How many neutrons does beryllium
NameBerylliumNeutrons5Electrons4Atomic Number (Z)49
How many neutrons does beryllium have?
NameBerylliumSymbolBeAtomic Number4Protons4Neutrons5
How many neutrons does beryllium 7 have?
Beryllium 7 has four protons and four neutrons.
How many valence neutrons does beryllium have?
Element NameBoronSymbolBAtomic Number5Electron Configuration1s22s22p1How does beryllium have 5 neutrons?
The number of protons determines the element, so that if there are four protons, this means that the nucleus must be that of an atom of beryllium. … In the case of beryllium, a light metal, only the beryllium-9 isotope is stable with its 9 nucleons (i.e. 4 protons and 5 neutrons).
Why the atomic mass of beryllium is 9?
IsotopeAtomic mass (Da)Isotopic abundance (amount fraction)9Be9.012 1831(5)1
How do u find neutrons?
To find the number of neutrons, subtract the number of protons from the mass number. number of neutrons=40−19=21.
What type of particle contains 35 protons 36 electrons and 45 neutrons?
So Bromine Br−1 has 35 protons, 36 electrons and 45 neutrons.Does sodium have 1 valence electrons?
A: An atom of a group 1 element such as sodium has just one valence electron. It is “eager” to give up this electron in order to have a full outer energy level, because this will give it the most stable arrangement of electrons.
How many electrons does beryllium 8 have?So… for the element of BERYLLIUM, you already know that the atomic number tells you the number of electrons. That means there are 4 electrons in a beryllium atom.
Article first time published onWhich is the isotopes of 5b?
Boron (5B) naturally occurs as isotopes 10B and 11B, the latter of which makes up about 80% of natural boron.
Why is 8 unstable?
Beryllium-8 has an extremely short half-life (8.19×10−17 seconds), and decays back into two helium-4 nuclei. This, along with the unbound nature of 5He and 5Li, creates a bottleneck in Big Bang nucleosynthesis and stellar nucleosynthesis, for it necessitates a very fast reaction rate.
What has 5 protons and 5 neutrons?
NameProtonsNeutronsHelium22Lithium34Beryllium45Boron56
What ion has 4 protons 4 neutrons and 2 electrons?
The element with atomic number 4 is beryllium, which means each atom of beryllium has 4 protons. A stable atom would have 4 neutrons and 4 electrons.
How many neutrons does bromine have?
a) How many neutrons does the atom of bromine have? The mass number = protons + neutrons. Bromine has a mass number of 80 and 35 protons so 80-35 = 45 neutrons.
How a neutron is formed?
Neutrons are produced copiously in nuclear fission and fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes. The neutron is essential to the production of nuclear power.
How do you find neutrons without atomic mass?
The number on the top is the atomic number, while the number near the bottom is the atomic mass. The atomic mass is the weighted average mass of all the isotopes of an element. If we round off the atomic mass to the nearest whole number and subtract the atomic number from it, we get the number of neutrons.
What does the number 84 in the name krypton 84 represent?
In the case of Krypton-84, this means that you have 84 nucleons, where 36 of these are protons, and the remaining 48 are neutrons. This becomes relevant in nuclear chemistry, where sometimes, you have to balance nuclear equations with respect to the mass numbers (nucleon numbers).
How many protons does be 9?
The stable isotope of beryllium is Be-9 which has 4 protons and 5 neutron. Radioactive isotopes will vary in the number of neutrons as the number of protons determine its identity.
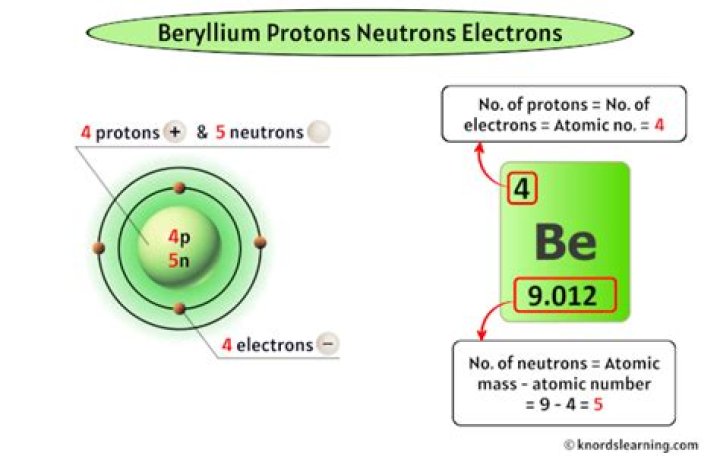
How many protons neutrons and electrons are in an atom of beryllium with a mass number of nine?
The Bohr Model for Beryllium (Be) has 4 protons in the nucleus due to the atomic number of Be being 4. The Mass number is 9 which means Beryllium needs 5 neutrons in the nucleus. (Mass number = protons + neutrons, 9 = 4 + n). Beryllium has four electrons to balance the four protons.
How do you find the mass of beryllium?
Atomic mass of Beryllium is 9.0122 u. Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. For 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it.
How do u calculate atomic mass?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
What is number 32 on the periodic table?
Germanium – Element information, properties and uses | Periodic Table.
What is beryllium-9 used for?
Beryllium is used in alloys with copper or nickel to make gyroscopes, springs, electrical contacts, spot-welding electrodes and non-sparking tools. Mixing beryllium with these metals increases their electrical and thermal conductivity.
What is the atomic number of be 9?
The atomic number of beryllium-9 is four.
What is the half life of beryllium-9?
Mass NumberHalf-lifeDecay Mode753.24 daysElectron Capture88.181×10 -17 secondsAlpha Decay9STABLE-101.387×10 +6 yearsBeta-minus Decay
How do you write the electron configuration for beryllium?
Beryllium is the fourth element with a total of 4 electrons. In writing the electron configuration for beryllium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the remaining 2 electrons for Be go in the 2s orbital. Therefore the Be electron configuration will be 1s22s2.
What is the electron configuration for beryllium atomic number 4?
ElementAtomic numberElectron configurationhydrogen11s1helium21s2lithium31s22s1beryllium41s22s2
What is the standard notation for beryllium?
Beryllium is a chemical element with the symbol Be and atomic number 4.
How many neutrons does sodium have?
Given the fact that there are 12 neutrons in sodium, the complete symbol for sodium is 2311Na.
How many atomic shells does sodium have?
ZElementNo. of electrons/shell11Sodium2, 8, 112Magnesium2, 8, 213Aluminium2, 8, 314Silicon2, 8, 4



