How many protons does Protium have
protium, isotope of hydrogen (q.v.) with atomic weight of approximately 1; its nucleus consists of only one proton.
How many protons neutrons and electrons are in Protium?
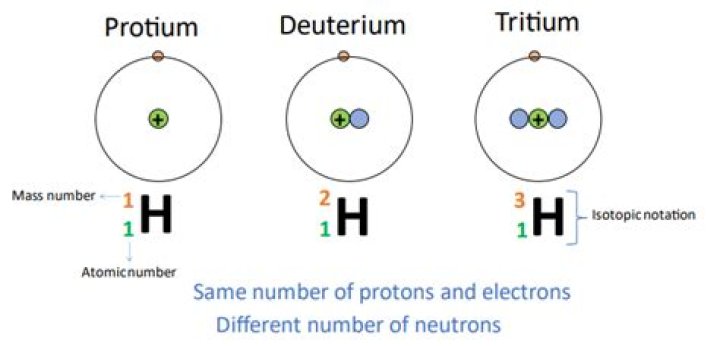
Protium is the most prevalent hydrogen isotope, with an abundance of 99.98%. It consists of one proton and one electron. It is typically not found in its monoatomic form, but bonded with itself (H2) or other elements. Deuterium is a hydrogen isotope consisting of one proton, one neutron and one electron.
How many protons does deuterium have?
deuterium, (D, or 2H), also called heavy hydrogen, isotope of hydrogen with a nucleus consisting of one proton and one neutron, which is double the mass of the nucleus of ordinary hydrogen (one proton). Deuterium has an atomic weight of 2.014.
How many neutrons does an Protium have?
The main difference between protium, deuterium, and tritium is that protium has no neutrons in its nuclei, whereas deuterium is composed of one neutron and tritium is composed of two neutrons [17]. Protium is an isotope of hydrogen that is composed of one proton and one electron.How many protons electrons and neutrons do the three isotopes of hydrogen have?
All three forms have one proton (pink) and one electron (dark green) but differ in the number of neutrons (gray) in the nucleus. Protium, or ordinary hydrogen (top), has no neutrons. Deuterium, or hydrogen-2 (bottom left) has one neutron.
How do you write protium?
Protium is represented by the symbol H. Its atomic number is 1 and mass number is also 1. It has one proton in its nucleus and one electron in its 1s orbital. Naturally occurring hydrogen contains 99.985% of this isotope.
What is the chemical formula of protium?
PubChem CID5460654Molecular FormulaHSynonymsmonoprotium hydrogen radical protium(.) CHEBI:29305 p(+)e(-) More…Molecular Weight1.0078250319DatesModify 2021-12-18 Create 2004-09-16
Is protium an isotope of hydrogen?
protium, isotope of hydrogen (q.v.) with atomic weight of approximately 1; its nucleus consists of only one proton. Ordinary hydrogen is made up almost entirely of protium.Why are there no neutrons in protium?
Ans- Because in hydrogen, you’ve only got one proton in the nucleus. So no repulsion, and no need for neutrons.
Is hydrogen the same as protium?Hydrogen can refer to any isotope of the element, while protium is a specific isotope, similar to dueterium and tritium.
Article first time published onWhat's the definition of Protium?
Definition of protium : the ordinary light hydrogen isotope of atomic mass 1.
What is protium deuterium and tritium?
Definition. Protium: Protium is an isotope of Hydrogen that is composed of one proton and one electron. Deuterium: Deuterium is an isotope of Hydrogen that is composed of one proton, one neutron, and one electron. Tritium: Tritium is an isotope of hydrogen that is composed of one proton, two neutrons, and one electron.
How many more neutrons does thorium 230 have than protons?
Therefore, thorium-230 has 50 more neutrons than it does protons. Therefore, thorium-230 has 90 electrons.
Why is Protium most abundant?
This is because the nucleus of the Protium isotope contains a single proton. … Tritium contains one proton and two neutrons. Only a trace amount of tritium exists in the environment. Thus the most abundant isotope of hydrogen is Protium.
How many protons are in the isotope hydrogen?
Elementary ParticleChargeMassElectron−1~0
What hydrogen cells is protium used in?
