How is a delta bond formed
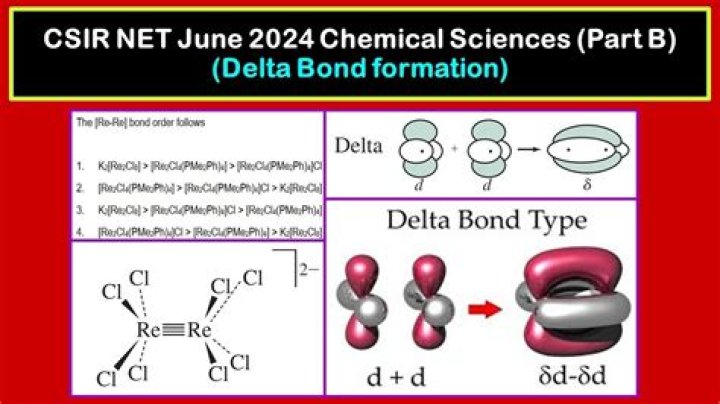
Delta bonds are formed by side way overlap of four lobes of d-orbitals. For example: the d(xy) of one metal can overlap with d(xy) of other metal.
Which orbitals form Delta molecular orbitals?
Delta bonding molecular orbital is formed by the overlap of \[{{d}_{{{x}^{2}}-{{y}^{2}}}}\]and \[{{d}_{{{x}^{2}}-{{y}^{2}}}}\] orbitals when z is the internuclear axis.
What is a Delta orbital?
Chemical bonding involving delta (δ) orbitals. A δ orbital is so called because it resembles a d-orbital when viewed along the axis of a molecule and has two units of orbital angular momentum around the internuclear axis. The formation of δ bonds originates in the overlap of d-orbitals on different atoms.
How many nodes does a Delta bond have?
Sigma, pi and delta denote how many planar nodes are in the bond. Sigma bonds have no node, pi bonds have one and delta bonds have two. You can tell what kind of bond forms by how the orbitals overlap.Do d-orbitals form pi bonds?
The Greek letter π in their name refers to p orbitals, since the orbital symmetry of the pi bond is the same as that of the p orbital when seen down the bond axis. One common form of this sort of bonding involves p orbitals themselves, though d orbitals also engage in pi bonding.
What is Delta en in chemistry?
Illustrated Glossary of Organic Chemistry – δ+ δ+: A symbol which indicates that an atom or region with a deficiency of electron density, often because of resonance delocalization, electronegativity differences, or inductive effects. EN C (2.5) < EN Cl (3.0) ΔEN = 3.0 – 2.5 = 0.5.
What is meant by Delta bonds?
In chemistry, delta bonds (δ bonds) are covalent chemical bonds, where four lobes of one involved atomic orbital overlap four lobes of the other involved atomic orbital. … The first compound identified as having a δ bond was potassium octachlorodirhenate(III).
Can two's orbitals make a π bond?
A π bond has a plane of symmetry along the bond axis. It cannot be formed by s-orbitals; it needs at least p-orbitals to be created.Are there quadruple bonds?
A quadruple bond is a type of chemical bond between two atoms involving eight electrons. … Stable quadruple bonds are most common among the transition metals in the middle of the d-block, such as rhenium, tungsten, technetium, molybdenum and chromium.
What is Delta and Delta in chemistry?Delta (uppercase/lowercase Δ δ), is a letter of the Greek alphabet, used to represent the “d” sound in Ancient and Modern Greek. … In molecular chemistry, the lower case Greek letter δ is used to represent a partial charge. This is always less than the unit charge associated with an ion, but has no real fixed value.
Article first time published onCan 2 d orbitals form sigma bonds?
σ bonding with d orbitals An example of a σ bond formed by \d orbitals is that of two dz2 orbitals (see Figure 5.1. 3. 2). If a bonded atom is in a position other than on the z axis (in an octahedral geometry, for example), sigma bonds can also form.
Can dx2 y2 form pi bond?
If dx^2 – y^2 and py orbitals come close together along z – axis, then they can form pi – bond by sideways overlapping.
Which of the following orbital can not form pi as well as Delta bond?
dz2 can not form π as well δ bond but it can form σ-bond.
How is a sigma bond formed?
Sigma bond (σ bond): A covalent bond formed by overlap of atomic orbitals and/or hybrid orbitals along the bond axis (i.e., along a line connected the two bonded atoms). The sigma bond in the a hydrogen molecule (shown in red) is formed by overlap of a pair of 1s orbitals, one from each hydrogen atom.
