How does the quantum mechanical model describe electrons
Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. … Electrons have an intrinsic property called spin, and an electron can have one of two possible spin values: spin-up or spin-down. Any two electrons occupying the same orbital must have opposite spins.
What does the quantum mechanical model say about electrons?
In the quantum-mechanical model of an atom, electrons in the same atom that have the same principal quantum number (n) or principal energy level are said to occupy an electron shell of the atom. Orbitals define regions in space where you are likely to find electrons.
How do quantum numbers describe the properties of an electron?
The principal quantum number defines the general size and energy of the orbital. The l value specifies the shape of the orbital. Orbitals with the same value of l form a subshell. In addition, the greater the angular momentum quantum number, the greater is the angular momentum of an electron at this orbital.
How are electrons described in the quantum mechanical model chegg?
Quantum Model For this model and study, Schrodinger considered electrons as matter waves. This consideration comes from the theory put forth by Louis De Broglie. He stated that all particles can be considered as matter waves and that it had wavelength (ℷ).How are the models different in their description of electron location?
In the Bohr Model, the electron is treated as a particle in fixed orbits around the nucleus. In the Quantum Mechanical Model, the electron is treated mathematically as a wave. The electron has properties of both particles and waves.
How are the Bohr model of the hydrogen atom and the quantum mechanical model of atoms similar How are they different?
The Bohr model and quantum model are models that explain the structure of an atom. … The key difference between Bohr and quantum model is that Bohr model states that electrons behave as particles whereas quantum model explains that the electron has both particle and wave behavior.
How many electrons can the fourth Shell hold?
The fourth shell holds 32 electrons; 2 in a 4s orbital; 6 in three 4p orbitals; 10 in five 4d orbitals; and 14 in seven 4f orbitals. The exact arrangement of electrons in an atom’s shells and subshells is the atom’s electron configuration. It can be predicted by applying three rules.
What is being described by the quantum number?
We use a series of specific numbers, called quantum numbers, to describe the location of an electron in an associated atom. Quantum numbers specify the properties of the atomic orbitals and the electrons in those orbitals. An electron in an atom or ion has four quantum numbers to describe its state.What are the rules for the possible values of the four quantum numbers?
Rules Governing the Allowed Combinations of Quantum Numbers The principal quantum number (n) cannot be zero. The allowed values of n are therefore 1, 2, 3, 4, and so on. The angular quantum number (l) can be any integer between 0 and n – 1. If n = 3, for example, l can be either 0, 1, or 2.
Which quantum number describes the possible orientation of the orbital occupied by an electron?Magnetic Quantum Number (ml): ml = -l, …, 0, …, +l. Specifies the orientation in space of an orbital of a given energy (n) and shape (l). This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell.
Article first time published onWhat is quantum mechanical model explain?
Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. … Electrons have an intrinsic property called spin, and an electron can have one of two possible spin values: spin-up or spin-down. Any two electrons occupying the same orbital must have opposite spins.
How does quantum mechanical model help explain shapes of p orbitals?
The quantum mechanical model is based on mathematics. … The quantum mechanical model of the atom uses complex shapes of orbitals (sometimes called electron clouds), volumes of space in which there is likely to be an electron. So, this model is based on probability rather than certainty.
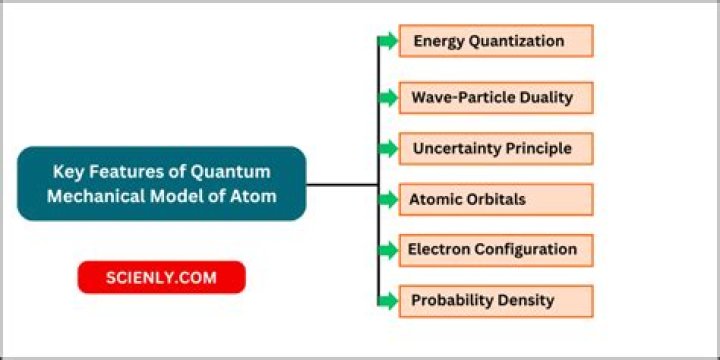
What are the main features of quantum mechanical model of atom?
- The energy of electrons in an atom is quantized (it can only have certain specific values).
- The existence of quantized. …
- All the information about the electron in an atom is contained in its orbital wave function .
What is SPDF rule?
