How many valence electrons are in a neutral atom of potassium
Potassium, an element in Group 1 of the periodic table(alkaline metals) has 1 valence electron. Number of valence(outer shell) electrons corresponds to the element’s group number and therefore it has 1 valence electron.
How many valence electrons are in a neutral atom of potassium? - Google Search
b. This tells us that in an atom of K there are 19 protons and 19 electrons.
Is potassium a neutral atom?
A neutral atom of potassium has an atomic number of 19 and an atomic mass number of 39.
Why is potassium neutral?
In a neutral atom, the number of protons and electrons are equal to each other. … If, however, there are fewer electrons than protons as in the case of the potassium ion, the net charge is positive since there is one more positive proton than negative electrons [(+19) + (−18) = +1].Does K+ have 8 valence electrons?
Potassium has one valence electron. This means that there is one electron in its outermost shell (4th shell). Potassium ion, on the other hand, loses an electron and has a complete octet (has eight valence electrons) in its 3rd shell.
How many valence electrons does CL have?
The atomic number of chlorine is 17. Hence it has got 7 electrons in its outermost shell. There are 7 valence electrons in the chlorine atom.
How many valence electrons do have?
Iodine has seven valence electrons.
How many electron does potassium have?
So if you look for potassium (symbol K in the periodic table), you will find that it has the atomic number of 19. This tells you that potassium has 19 protons and – since the number of protons is the same as the number of electrons- also 19 electrons.Why does potassium only have 1 valence electron?
Potassium is an alkali metal. So it contains 1 electron in its valenced shell or orbital. So its valency is also 1.
What happens to potassium valence electrons?In order to obtain eight valence electrons (an octet), the potassium atom will transfer its single valence electron to the fluorine atom. … This also produces a potassium cation with a 1+ charge with the formula K+ , and a fluoride anion with a 1− with the formula F− .
Article first time published onHow do you find the valence electrons in a neutral atom?
For neutral atoms, the number of valence electrons is equal to the atom’s main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.
How many valence electrons does a neutral atom with 4 protons have?
As you can see, a neutral calcium atom has 2 valence electrons. You can tell by looking at the number of electrons present in the outermost energy shell of the atom, i.e. in the valence shell. In this case, calcium’s outermost energy level, which corresponds to n=4 , contains 2 electrons.
How many protons electrons and neutrons are in a neutral atom of potassium having an atomic weight of 39 and atomic number 19?
19 protons, 20 neutrons and 18 electrons.
How many electrons does potassium have in its outside electron shell?
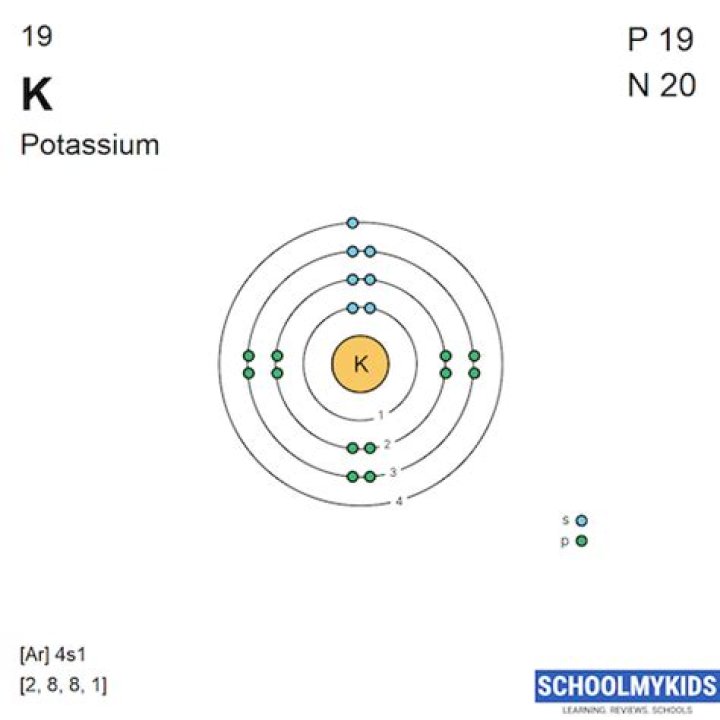
Potassium atoms have 19 electrons and 19 protons with one valence electron in the outer shell.
How many valence electrons does an atom acquire?
Referring to the octet rule, atoms attempt to get a noble gas electron configuration, which is eight valence electrons.
How many electrons are in the atom?
ElementOxygenSymbolOAtomic No.8Number of Electrons in Each ShellFirst2Second6
What are the orbital of potassium?
In writing the electron configuration for Potassium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Potassium go in the 2s orbital. The next six electrons will go in the 2p orbital. The p orbital can hold up to six electrons.
Does potassium have 2 valence electrons?
Explanation: Just like sodium potassium has one valence electron and is highly reactive.
Does potassium have three valence electrons?
The total number of electrons in a valence shell(last shell) is called a valence electron. The electron configuration of potassium(K) shows that the last shell(orbit) of potassium has one electron. Therefore, the valence electron of potassium(K) is one.
Does potassium have 4 orbitals?
Potassium has access to its 1s , 2s , 2p , 3s , 3p , and 4s orbitals. Its electron configuration is 1s22s22p63s23p64s1 .
What is valence electron with example?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen’s valence electrons as 2s²2p⁴.
Does CL have 1 valence electron?
Chlorine has seven valence electrons. Chlorine has atomic number 17. The atomic number is the number of protons present in the nucleus of an atom. All the atoms of an element have same atomic number.
Can there be 0 valence electrons?
Valence electrons are the outer electrons that are involved in bonding. Only electrons in the s and p orbitals are valance electrons, so a given atom can have between 0 and 7 valance electrons. Atoms with 0 valence electrons are called noble gases and don t like form bonds.
How many core and valence electrons does potassium have?
There is 1 valence electron in potassium.
How many electrons in a potassium atom can form a bond?
Because there is only one electron, the element is very reactive and searches out other elements to make new compounds. You will learn about many salts where alkali metals bond with halogens that are looking for an extra electron. Potassium is so reactive that it can burn when introduced to water.
How many energy levels and valence electrons does potassium have?
Explain that potassium has 19 protons and 19 electrons. There are 2 electrons on the first energy level, 8 electrons on the second level, 8 electrons on the third energy level, and 1 on the fourth energy level.



