How is an ionic bond used in DNA
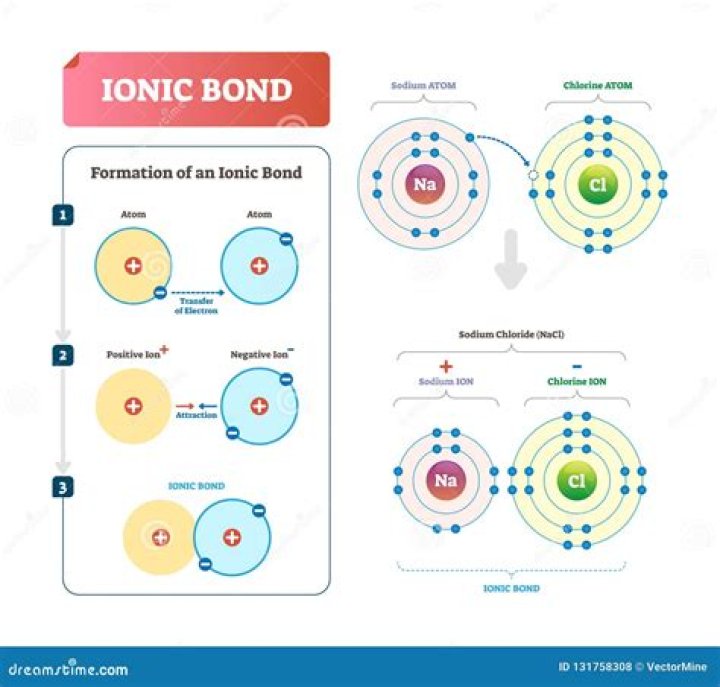
An ionic bond is a bond that is formed between ions of opposite charge. … These negative charges allow the DNA molecule to be “salted out” during DNA purification by using positively charged ions (divalent cations) such as Mg2+, Mn2+, Ca2+, etc.
What type of bond is used in DNA?
The nucleotides in a base pair are complementary which means their shape allows them to bond together with hydrogen bonds. The A-T pair forms two hydrogen bonds. The C-G pair forms three. The hydrogen bonding between complementary bases holds the two strands of DNA together.
How are ionic bonds used?
Ionic bonds are important because they allow the synthesis of specific organic compounds. Scientists can manipulate ionic properties and these interactions in order to form desired products. Covalent bonds are especially important since most carbon molecules interact primarily through covalent bonding.
How are ionic bonds used in biology?
As ions move in and out of cells, they can build up positive and negative charges on different sides of those cells. Once these charges build up, they can trigger a reaction such as muscle or nerve contraction. Ionic bonds are also involved in determining cell shapes, catalytic reactions, and neuron functions.What bonds hold nucleotides together in DNA?
Nucleotides are joined together by covalent bonds between the phosphate group of one nucleotide and the third carbon atom of the pentose sugar in the next nucleotide. This produces an alternating backbone of sugar – phosphate – sugar – phosphate all along the polynucleotide chain.
What are two examples of ionic bond uses in real life?
- Table Salt.
- Iodized Salt.
- Fluoride in Toothpaste.
- Baking Soda.
- Washing Soda.
- Household Bleach.
- Preservative.
- Anti-caking Agent.
Are there peptide bonds in DNA?
Nucleic acids (i.e., DNA and RNA) are made up of nucleotide monomers. … Peptide bonds are formed between the carboxylic acid group of one amino acid and the amine group of a second amino acid. One of the twenty amino acids, called cysteine, contains a sulfhydryl group as its side chain group.
What is an ionic bond example?
Ionic bonds involve a cation and an anion. The bond is formed when an atom, typically a metal, loses an electron or electrons, and becomes a positive ion, or cation. … One example of an ionic bond is the formation of sodium fluoride, NaF, from a sodium atom and a fluorine atom.Why are ionic bonds used in medicine?
Ionic liquids are highly tunable and can be readily optimized to connect with a variety of pharmaceutical ingredients. 3. Variable linker for regulating the distance between the ionic core and active pharmaceutical ingredient.
What compound uses ionic bonds?Examples of Ionic Bonds Some ionic bond examples include: NaCl: sodium chloride. NaBr: sodium bromide. NaF: sodium fluoride.
Article first time published onWhat objects are ionic bonds?
- Sodium chloride – NaCl – table salt.
- Calcium chloride – CaCl2 – rock salt.
- Sodium hydroxide – NaOH – Lye used in soap.
- Sodium fluoride – NaF – fluoride in toothpaste.
- Iron Oxide – Fe2O3 – rust.
- Calcium hydroxide – Ca(OH)2 – basic salt in antacid tablets.
Why are ionic bonds called ionic bonds?