99.98% of hydrogen atoms are protium. It is used in hydrogen fuel cells and the production of plastics.
Is protium an element?
Protium may refer to: Protium (isotope) or hydrogen-1, the most common isotope of the element hydrogen, with one proton, one electron, and no neutrons.
What is difference between proton and protium?
is that proton is (particle) positively charged subatomic particle forming part of the nucleus of an atom and determining the atomic number of an element; the nucleus of the most common isotope of hydrogen; composed of two up quarks and a down quark while protium is (isotope) the lightest and most common isotope of …
Where is Protium on the periodic table?
Protium atom is the stable isotope of hydrogen with relative atomic mass 1.007825 and a natural abundance of 99.9885 atom percent (from Greek pirhoomegatauomicronsigma, first). It contains a proton. The first chemical element in the periodic table with atomic symbol H, and atomic number 1.
How many quarks are in Protium?
Although we usually say that a proton contains three quarks (up, up and down), there are many more quark-antiquark pairs at fine scales. Matter is made of molecules, which are made of atoms, which are primarily made of protons and neutrons, which are made of quarks.
How is Protium used?
Protium is a selective “proton pump inhibitor”, a medicine which reduces the amount of acid produced in your stomach. It is used for treating acid-related diseases of the stomach and intestine.
How do u find neutrons?
Subtract the atomic number from the atomic mass. Since the vast majority of an atom’s mass is made up of its protons and neutrons, subtracting the number of protons (i.e. the atomic number) from the atomic mass will give you the calculated number of neutrons in the atom.
Why is the atomic mass of hydrogen 1?
Hydrogen is the first and the lightest element of the periodic table, which contains only 1 electron, and so has an atomic number of 1. Apart from 1 electron, an isolated hydrogen atom contains 1 proton, and no neutrons. … As this factor is simplified as 1.007 amu, therefore, hydrogen has a mass of 1.007 amu.
Why is Protium an isotope?
Hydrogen-1 (Protium) Because the nucleus of this isotope consists of only a single proton, it is given the formal name protium. The proton has never been observed to decay, and hydrogen-1 is therefore considered a stable isotope.
What is Protium 40mg?
Protium is a selective “proton pump inhibitor”, a medicine which reduces the amount of acid produced in your stomach. It is used for treating acid-related diseases of the stomach and intestine. Protium is used to treat adults and adolescents 12 years of age and above for. • Reflux oesophagitis.
How many neutrons are present in protium deuterium and tritium respectively?
Protium contains a proton, an electron and one neutron, whereas deuterium has 1 proton and a neutron. In tritium, only a proton and 2 neutrons are present.
How many neutrons are in Boron?
The mass number for boron is 5; this The atomic mass of boron (symbol B) is 10.8. To determine the number of neutrons we round 10.8 to 11 and subtract the atomic number (5) and get 6; therefore, boron has 6 neutrons.
How many protons are in a carbon atom?
Every carbon atom has six protons, and the majority of carbon atoms have six neutrons. A carbon-12 atom has 6 protons (6P) and 6 neutrons (6N). But some types of carbon have more than six neutrons. We call forms of elements that have a different number of neutrons, isotopes.
Who discovered protium?
Hydrogen discovery The element was named hydrogen by the French chemist Antoine Lavoisier. Hydrogen has three common isotopes: protium, which is just ordinary hydrogen; deuterium, a stable isotope discovered in 1932 by Harold C. Urey; and tritium, an unstable isotope discovered in 1934, according to Jefferson Lab.
Can protium be used in nuclear fusion?
The reason the sun can fuse protium is because the sun crushes the protium so close together that it actually slows down proton emission. Hence, it has a higher chance of beta decaying to form deuterium. Fusing protium is possible because the sun fuses protium to make deuterium. It just is very impractical.
Where does the word protean come from?
Protean (adj.) “of or pertaining to the Greek sea-god Proteus,” 1590s, from Greek Prōteus, son of Oceanus and Tethys, who could change his form at will; hence, “readily assuming different shapes, exceedingly variable.” His name is literally “first,” from prōtos “first” (see proto-).



