How does lipid solubility affect drug absorption
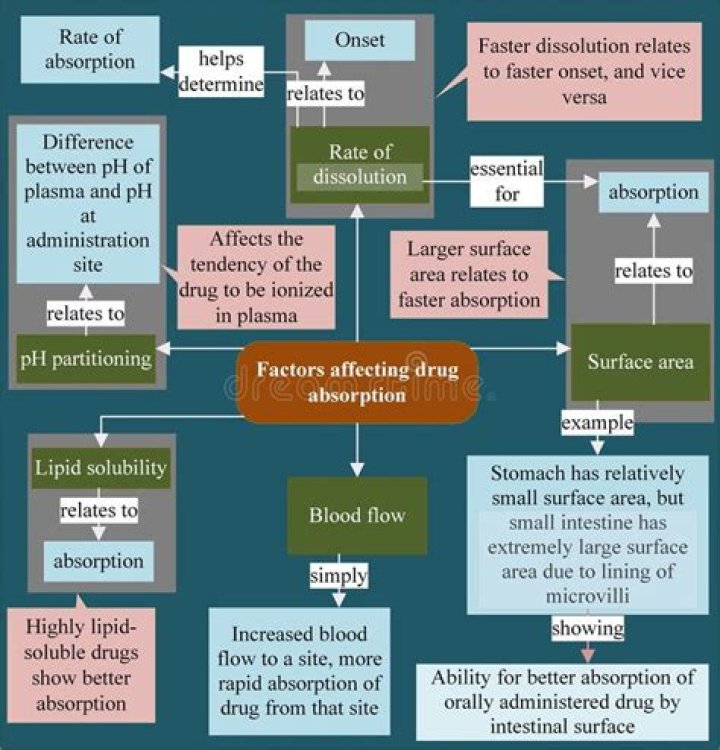
Absorption. Both the lipid solubility of the drug and the pH of the gastric tissues affect drug absorption from the GI tract. Lipid-soluble drugs are absorbed more rapidly than non–lipid-soluble drugs. Gastric fluid has a pH of approximately 1.4.
How does lipid solubility affect drug distribution?
Drug absorption depends on the lipid solubility of the drug, its formulation and the route of administration. A drug needs to be lipid soluble to penetrate membranes unless there is an active transport system or it is so small that it can pass through the aqueous channels in the membrane.
How lipid solubility of the drug is determined?
A substance will become more lipid soluble in a solution with a pH similar to its own pH. A weak base is more lipid-soluble in an alkaline solution. A weak base is more WATER-soluble in an acidic solution.
What is the relationship between solubility and absorption?
The relationship between drug permeability, solubility, and absorption in humans suggests that drugs in the high permeability-high solubility group and those in the high permeability-low solubility group are absorbed completely.Why are lipid soluble drugs more easily absorbed?
Because the cell membrane is lipoid, lipid-soluble drugs diffuse most rapidly. Small molecules tend to penetrate membranes more rapidly than larger ones.
How does molecular weight affect drug absorption?
Although numerous studies have focused on its applications as pharmaceutical excipients or bioactive reagents, molecular weight (MW)-dependent pharmaceutical and bioactive properties remain unclear. … As the MW increases, the absorption decreases.
How does low solubility affect drug absorption?
The negative effect of compounds with low solubility include poor absorption and bioavailability, insufficient solubility for IV dosing, development challenges leading to increasing the development cost and time, burden shifted to patient (frequent high-dose administration) [11].
How does permeability affect drug absorption?
A drug’s permeability across biological membranes is a key factor that influences the absorption and distribution. This is because if a drug wants to reach to the systemic circulation, it needs to cross several semipermeable cell membranes firstly.What factors affect absorption of a drug?
- Lipid water solubility. Lipid water solubility coefficient is the ratio of dissolution of drug in lipid as compared to water. …
- Molecular size. …
- Particle size. …
- Degree of Ionization. …
- Physical Forms. …
- Chemical Nature. …
- Dosage Forms. …
- Formulation.
Lipids play an important role in energy metabolism and storage, as structural components, in signaling, and as hormones. … The presence of dietary lipids or lipids from drug formulations/lipidic prodrugs can influence drug absorption by incorporating to the natural lipid metabolic pathways.
Article first time published onCan lipid soluble drugs cross blood brain barrier?
Large molecules do not pass through the BBB easily. Low lipid (fat) soluble molecules do not penetrate into the brain. However, lipid soluble molecules, such as barbituate drugs, rapidly cross through into the brain.
How Does pH and pKa affect drug absorption?
The weak base is absorbed at a faster rate from the intestine (pH 7.50 – 8), this is because the basic substances can’t be ionized in basic medium. So the uncharged substances can be passed easily due to its lipid solubility. … So, in this case pH = pKa. Hence, when pH is equal to pKa, the drug is ionized halfly.
