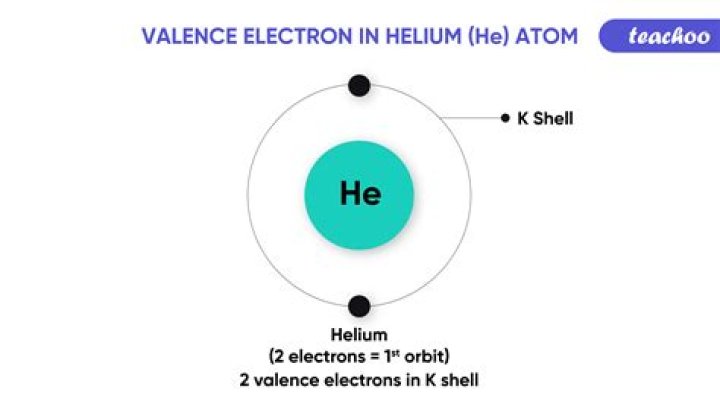
How many valence electrons are in thallium
Characteristics. A thallium atom has 81 electrons, arranged in the electron configuration [Xe]4f145d106s26p1; of these, the three outermost electrons in the sixth shell are valence electrons.
How many electrons does thallium have in its outermost shell?
Aluminum, gallium, indium, and thallium have three electrons in their outermost shell (a full s orbital and one electron in the p orbital) with the valence electron configuration ns2np1.
What is an example of a valence electron?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell.
How many valence electrons are in the element be?
Lithium has a single electron in the second principal energy level and so we say that lithium has one valence electron. Beryllium has two valence electrons.How many valence electrons does a copper atom have?
Copper (Cu) has two valences Cu I (cuprous) has one valence electron and Cu II (cupric) has two valence electrons.
How many valence electrons does xenon have?
Xenon right over here. It is a noble gas. It has eight valence electrons. One, two, three, four, five, six, seven, eight in that fifth shell.
What is the group number of thallium?
Group13304°C, 579°F, 577 KPeriod61473°C, 2683°F, 1746 KBlockp11.8Atomic number81204.38State at 20°CSolid205Tl
Can there be 9 valence electrons?
Re: Having more than 8 valence electrons Hey there! Some molecules can accommodate more than 8 valence electrons because they have expanded valence shells. These molecules are from periods 3 or higher because starting from n=3, atoms have d-orbitals that can accommodate for more that 9 valence electrons.Why does thallium form a 1+ ion?
It has the electron configuration [Xe] 2s22p1 and has a +3 or +1 oxidation state. As stated above, because thallium is heavy, it has a greater stability in the +1 oxidation state (inert pair effect). Therefore, it is found more commonly in its +1 oxidation state.
Does iodine have 5 valence?Iodine has seven valence electrons. Valence electrons of an atom are located in the outermost shell of the atom and participate in bonding. There are 7 valence shell electrons and 46 inner shell electrons in the ground state of iodine.
Article first time published onWhich of the following has 8 valence electrons?
Atoms of neon (Ne), argon (Ar), krypton (Kr), and xenon (Xe) have 8 valence electrons. These elements are nonreactive, or stable.
How many valence electrons does chlorine?
The atomic number of chlorine is 17. Hence it has got 7 electrons in its outermost shell. There are 7 valence electrons in the chlorine atom.
What is the importance of 8 valence electrons?
The rule of 8 or the Octet rule is the tendency of atoms to have eight electrons in their valence shell. Eight electrons in this final shell allow atoms to be stable and non-reactive. Noble gases, for example, are some of the most non-reactive chemical elements one can find in nature.
What is meant by valence electron?
Definition of valence electron : a single electron or one of two or more electrons in the outer shell of an atom that is responsible for the chemical properties of the atom.
Does copper have 1 or 11 valence electrons?
The element copper has 11 valence electrons, distributed amongst the outermost d and s orbitals. These electrons are the ones that engage in chemical reactions that involve photons corresponding to visible light. The inner electrons require far higher energies to liberate, and do not figure in chemical reactions.
Why does copper only have 1 valence electron?
Copper has excpetional electronic configuration of [Ar]3d10 4s1 rather than the normal configuration of [Ar]3d9 4s2 . The former is more stable due to increase in exchange energy and you can see it has a single electron in its outermosrt 4s shell. Therefore it has a single Valence electron in the ground state.
What is the outermost electron of copper?
Outermost electron in copper is 4s.
What classification is thallium in?
Thallium is a chemical element with symbol Tl and atomic number 81. Classified as a post-transition metal, Thallium is a solid at room temperature.
What is thallium in?
Thallium uses today commonly include the production of electronic devices, fiber optics, camera lenses, switches, and closures. Thallium metal is used most notably by the semiconductor, fiber optic, and the glass lens industries.
How do you find the valence electrons of xenon?
The atomic number of xenon is 54. The electronic configuration of xenon is $[Kr]4{d^{10}}5{s^2}5{p^6}$. It has 8 valence electrons in its outermost electronic configuration so it can share its 8 electrons with other atoms.
How many valence electrons are in sulfur?
The superscripts associated with these orbitals total to 6. Therefore, sulfur has 6 valence electrons.
How can thallium form a 1+ ion rather than a 3+ ion in some of its compounds?
Thallium forms its oxides in two different oxidation states, +1 (Tl2O) and +3 (Tl2O3). … Thus, thallium, unlike the other boron group elements, predominantly forms singly charged thallium salts having thallium in the +1 rather than the +3 oxidation state (the 6s2 electrons remain unused).
When was thallium banned?
Thallium was also widely used as a rodenticide. Its use as a household rodenticide was banned in the United States in 1965 after multiple unintentional poisonings.
How much does thallium cost?
Commercial thallium metal (99%) costs about $40/lb.
Why can iodine have 10 electrons?
So, iodine can use all of these electrons in chemical bonding: 2 in the 5s, 5 in the 5p and up to 10 in the 4d, although it only needs to use 4 of its 4d electrons to do the bonding in this molecule. Hence it can have more than 8 electrons involved in its bonding.
Do metals have 8 valence electrons?
Periodic table blockPeriodic table groupValence electronspGroup 18 (VIII or 0) (noble gases) except helium8
How many valence electrons are in rhodium?
Atomic Number45Electron Configuration[Kr] 4d8 5s1Valence Electrons4d8 5s1Oxidation State-3;-1 1;2;3;4;5;6Atomic Term Symbol (Quantum Numbers)4F9/2
Does iodine have 7 valence electrons?
Iodine has 7 valence electrons in the valence shell which means that there are 7 electrons in its outer shell. A neutral iodine atom will have a total of 53 electrons around its nucleus.
How many valence electrons does iodine and chlorine have?
Iodine has 7 electrons in the outer shell and Chlorine has 7 electrons in its outermost shell so will gain 1 more to complete the octet.
How many valence electrons does Group 2 have?
Group 1 has 1 valence electron. Group 2 has 2 valence electrons. Group 13 has 3 valence electrons.
What family has 7 valence electrons?
Any element in the halogen group will have seven valence electrons. These elements include fluorine, chlorine, bromine, iodine, and astatine.