What is pi molecular orbital?
Pi orbital (π orbital): The bonding molecular orbital component of a pi bond. The π orbital of ethylene’s carbon-carbon pi bond has two orbital lobes, one above the plane of the atoms, and another below the plane. This is a bonding molecular orbital. The plane containing the atoms is also the pi orbital’s one node.
What are the types of covalent bond?
- Single Covalent Bond.
- Double Covalent Bond.
- Triple Covalent Bond.
What is banana bond?
A banana bond is also known as a bent bond. It is a type of chemical bonding where the ordinary hybridization state of two atoms making up a chemical bond are modified with increased or decreased s-orbital character in order to accommodate a particular molecular geometry (that of 3 bananas in a ring shape).
What is Gamma bond?
Gamma is the rate of change for an option’s delta based on a single-point move in the delta’s price. Gamma is at its highest when an option is at the money, and is at its lowest when it is further away from the money.
How do you write Delta in chemistry?
Go to chem draw, click the A in the left tab or insert text option, right click in the box, go to font and choose symbol. Now hit caps lock and press D. You will have the Δ. Symbol options can be used to add all types of chemical entities.
How can you tell which bond is formed?
Bond type is predicated on the difference in electronegativity of the two elements involved in the bond. The degree to which a given bond is ionic or covalent is determined by calculating the difference in electronegativity between the two atoms involved in the bond.
How do you write Delta?
Using an Alt keyboard shortcut to insert the Delta symbol You can press the Alt key in combination with numbers on the numeric keypad to insert the Delta symbol: Press Alt + 235 to enter lower case Delta (δ). Press Alt + 916 to enter upper case or capital letter Delta (Δ).
How are quadruple bonds formed?
Double bond occurs when four electrons are shared, occupying one σ (sigma) and one π (pi) orbitals. Finally, triple bond occurs by sharing of six electrons into one σ and two π orbitals. Therefore, quadruple bond is supposed to occur when eight electrons are shared by two atoms.
What is O2 bond?
The O2 Lewis structure has a double bond between two oxygen atoms. According to the octet rule, oxygen atoms need to bond twice. The O2 molecule is diatomic, meaning that two atoms of the same element are connected in a pair.
What are the strongest covalent bonds?
The strongest type of covalent bonds are sigma bonds, which are formed by the direct overlap of orbitals from each of the two bonded atoms. Regardless of the atomic orbital type, sigma bonds can occur as long as the orbitals directly overlap between the nuclei of the atoms.
Why pi bond is never formed alone?
It is formed along with a sigma bond . Pi bond is formed by sideway overlap of p-orbitals only. …
How sigma bond and pi bonds are formed?
Sigma and pi bonds are formed by the overlap of atomic orbitals. Sigma bonds are formed by end-to-end overlapping and Pi bonds are when the lobe of one atomic orbital overlaps another. Both acquired their names from the Greek letters and the bond when viewed down the bond axis.
How are sigma and pi bonds formed?
Sigma and pi bonds are formed by atomic orbital overlap. Sigma bonds are formed by the overlapping end – to – end and Pi bonds occur when one atomic orbital lobe overlaps another. … This type of covalent bond is formed by the overlap of bonding orbitals along the internuclear axis from end to end (head-on).
Who created delta?
The Hamiltonian operator. The symbol , which is also called a “del,” “nabla,” or “atled” (delta spelled backwards), was introduced by William Rowan Hamilton (1805-1865) in 1853 in Lectures on Quaternions, according to Cajori vol. 2, page 135.
What does ∆ stand for?
Delta (/ˈdɛltə/; uppercase Δ, lowercase δ or 𝛿; Greek: δέλτα, délta, [ˈðelta]) is the fourth letter of the Greek alphabet. In the system of Greek numerals it has a value of 4. It was derived from the Phoenician letter dalet 𐤃, Letters that come from delta include Latin D and Cyrillic Д.
What is simple delta?
A delta is an area of low, flat land shaped like a triangle, where a river splits and spreads out into several branches before entering the sea.
Can P and D form sigma bonds?
A sigma bond is formed due to overlapping of singular lobes of two orbitals. A d-orbital has four lobes, so it can form a sigma, pi or even a delta bond. In Dimeric Chromium Acetate, a dz2 orbital overlaps in a sigma fashion to form a bond. In the same compount.



