How does pepsin break down protein
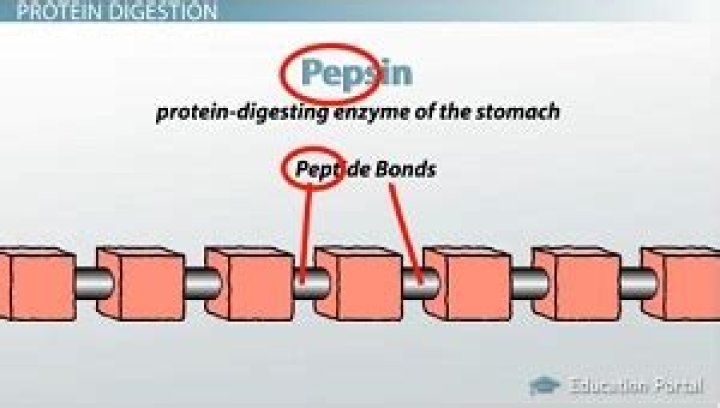
The specific reaction catalyzed by pepsin is the acid hydrolysis of the peptide bond. This reaction will break down proteins into smaller units to enable the digestive process. Pepsin demonstrates an unusual property for an enzyme; it does not actually form chemical bonds with its substrate.
How does pepsin break down amino acids?
Pepsin cleaves peptide bonds in the amino-terminal side of the cyclic amino acid residues (tyrosine, phenylalanine, and tryptophan), breaking the polypeptide chains into smaller peptides (Fange and Grove, 1979).
What does a pepsin do?
An enzyme made in the stomach that breaks down proteins in food during digestion. Stomach acid changes a protein called pepsinogen into pepsin.
What is it called when pepsin breaks down proteins?
Pepsin Breaks Down Food Proteins The acid in the stomach causes food proteins to unfold in a process called denaturation. Denaturation exposes the protein’s molecular bonds so that pepsin can access them and break the proteins into smaller fragments, called peptides or polypeptides.Where does HCl and pepsin break down proteins?
From the Mouth to the Stomach The stomach releases gastric juices containing hydrochloric acid and pepsin (an enzyme) which initiate the chemical breakdown of protein. Hydrochloric acid (HCl) in the stomach begins protein digestion by denaturing the protein; denaturation results in the loss of the protein’s function.
What is the role of pepsin in the digestive process at what pH does it work?
Answer: Pepsin basically breaks down proteins into peptides…. it works with acidic pH (HCL in stomach) . In Acidic pH Pepsinogen is converted in enzyme Pepsin thus acidic pH is required.
What is the main role of pepsin in this metabolic reaction?
Pepsin is an enzyme that acts in the stomach for protein digestion. Which of these is most likely the main role of pepsin in this metabolic reaction? Enzymes enable reactions to occur at lower temperatures.
What happens to pepsin as it enters the small intestine?
When pepsin enters the small intestine, it becomes inactive. Pepsin is a strong enzyme that becomes active once in the presence of hydrochloric acid…Why pepsin does not digest proteins in the stomach wall?
Pepsin is secreted by cells lining the walls of our stomach to digest proteins that are ingested in our food. … Pepsin does not end up digesting the stomach because the epithelial cells of the stomach secrete a protective mucus layer.
Why pepsin is active at pH 2?The reason pepsin functions best at pH 2 is because the carboxylic acid group on the amino acid in the enzyme’s active site must be in its protonated state, meaning bound to a hydrogen atom. At low pH the carboxylic acid group is protonated, which allows it to catalyze the chemical reaction of breaking chemical bonds.
Article first time published onHow does pepsin break down a substrate?
The specific reaction catalyzed by pepsin is the acid hydrolysis of the peptide bond. This reaction will break down proteins into smaller units to enable the digestive process. Pepsin demonstrates an unusual property for an enzyme; it does not actually form chemical bonds with its substrate.
Does pepsin digest carbohydrates?
Protein digestion occurs in the stomach and the duodenum through the action of three main enzymes: pepsin, secreted by the stomach, and trypsin and chymotrypsin, secreted by the pancreas. During carbohydrate digestion the bonds between glucose molecules are broken by salivary and pancreatic amylase.
Is pepsin intracellular or extracellular?
Question : Pepsin is an intracellular/extracellular enzyme. Which is correct? Video Solution: Pepsin is an intracellular/extracellular enzyme.
