How does HClO dissociate in water
Hypochlorous acid (HClO) is formed when chlorine dissolves in water, and it quickly dissociates into hypochlorite (ClO-) and hydronium (H3O+) ions with limited stability.
Why does HClO dissociate in water?
Hypochlorous acid has a partial dissociation in water because it is a weak acid and its acid dissociation constant (ka) is: 3.0 × 10-8. In water, hypochlorous acid occurs both as a chemical chlorine solution and as water.
Is HClO aqueous?
Hypochlorous acid is a weak acid which is typically formed when chlorine dissolves in water. It is referred to by several other names, such as: chloric acid, chloranol, hydrogen hypochlorite and chlorine hydroxide. … Physical properties: Hypochlorous acid only exists as an aqueous solution.
What happens to HClO in water?
Hypochlorous Acid (HCIO) is a weak acid that is formed when elemental Chlorine is dissolved in water and maintained within a defined pH range (between 3-6.5pH). Outside of this range HCIO will partially dissociates, forming hypochlorites (CIO-). … Uniquely this makes HCIO one of the only non-toxic disinfection agents.Does hypochlorite dissociate in water?
When sodium hypo chlorite dissolves in water, two substances form, which play a role in for oxidation and disinfection. These are hypochlorous acid (HOCl) and the less active hypochlorite ion (OCl-). The pH of the water determines how much hypochlorous acid is formed.
Why is HOCl unstable?
It is a weak acid. The chlorine atom possesses oxidation state +3 in this acid. The pure substance is unstable and disproportionates to hypochlorous acid (Cl oxidation state +1) and chloric acid (chlorine oxidation state +5). Chlorite salts such as sodium chlorite are stable conjugate bases derived from this acid.
Does HClO dissociate completely in water?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−. HClO and ClO− are oxidizers, and the primary disinfection agents of chlorine solutions.
What is the concentration of HClO?
The active chemical is HClO, hypochlorous acid, which passes through the cell wall and oxidizes or “burns up” the interior of the bacterium. Christie read that the concentration of HClO should be kept above 1.0 ppm.Can I drink hypochlorous acid?
One solution is a sanitizer that at 50 parts per million is more effective than chlorine bleach at 200 parts per million, yet it’s so non-toxic you can drink it.
Is HClO polar?Hypochlorous acid is HOCl. Here Oxygen atom is sp3 hybridised. Hence, it has bent shape around oxygen owing to presence of two lone pairs. This causes net Dipole moment (0.37 D) and hence it is a polar molecule.
Article first time published onIs HOCl an element?
Hypochlorous acid is a chlorine oxoacid with formula HOCl; a weak, unstable acid, it is the active form of chlorine in water. It has a role as a human metabolite, an EC 3.1. 1.7 (acetylcholinesterase) inhibitor and an EC 2.5.
Is HOCl a bleach?
Chlorine bleach is a solution of sodium hypochlorite in water, which forms hypochlorous acid – HOCl. Hypochlorous acid is an oxidizing agent that attacks and destroys molecules in bacteria and viruses. Recent work has shown that hypochlorous acid causes proteins in bacteria to unfold – a process called denaturing.
How do you make HOCl?
Hypochlorous acid is formed when chlorine dissolves in water. One manufacturing method involves the electrochemical activation of a saturated salt solution (e.g., brine) to form HOCl. Another production method involves the disproportionation of chlorine gas in alkaline solutions.
How do acids dissociate in water?
Figure 2.4.1 (a) In aqueous (watery) solution, an acid dissociates into hydrogen ions (H+) and anions. Every molecule of a strong acid dissociates, producing a high concentration of H+. (b) In aqueous solution, a base dissociates into hydroxyl ions (OH–) and cations.
What completely dissociates in water?
For all practical purposes, strong acids completely dissociate in water. That is the definition: A strong acid is an acid that completely dissociates in water.
