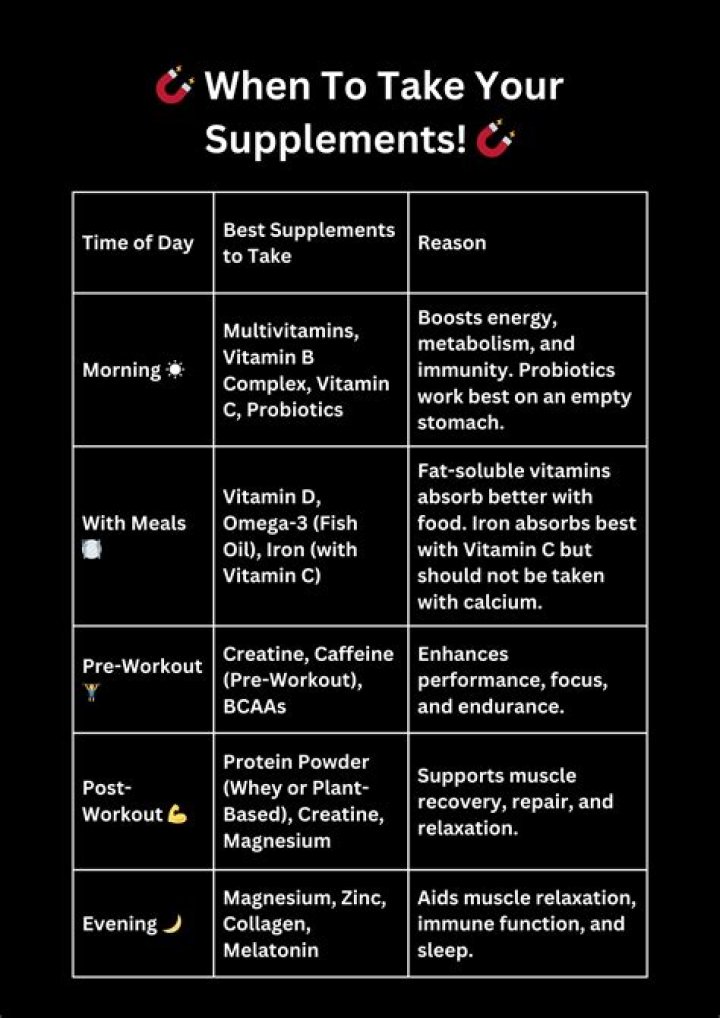
How many neutrons does deuterium have
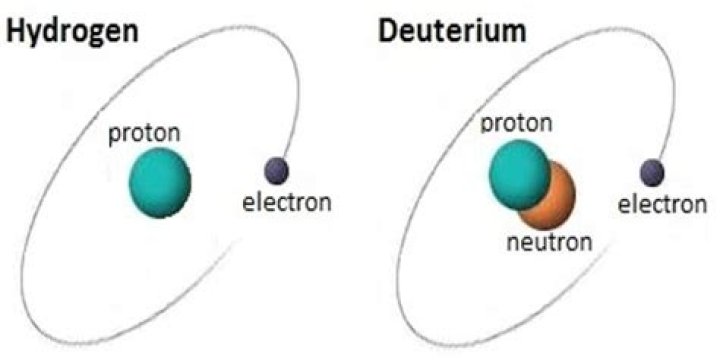
deuterium, (D, or 2H), also called heavy hydrogen, isotope of hydrogen with a nucleus consisting of one proton and one neutron, which is double the mass of the nucleus of ordinary hydrogen (one proton). Deuterium has an atomic weight of 2.014.
How many protons neutrons and electrons does deuterium have?
Deuterium is a hydrogen isotope consisting of one proton, one neutron and one electron. It has major applications in nuclear magnetic resonance studies.
How many neutrons are in hydrogen isotope?
Most hydrogen atoms have no neutron. However, rare isotopes of hydrogen, called deuterium and tritium, have one and two neutrons each, respectively.
How many neutrons does deuterium and tritium have?
Deuterium and tritium are isotopes of hydrogen, the most abundant element in the universe. Whereas all isotopes of hydrogen have one proton, deuterium also has one neutron and tritium has two neutrons, so their ion masses are heavier than protium, the isotope of hydrogen with no neutrons.How many neutrons does tritium have?
tritium, (T, or 3H), the isotope of hydrogen with atomic weight of approximately 3. Its nucleus, consisting of one proton and two neutrons, has triple the mass of the nucleus of ordinary hydrogen.
How do you isolate deuterium?
Extracting this deuterium from seawater is a simple and well proven industrial process. “Heavy water”, or D2O (water in which deuterium substitutes for hydrogen), is first separated from regular water by chemical exchange processes, and is then submitted to electrolysis in order to obtain deuterium gas.
How many protons does deuterium have?
deuterium, (D, or 2H), also called heavy hydrogen, isotope of hydrogen with a nucleus consisting of one proton and one neutron, which is double the mass of the nucleus of ordinary hydrogen (one proton). Deuterium has an atomic weight of 2.014.
Why does deuterium have a spin of 1?
