosteric site. The binding of the inhibitor causes the tertiary structure of the enzyme to change, meaning the active site changes shape.
Which statement best describes the action of a noncompetitive inhibitor?
Which statement best describes the action of a non-competitive inhibitor? A non-competitive inhibitor binds outside of the active site and alters the shape of the enzyme.
Which statement is correct about noncompetitive enzyme inhibitors?
Correct answer: Noncompetitive inhibition is characterized by a decrease in the maximum velocity (or efficacy) of an enzyme. Noncompetitive inhibitors bind irreversibly to the enzyme and prevent the substrate-enzyme activity. This decreases the efficacy of the enzyme.
What is true about competitive and noncompetitive inhibitors quizlet?
Competitive inhibitors bind to the active site of an enzyme while noncompetitive inhibitors bind to an enzyme away from the active site.
How does inhibitor affect Vmax?
Vmax is the maximum velocity of the enzyme. Competitive inhibitors can only bind to E and not to ES. They increase Km by interfering with the binding of the substrate, but they do not affect Vmax because the inhibitor does not change the catalysis in ES because it cannot bind to ES.
How does noncompetitive inhibition affect Km and Vmax?
For the competitive inhibitor, Vmax is the same as for the normal enzyme, but Km is larger. For the noncompetitive inhibitor, Vmax is lower than for the normal enzyme, but Km is the same.
How does inhibitors affect enzymatic activity?
By binding to enzymes’ active sites, inhibitors reduce the compatibility of substrate and enzyme and this leads to the inhibition of Enzyme-Substrate complexes’ formation, preventing the catalysis of reactions and decreasing (at times to zero) the amount of product produced by a reaction.
Do non-competitive inhibitors denature enzymes?
Some non-competitive inhibitors are irreversible and permanent because they denature the enzymes effectively. However, there are also non-competitive inhibitors reversibly and non-permanently stops the enzymatic reaction, which are vital in controlling metabolic functions in organisms.
Does inhibitor increase or decrease activation energy?
The binding of this allosteric inhibitor changes the conformation of the enzyme and its active site, so the substrate is not able to bind. This prevents the enzyme from lowering the activation energy of the reaction, and the reaction rate is reduced.
Is noncompetitive inhibition same as mixed inhibition?
If the ability of the inhibitor to bind the enzyme is exactly the same whether or not the enzyme has already bound the substrate, it is known as a non-competitive inhibitor. … In mixed inhibition, the inhibitor binds to an allosteric site, i.e. a site different from the active site where the substrate binds.
What is the difference between noncompetitive and uncompetitive inhibition?
Non-competitive inhibitors bind equally well to the enzyme and enzyme–substrate complex. Uncompetitive inhibitors bind only to the enzyme–substrate complex. These different inhibitory mechanisms yield different relationships between the potency of the inhibitor and the concentration of the substrate.
How does the competitive inhibitor at low substrate concentrations alter the affinity of the enzyme for the substrate?
The reason is that the competitive inhibitor is reducing the amount of active enzyme at lower concentrations of substrate. When the amount of enzyme is reduced, one must have more substrate to supply the reduced amount of enzyme sufficiently to get to Vmax/2.
How is non-competitive inhibition reversed?
Non-competitive inhibition [Figure 19.2(ii)] is reversible. The inhibitor, which is not a substrate, attaches itself to another part of the enzyme, thereby changing the overall shape of the site for the normal substrate so that it does not fit as well as before, which slows or prevents the reaction taking place.
How does inhibition by a competitive inhibitor differ from inhibition by a noncompetitive inhibitor quizlet?
How does inhibition of an enzyme-catalyzed reaction by a competitive inhibitor differ from inhibition by a noncompetitive inhibitor? Competitive inhibitors bind to the active site of the enzyme; noncompetitive inhibitors bind to a different site.
How does a competitive inhibitor differ from a noncompetitive inhibitor quizlet?
-COMPETITIVE inhibition= inhibitor & substrate both bind to the active site of the enzyme. … -NONCOMPETITIVE inhibition= inhibitor & substrate bind to different sites. binding of an inhibitor distorts the enzyme, inhibiting substrate binding or reducing catalytic activity.
Which of the following statements is true about noncompetitive inhibitors?
Which of the following statements is true about uncompetitive inhibitors? Explanation: They bind non-covalently at a site distinct from the substrate active site. They decrease the measured Vmax and also apparent Km. … Explanation: DIPF, Penicillin and Iodoacetamide are irreversible inhibitors.
Do nonprotein cofactors alter the substrate specificity of enzymes?
Enzyme function is independent of physical and chemical environmental factors such as pH and temperature. … Nonprotein cofactors alter the substrate specificity of enzymes.
Which of the following best describes competitive inhibitor?
Which of the following best describes the function of competitive inhibitors? Explanation: Competitive inhibitors bind to the substrate binding site of an enzyme and have the following effect: Increase , No change in .
What is true about competitive and noncompetitive inhibitors?
The competitive inhibitor binds to the active site and prevents the substrate from binding there. The noncompetitive inhibitor binds to a different site on the enzyme; it doesn’t block substrate binding, but it causes other changes in the enzyme so that it can no longer catalyze the reaction efficiently.
Which of the following statements describes a non kinetic enzyme assay?
Which of the following statements describes a non kinetic enzyme assay? … An enzyme will accelerate the rate of a reaction, reducing the time required to reach equilibrium. The concentration of reactants and products at equilibrium will be the same with or without the enzyme.
Which of the following environments or actions does not affect enzyme activity?
Which of the following environments or actions would NOT affect the rate of an enzyme reaction? None of the listed responses is correct. Correct. Changes in temperature, substrate concentration, and pH are all likely to affect enzyme activity.