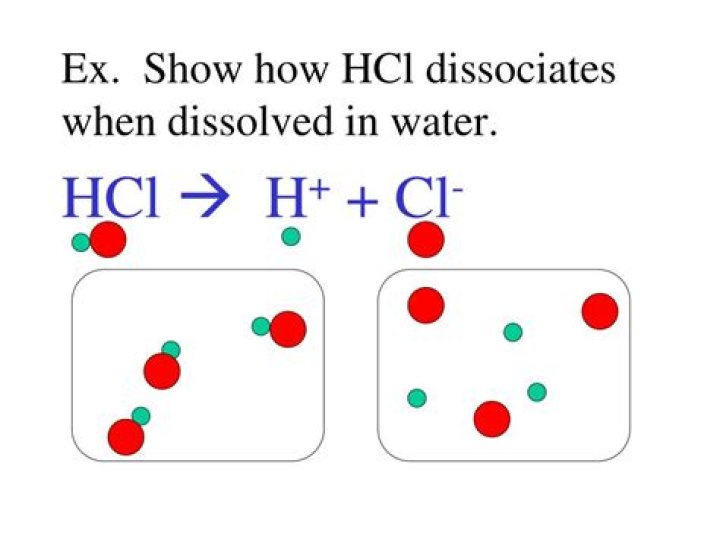
How would HCl dissociate in water?
HCl will dissociate in water because HCl is also a polar molecule (Cl is slightly negative and H is slightly positive). The Cl will be attracted to the slightly positive H in water and the H will be attracted to the slightly negative O in water.
How stable is HOCl?
Hypochlorous acid (HOCl) solution (200 ppm, pH 6) was prepared and evaluated for their stabilities and microbicidal activities. We demonstrated that HOCl is unstable against ultraviolet (UV) light, sunshine, contact with air, and elevated temperature (≧25℃).
How long is HOCl stable?
HOCl from Monarch Chemicals, however, has a shelf-life of up to 24 months with nothing new added – just improved stabilisation during the manufacturing process.
What is the pH of HOCl?
In aqueous solution, HOCl is the predominant species at the pH range of 3 to 6. At pH values less than 3.5, the solution exists as a mixture of chlorine in aqueous phase, chlorine gas, trichloride (Cl3−), and HOCl.
Can you make electrolyzed water?
hide Authority controlOtherMicrosoft Academic
Can I drink electrolyzed water?
Electrolyzed Water Is Non-Toxic It is a non-toxic liquid that spells doom only for harmful germs, not natural human body processes. Empowered Water is a gentle substance that doesn’t have any harmful elements. You can use it for just about anything without worrying about toxic traces or chemical films.
Do hypochlorous acid generators work?
Hypochlorous acid water generator highly effective in removing bacteria and deodorizing. … It is produced by electrolyze of water and salt, without any need of hazardous chemical materials, and is immediately effective against a wide range of bacteria and viruses, including influenza and noroviruses.
What is the dissociation equation for HClO?
Problem: Hypochlorous acid, HClO, is a weak acid. The following is the equilibrium equation for its reaction with water:HClO (aq) + H2O (l) ⇌ H3O+ (aq) + ClO- (aq) Ka = 2.9 x 10-8What is the hydroxide ion concentration, [OH-], in a 1.98 M HClO solution?
Can I make hypochlorous acid at home?
After getting us all hooked on the wonders of Hypochlorous acid, I discovered that it’s easily made at home. I found several machines on Amazon to make it easily at home. … The basic premise for all of them is similar: water, salt and vinegar are combined with an electrical charge to create the HOCI solution.
Why is HClO a weak acid?
These acids are the only ones that fully dissociate in water. All other acids are weak acids. Since HClO is not one of these seven, and there is no -OH group present as there is in bases, HClO is a weak acid. Another way to verify that this is an acid instead of a base is by looking at the ion it dissociates into.
Does HOCl have dipole dipole forces?
hclo intermolecular forces have dipole-dipole force. it is windly attack between positive end to negative end. Hello, reders welcome to another fresh article on “textilesgreen.in” today we will discuss about hclo intermolecular forces. and its types.
Does HOCl have a dipole moment?
The dipole moment of HOCl in vOH=4 – ScienceDirect.
Does HOCl have hydrogen bonding?
► The Cl atom of HOCl molecule can act as a hydrogen-bond or halogen-bond acceptor, or as a halogen-bond donor. ► The HOCl molecule can dimerize to form six different complexes but not at room temperature.
What does HOCl?
HOCl attacks invading pathogens, breaking down the cell walls before destroying unhealthy invaders. The antimicrobial acid is lethally effective in carrying out its protective mission.
Does HOCl have a resonance structure?
Hypochlorous acid has two resonance structures: one with oxygen as the central atom, and the other with chlorine as the center.
What is the hybridization of HClO?
Hybridisation of HClO is sp3.