In the first case the deuteron is a spin triplet, so that its total spin s is 1. It also has an even parity and therefore even orbital angular momentum l ; The lower its orbital angular momentum, the lower its energy. Therefore, the lowest possible energy state has s = 1, l = 0.How many neutrons does sulfur have?
NameCarbonProtons6Neutrons6Electrons6Atomic Number (Z)6
How many neutrons does calcium have?Atomic elements in calcium Since a stable atom has a net charge of 0, we must have 20 electrons. The number of neutrons will be the same as the number of protons: it is also 20.
Article first time published onHow many neutrons does hydrogen 2 deuterium have?
GeneralNamesdeuterium, H-2, hydrogen-2, DProtons1Neutrons1Nuclide data
How is deuterium formed?
Deuterium is made by separating naturally-occurring heavy water from a large volume of natural water. Deuterium could be produced in a nuclear reactor, but the method is not cost-effective.
How do u find neutrons?
The atomic mass equals the number of protons plus the number of neutrons, so you find the number of neutrons by subtracting the number of protons (i.e. the atomic number) from the atomic mass (in atomic mass units).
What is protium deuterium and tritium?
Definition. Protium: Protium is an isotope of Hydrogen that is composed of one proton and one electron. Deuterium: Deuterium is an isotope of Hydrogen that is composed of one proton, one neutron, and one electron. Tritium: Tritium is an isotope of hydrogen that is composed of one proton, two neutrons, and one electron.
How many neutrons are in Boron?
The mass number for boron is 5; this The atomic mass of boron (symbol B) is 10.8. To determine the number of neutrons we round 10.8 to 11 and subtract the atomic number (5) and get 6; therefore, boron has 6 neutrons.
How many neutrons are in fluorine?
Reason: Consider fluorine atoms with 9 protons and 10 neutrons.
What is the molar mass of deuterium?
PubChem CID24523Molecular FormulaH2SynonymsDEUTERIUM Dideuterium 7782-39-0 Heavy hydrogen UNII-AR09D82C7G More…Molecular Weight4.0282035557DatesModify 2021-12-05 Create 2004-09-16
What is the mass of deuteron?
deuteron massNumerical value3.343 583 7724 x 10-27 kgStandard uncertainty0.000 000 0010 x 10-27 kgRelative standard uncertainty3.0 x 10-10Concise form3.343 583 7724(10) x 10-27 kg
Do all carbon atoms have 6 neutrons?
Every carbon atom has six protons, and the majority of carbon atoms have six neutrons. A carbon-12 atom has 6 protons (6P) and 6 neutrons (6N). But some types of carbon have more than six neutrons. We call forms of elements that have a different number of neutrons, isotopes.
How much deuterium is in tap water?
Deuterium-depleted water usually contains about 125 ppm.
How much deuterium is in a liter of water?
It is water with reduced deuterium content. Deuterium is a “bad” isotope form of hydrogen, found in any naturally accrued water. On average every natural water has 150 parts per million of deuterium per litre, so every 1 hydrogen atom out of 6000 is deuterium.
Can you drink deuterium?
While heavy water isn’t radioactive, it’s not entirely safe to drink. … Basically, the mass difference slows biochemical reactions that use water. Also, deuterium forms stronger hydrogen bonds than protium, resulting in a different reactivity. You can drink a glass of heavy water and won’t suffer any ill effects.
How many neutrons does Copernicium have?
On a microscopic scale, this is the atom, with its electrons orbiting around the nucleus and, in the case of copernicium, 112 electrons moving around a nucleus consisting of 112 protons and 165 neutrons.
How many electrons does sulfide have?
A neutral sulfur atom has an atomic number equal to 16 , which means that it has 16 protons inside its nucleus and 16 electrons surrounding its nucleus.
How many electrons does ca2+ have?
Ca2+ represents an ion with 20 protons and 18 electrons.
What is the spin of deuterium?
Deuterium is an isotope with spin = 1, unlike hydrogen which is spin = 1/2. Deuterium NMR has a range of chemical shift similar to proton NMR but with poor resolution.
Which country is rich in deuterium?
The Philippines was identified by some in the scientific community to hold the greatest amount of deuterium deposits in the world somewhere in the area called the Philippine Trench or Mindanao Deep, the part of the Pacific Ocean running mostly the entire length of the country.
Why is deuterium 2h1 called heavy hydrogen?
Deuterium is also known as “heavy hydrogen” because of the extra neutron in the atomic nucleus. [Hydrogen has a third isotope called tritium, with a proton and two neutrons, but it radioactively decays in a short time and is unimportant for the observations described here.]
How many neutrons does platinum have?
Platinum atoms have 78 electrons and 78 protons with 117 neutrons in the most abundant isotope.
Which of the following pairs are isobars?
Here, Calcium and Argon are different elements with the same mass numbers and different atomic numbers. Hence they are isobars.
What is the mass and charge of deuteron?
Deuteron Mass and Charge Deuterium is a stable atomic particle containing a proton and neutron. It is denoted by D or 2H and is called Hydrogen-2. Mass of deuteron is expressed in terms of an atomic mass unit (amu) or electron volts (eV). The Charge of deuteron is +1e.