What is the role of pepsin in stomach Class 10?
The Function Of Pepsin are: It helps in the digestion. It breaks down the large polypeptides into smaller peptide fragments. Help break down dietary proteins like eggs, meat, seeds, and other dairy products.
How are proteins broken down into amino acids?
Once a protein source reaches your stomach, hydrochloric acid and enzymes called proteases break it down into smaller chains of amino acids. Amino acids are joined together by peptides, which are broken by proteases. From your stomach, these smaller chains of amino acids move into your small intestine.
What are amino acids broken down into?
The individual amino acids are broken down into pyruvate, acetyl CoA, or intermediates of the Krebs cycle, and used for energy or for lipogenesis reactions to be stored as fats.
How are proteins broken down in the body?
Proteins. Proteins are digested in the stomach and small intestine. Protease enzymes break down proteins into amino acids.
How does pH affect pepsin activity?
Pepsin is an aspartic protease and its activity is directly dependent on the pH of the solution environment [41]. At pH values >2.5, pepsin activity begins to decline, with incomplete degradation of the three susceptible substrate proteins by pepsin being observed.
At what pH is pepsin likely to denature?
Pepsin undergoes a conformational transition from the native (at acidic pH) to the denatured (at alkaline pH) state in a narrow pH range (between 6 and 7).
Why is pepsin released in an inactive form?
Complete answer: Protein digesting enzymes are secreted in an inactive form to protect the organs and glands from digestion by the enzymes. … The proenzyme pepsinogen, on exposure to hydrochloric acid, gets converted into the active enzyme pepsin which is the proteolytic enzyme present in the stomach for digestion.
Why is pepsin released from chief cells of the stomach as pepsinogen rather than being released as pepsin itself?
Pepsin enzyme is secreted by gastric glands of the stomach as inactive pepsinogen to protect the cells of these secretory glands from strong protein digesting action of the enzyme. The inactive form of the enzyme is activated by acidic pH of stomach lumen and the stomach wall is protected by mucus lining.
What happens when pepsin enters the small intestine nutrition quizlet?
What happens when pepsin enters the small intestine? It cleaves proteins into smaller peptides and some free amino acids.
What happens when pepsin enters the small intestine quizlet?
What happens when pepsin enters the small intestine? It is digested due to the high pH.
How the structure and function of pepsin will change as it enters the small intestine?
Which of the following best predicts how the structure and function of pepsin will change as it enters the small intestine? Pepsin will change in shape because of the basic environment of the small intestine; therefore, its enzymatic activity will decrease.
What macromolecules are broken down by pepsin?
Protein. A large part of protein digestion takes place in the stomach. The enzyme pepsin plays an important role in the digestion of proteins by breaking down the intact protein to peptides, which are short chains of four to nine amino acids.
Why is pepsin not denatured at low pH?
Pepsin has several properties which are markedly different from those common for other proteins. It has a very low pH optimum for the hydrolysis of different substrates and a high activity at pH 2. This implies a very stable tertiary structure under conditions in which many proteins are fully denatured.
Does pepsin denature protein?
Pepsin is an endopeptidase that breaks down proteins into smaller peptides. It is produced in the gastric chief cells of the stomach lining and is one of the main digestive enzymes in the digestive systems of humans and many other animals, where it helps digest the proteins in food.
Is pepsin hydrophobic or hydrophilic?
The central hydrophobic core of pepsin (catalytic aspartic acid residues at position 32 and 215) comprises the active site of the enzyme. This site can accommodate an approximately 8-amino-acid portion of protein substrate. FIGURE 1. Schematic structure of human pepsin (EC 3.4.
Is pepsin a molecule?
The Pepsin molecule – rotatable in 3 dimensions Pepsin is a protease (protein-digesting enzyme), which is active in the stomach. The porcine pepsin molecule displayed here consists of 327 amino acid residues – 5053 atoms, now including hydrogen atoms, but this also includes oxygens from 375 water molecules!
Does pepsin break down starches?
It helps break down large starch molecules into smaller sugar molecules. Pepsin, produced in the stomach. Pepsin helps break down proteins into amino acids.
What happens to pepsin after digestion?
In the digestive tract pepsin effects only partial degradation of proteins into smaller units called peptides, which then either are absorbed from the intestine into the bloodstream or are broken down further by pancreatic enzymes.