There are different orbital shapes (s,p,d,f) Each orbital can only hold 2 electrons max. There is a hierarchy, i.e. s orbitals will be filled before p orbitals which will be filled before d orbitals and so on. (s<p<d<f) (note, this is a general rule but there are exceptions)
Which quantum number signifies the size of the electron cloud?
All electrons have four quantum numbers which describe the location of electrons in the electron cloud of an atom. The principle quantum number (n) describes the size of the orbital the electron is in. The angular momentum quantum number (l) describes the shape of the orbital.
How many electrons in an atom could have these sets of quantum numbers?
Only 2 electrons.It is because from these quantum numbers,it indicates an orbital of 6p. As only 2 electrons can present in an orbital,so only 2 electrons in an atom can have this set of quantum numbers.
Does the quantum mechanical model estimate the probability of finding an electron?
An electron moves within one of many orbitals of varying size and shape. … The quantum mechanical model of the atom estimates the probability of finding an electron in a certain position.
How did Bohr make use of the concept of quantum in explaining the state of electrons in the atom?
Bohr explained that electrons can be moved into different orbits with the addition of energy. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy—a quantum of light, or photon. This was the basis for what later became known as quantum theory.
How do the principal quantum numbers of the quantum mechanical model relate to the Bohr model of the atom?
These are all associated with particular physical properties. n, the principal quantum number, is associated with the total energy, the same way it is in the Bohr model. … , the orbital quantum number, is connected to the total angular momentum of the electron.
What values are needed to determine the energy of an electron in a many electron atom?
The energy of an electron in many–electron atom is determined by both principal quantum number (n) and azimuthal quantum number (l). The size of an orbital can be described by principal quantum number (n).
What are the possible sets of quantum numbers that can describe a 2p electron in an atom?
A For a 2p orbital, we know that n = 2, l = n − 1 = 1, and ml = −l, (−l +1),…, (l − 1), l. There are only three possible combinations of (n, l, ml): (2, 1, 1), (2, 1, 0), and (2, 1, −1).
Which of the following sets of quantum numbers can describe a 3p electron?
For a 3p electron, the number 3 represents n, whereas s represents ℓ. ℓ corresponds to the following values: s = 0, p = 1, d = 2, f = 3; this means that in this orbital, ℓ = 1. The value for mℓ must be between -ℓ and ℓ, so if ℓ = 1, mℓ must be -1, 0, or 1.
What are quantum numbers how they are classified explain?
The set of numbers used to describe the position and energy of the electron in an atom are called quantum numbers. There are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. The values of the conserved quantities of a quantum system are given by quantum numbers.
What are quantum numbers explain the significance of four quantum numbers associated with an electron?
The four quantum numbers n, ℓ, m, and s specify the complete and unique quantum state of a single electron in an atom, called its wave function or orbital. Two electrons belonging to the same atom cannot have the same values for all four quantum numbers, due to the Pauli exclusion principle.
Which quantum number governs spatial orientation of an orbital?
Magnetic quantum number specifies orientation of orbitals in space.
Which of the following is a correct set of quantum numbers for an electron in a 5f orbital?
The correct set of quantum numbers is choice “a”. Choice “a” is n = 5, l = 3, ml = +1, which is the 5f orbital…
How do you determine the quantum number of an element?
Look at the Periodic Table of Elements and find the element that you want to know the quantum number for. Find the principal number, which denotes the element’s energy, by looking in which period the element is found. For example, sodium is in the third period of the table, so its principal quantum number is 3.
How does an electron configuration denote the atoms of elements?
The electron configuration of an atom is the representation of the arrangement of electrons distributed among the orbital shells and subshells. … The valence electrons, electrons in the outermost shell, are the determining factor for the unique chemistry of the element.
How is the concept of electron density used to describe the position of an electron in the quantum mechanical treatment of an atom?
What is electron density? The theory of quantum mechanics says that an electron’s position can only be described statistically. … This calculation produce a quantity called electron density, a number that tells us the relative probability of finding an electron at a particular point in space.
How the quantum mechanics determine the electron configuration?
An electron configuration simply lists the shell and subshell labels, with a right superscript giving the number of electrons in that subshell. … Electrons are typically organized around an atom by starting at the lowest possible quantum numbers first, which are the shells-subshells with lower energies.
How do quantum numbers describe the properties of an electron?
The principal quantum number defines the general size and energy of the orbital. The l value specifies the shape of the orbital. Orbitals with the same value of l form a subshell. In addition, the greater the angular momentum quantum number, the greater is the angular momentum of an electron at this orbital.