Thus, the term “ionic bonding” is given when the ionic character is greater than the covalent character – that is, a bond in which a large electronegativity difference exists between the two atoms, causing the bonding to be more polar (ionic) than in covalent bonding where electrons are shared more equally.
What types of bonds are involved in stabilizing the DNA backbone and what types of bonds are found between complementary base pairs?
The base pairs are stabilized by hydrogen bonds; adenine and thymine form two hydrogen bonds between them, whereas cytosine and guanine form three hydrogen bonds between them. Figure 7. Hydrogen bonds form between complementary nitrogenous bases on the interior of DNA.
What forms covalent bonds between nucleotides?
Nucleotides are linked together by the formation of a phosphodiester bond which is formed between the 3′ -OH group of one sugar molecule, and the 5′ phosphate group on the adjacent sugar molecule.
What does a peptide bond do in DNA?
The energy of DNA interaction with the peptide is higher than with individual amino acids. The peptide bonds increase the interaction of Lys-Glu peptide with DNA, which potentiates the biological effect on cell proliferation in organotypic culture of splenic cells.
Which bond is a peptide bond?
In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 (carbon number one) of one alpha-amino acid and N2 (nitrogen number two) of another, along a peptide or protein chain.
Are DNA strands held together by ionic bonding?
In general, almost all of the bonds that hold together a single strand of DNA are covalent bonds. As you know, a DNA molecule has three principal components; there are the nitrogenous bases, the deoxyribose sugars, and the phosphate groups.
How are ions used in real life?
Ion exchange, or the replacement of one ion by another, is used in applications such as water purification, while chemists and physicists use ions in mass spectrometry, to discover mass and structural information concerning atoms and molecules.
Is ibuprofen covalent or ionic?
Example 1: Computing Molecular Mass for a Covalent Compound Ibuprofen, C13H18O2, is a covalent compound and the active ingredient in several popular nonprescription pain medications, such as Advil and Motrin.
How do ionic bonds stay together?
An ionic bond is held together by the electrostatic attraction between ions that are near one another. Electrostatic attraction is the attraction between atoms that have opposite charge and holds the atoms together in ionic bonds. … The atom actually “accepts” or “takes” the electrons that the other atom is giving up.
How can the name of a compound be used to determine if ionic bonds or covalent bonds are present?
By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.
What is an ionic bond with two suitable examples explain the difference between an ionic bond and a covalent bond?
Covalent BondsIonic BondsRoom Temp. StateGas/LiquidSolidShapeNot definiteDefiniteCommon ExampleHydrogen Chloride (HCl)Sodium chloride (NaCl)Happens BetweenMetal and non-metalTwo non-metals
What is an ionic bond with two suitable examples explain the difference between an ionic and a covalent bond?
Ionic bondsCovalent bondsIn ionic bonds, one atom donates an electron to stabilize the other atom.In a covalent bond, the atoms are bound by the sharing of electrons.
Why are electrons transferred in ionic bonds?
It is the power of an atom in a molecule to attract electrons to itself. When the electronegativity difference between the two atoms is greater than 2.0, the bond is considered ionic; i.e. there is complete transfer (between the two atoms) of the electrons in the bond.
How do ionic bonds form and how are the ions arranged in an ionic compound?
The ions in a solid ionic compound are not randomly arranged. Instead, they have a regular, repeating arrangement called an ionic lattice . The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other.
How are ionic bonds formed quizlet?
ionic bonds form when electrons are transferred from one atom to another atom. ions of different elements can combine by forming ionic bonds . positive ions & negative ions form when atom s lose or gain electrons. … Atoms ,non-metal of elements tend to lose electrons when they form bonds.
Why are ionic bonds stronger than covalent bonds?
They tend to be stronger than covalent bonds due to the coulombic attraction between ions of opposite charges. To maximize the attraction between those ions, ionic compounds form crystal lattices of alternating cations and anions. … The opposing strong internuclear repulsion maintains the separation between ions.
What bonds and interactions stabilize the DNA double helix?
The structure of the DNA helix is stabilized by van der Waals forces, hydrogen bonds between complementary organic bases (a base pair), and hydrophobic interactions between the nitrogenous bases and the surrounding sheath of water.
Why are hydrogen bonds used in DNA?
Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.
What types of bonds are involved in stabilizing the DNA backbone and what types of bonds are found between complementary base pairs quizlet?
Complementary base pairs are stacked in the center of the helix and are stabilized by hydrogen bonds. There are three hydrogen bonds between G and C, but only two between A and T.
How do the nucleotides in DNA bond with each other within a strand how do they bond with each other across strands?
Each strand is composed of nucleotides bonded together covalently between the phosphate group of one and the deoxyribose sugar of the next. From this backbone extend the bases. The bases of one strand bond to the bases of the second strand with hydrogen bonds.