How does solubility affect bioavailability?
If a drug is not soluble in water or has a very low solubility it can not be absorbed through the membranes and thus its permeability will be negligible and consequently its bioavailability will be too low.
Which drug is lipid soluble?
Propranolol, oxprenolol, metoprolol, and timolol are the most lipid-soluble beta-adrenoceptor antagonists, and atenolol, nadolol, and sotalol are the most water-soluble; acebutolol and pindolol are intermediate [14].
How does solubility affect oral drug absorption?
When delivering an active ingredient orally, it must first dissolve in gastrointestinal fluids before it can then permeate the membranes of the gastrointestinal tract to reach systemic circulation. Therefore, a drug with poor aqueous solubility will exhibit dissolution rate-limited absorption.
What do you mean by the term absorption of drug discuss the various factors affecting drug absorption?
Absorption: depends on drug pKa and whether its an acidic or basic drug, GI ph influences drug absorption by determining amount of drug that would exist in unionised form at the site of absorption.
Does molecular weight affect drug distribution?
The results indicated that molecular weight significantly influenced the uptake of CM-chitosan from the lumen of abdomen and blood vessels to peripheral tissues, the distribution of this chemical and urinary excretion after intraperitoneal administration.
Why is molecular weight important for drugs?
To enable a drug to get absorbed by the body in the proper speed and the proper amount it is possible to spray a liquid on small drug cores which will form a membrane that the drug must get through before it reaches the body.
How does membrane permeability affect drug distribution?
Membrane permeability tends to restrict the transfer and distribution of drugs once they are delivered to the tissue. The other major factor which determines the rate of drug distribution is blood perfusion.
What makes a drug more permeable?
The ability of a drug to pass across biological membrane is defined as drug permeability. Passive diffusion is the major absorption pathway. Permeability can be improved either by incorporating permeation enhancers in formulations or by optimizing the molecule stucture.
Why is drug permeability important?
Permeability is important in governing the ability of drug substances to transport across gastrointestinal membrane and also crucial for proper drug distribution to pharmacological target organs and cells, and is therefore frequently utilized in drug discovery and development.
What is lipid based drug delivery system?
Lipid-based drug delivery systems (LBDDS) is a wide-ranging designation for formulations containing a dissolved or suspended drug in lipidic excipients. Lipids are esters of fatty acids – lipophilic hydrocarbon chains linked to a hydrophilic group like glycerol, polyglycerol, or polyalcohol (Figure 1).
What are the characteristics of lipid soluble drugs?
Number of half-livesFraction of drug remaining312.5%46.25%53.125%61.56%
What are the 4 main functions of lipids?
Within the body, lipids function as an energy reserve, regulate hormones, transmit nerve impulses, cushion vital organs, and transport fat-soluble nutrients.
Which type of drug penetrate CNS better?
Several anti-infectives (e.g., isoniazid, pyrazinamide, linezolid, metronidazole, fluconazole, and some fluoroquinolones) reach a CSF-to-serum ratio of the areas under the curves close to 1.0 and, therefore, are extremely valuable for the treatment of CNS infections.
Which drugs Cannot be filtered through glomerulus?
Which drugs cannot be filtered through glomerulus? Explanation: Drugs that are bound to plasma behave as macromolecules don’t get filtered through the glomerulus. Unbound free drug which are less than 300 Dalton get filtered by the glomerulus. 12.
What substances Cannot cross the blood brain barrier?
Furthermore, only a select number of substances can pass through the endothelial cells. Such substances include lipid-soluble substances (e.g., oxygen, carbon dioxide). Hydrophilic substances, for example, hydron and bicarbonate, are not permitted to pass through cells and across the blood-brain barrier.
How does pH affect the solubility of a drug?
Changes in the pH of gastrointestinal fluids can alter the solubility of drugs. Acidic drugs will be more poorly soluble in acidic media, while basic drugs will lose solubility in basic media and vice versa.
How does pKa affect drug?
The pKa of a drug is the hydrogen ion concentration (pH) at which 50% of the drug exists in its ionized hydrophilic form (i.e., in equilibrium with its un-ionized lipophilic form). All local anesthetic agents are weak bases. At physiologic pH, the lower the pKa the greater the lipophilicity.
How pH affect drug absorption?
GI pH is an important factor that can markedly affect oral drug absorption and bioavailability as it may have significant influence on drug dissolution & solubility, drug release, drug stability, and intestinal permeability. Different regions of the GI tract have different drug absorptive properties.
Why is solubility important in drug development?
Solubility, the phenomenon of dissolution of solute in solvent to give a homogenous system, is one of the important parameters to achieve desired concentration of drug in systemic circulation for desired (anticipated) pharmacological response.