How many covalent bonds are in h2o
By David Edwards
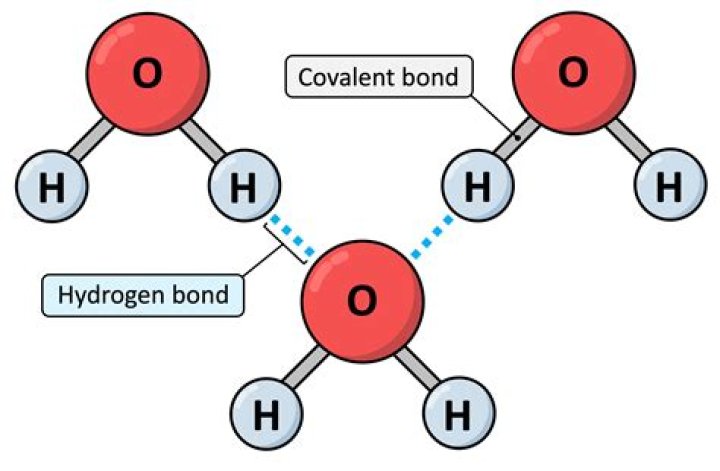
How many covalent bonds hold the water molecule together? A: The oxygen atom shares one pair of valence electrons with each hydrogen atom. Each pair of shared electrons represents one covalent bond, so two covalent bonds hold the water molecule together.
How many covalent bonds are in h2o? - Google Search
Strong linkages—called covalent bonds—hold together the hydrogen (white) and oxygen (red) atoms of individual H2O molecules. Covalent bonds occur when two atoms—in this case oxygen and hydrogen—share electrons with each other.
What type of bond is in H2O?
H2O is a Covalent bond, as the two elements are non-metals. A water molecule is a simple molecule, and simple molecules consist of a small number of atoms joined by covalent bonds.