In which reaction do the reactants have a lower potential energy than the products
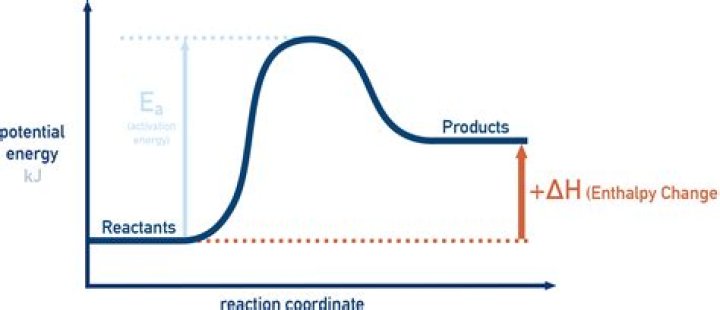
In an exothermic reaction, energy is released. The products have a lower potential energy than the reactants, and the sign of ΔH is negative. In an endothermic reaction, energy is absorbed.
In what type of reaction do the products of the reaction always possess less potential energy than the reactants?
In exothermic reactions, the products have less enthalpy than the reactants, and as a result, an exothermic reaction is said to have a negative enthalpy of reaction. This means that the energy required to break the bonds in the reactants is less than the energy released when new bonds form in the products.
Do the reactants or the products have the lower potential energy in an endothermic reaction?
The energy change ΔH is positive for an endothermic reaction. … The products have a lower potential energy than the reactants, and the sign of ΔH is negative. In an endothermic reaction, energy is absorbed. The products have a higher potential energy than the reactants, and the sign of ΔH is positive.
Why the energy of the product might be lower than the energy of the reactants?
Energy Diagrams In endothermic reactions, the reactants have higher bond energy (stronger bonds) than the products. Strong bonds have lower potential energy than weak bonds. Hence, the energy of the reactants is lower than that of the products.When the reactants have less enthalpy than the products?
In exothermic reactions, the products have less enthalpy than the reactants, and as a result, an exothermic reaction is said to have a negative enthalpy of reaction. This means that the energy required to break the bonds in the reactants is less than the energy released when new bonds form in the products.
How does the energy of the reactants and products differ between an energy absorbing reaction and an energy releasing reaction?
Energy is used to break bonds in reactants, and energy is released when new bonds form in products. Endothermic reactions absorb energy, and exothermic reactions release energy.
Which reaction generates the product lower in energy?
Endothermic and Exothermic Definitions Exothermic reactions release energy to their surroundings, because the products are lower in energy than the reactants.
How does the potential energy of reactants compare to the potential energy of products in an endothermic reaction?
In the case of an endothermic reaction, the reactants are at a lower energy level compared to the products—as shown in the energy diagram below. In other words, the products are less stable than the reactants.Do reactants in an exothermic reaction have a higher or lower energy than the products?
In an exothermic reaction, the reactants have a relatively high quantity of energy compared to the products. As the reaction proceeds, energy is released into the surroundings. … Since the energy of the system decreases during an exothermic reaction, the products of the system are more stable than the reactants.
Do the products have more or less potential energy than the reactants in an exothermic reaction?3 Answers. The simple answer is yes – as energy is required to be absorbed for the reaction, the products have more energy than the reactants.
Article first time published onWhen an reaction occurs the reactants are at a lower energy?
In an exothermic reaction, the reaction mixture releases heat to the surroundings. Therefore, the products will have a lower energy than the reactants and ΔH will be negative.
When the products of a reaction have a higher enthalpy than the reactants?
Endothermic reaction: In an endothermic reaction, the products are higher in energy than the reactants. Therefore, the change in enthalpy is positive, and heat is absorbed from the surroundings by the reaction.
How does the enthalpy of the products compare with the enthalpy of the reactants in an exothermic reaction an endothermic reaction?
An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat. … A system of reactants that absorbs heat from the surroundings in an endothermic reaction has a positive ΔH, because the enthalpy of the products is higher than the enthalpy of the reactants of the system.
What are the reactants for the endothermic reaction used in sports injury packs?
Everyday uses of endothermic reactions include instant ice packs which can be used to treat sports injuries. The slideshow describes an exothermic reaction between dilute sodium hydroxide and hydrochloric acid and an endothermic reaction between sodium carbonate and ethanoic acid.
Which reaction is exothermic quizlet?
All combustion reactions are exothermic.
What is an endothermic reaction equation?
The general equation for an endothermic reaction is: Reactants + Energy → Products. … In endothermic reactions, the temperature of the products is typically lower than the temperature of the reactants.
When the reactants have more potential energy than the products have and the extra energy is released to the surroundings it is called?
Energy level diagrams for exothermic reactions In an exothermic reaction, reactants have more energy than the products. The difference between these two energy levels is the energy released to the surroundings, shown as a vertical drop from a higher to a lower level.
Is the energy difference between reactants and products in a chemical reaction?
The balance between reactants and products in a reaction will be determined by the free energy difference between the two sides of the reaction. The greater the free energy difference, the more the reaction will favor one side or the other.
What is the relationship between the energy of the reactants and the energy of the products?
Explanation: The products have less energy than the reactants. The energy given off is experienced as heat and sometimes also light.
Do endothermic reactions have higher potential energy?
The total potential energy of the system increases for the endothermic reaction as the system absorbs energy from the surroundings. The total potential energy of the system decreases for the exothermic reaction as the system releases energy to the surroundings.
Which type of reaction produces greater energy in the reactants?
Endothermic reactionIn an endothermic reaction, the products are higher in energy than the reactants. Therefore, the change in enthalpy is positive, and heat is absorbed from the surroundings by the reaction.
Which reactions are exothermic?
- The thermite reaction.
- A neutralization reaction (e.g., mixing an acid and a base to form a salt and water)
- Most polymerization reactions.
- Combustion of a fuel.
- Respiration.
- Nuclear fission.
- Corrosion of metal (an oxidation reaction)
- Dissolving an acid in water.
Why do reactants have more energy than products in exothermic reactions?
In exothermic reactions, more energy is released when the bonds are formed in the products than is used to break the bonds in the reactants.
What is endothermic reaction Class 10th?
Endothermic reactions are chemical reactions in which the reactants absorb heat energy from the surroundings to form products.
How is total reaction energy calculated?
To calculate an energy change for a reaction: add together the bond energies for all the bonds in the reactants – this is the ‘energy in’ add together the bond energies for all the bonds in the products – this is the ‘energy out’
What happens to the rate of reaction when you decrease the temperature of a chemical reaction?
When you lower the temperature, the molecules are slower and collide less. That temperature drop lowers the rate of the reaction. … That greater density of molecules increases the number of collisions. When you decrease the pressure, molecules don’t hit each other as often and the rate of reaction decreases.
What does it mean to have a lower enthalpy?
Enthalpy (ΔH) refers to the total heat content of the system, if the change in enthalpy is positive, the reaction is endothermic in nature, and a lower enthalpy means it requires less energy.
How do you know if enthalpy increases or decreases?
It is the heat content of a system. The heat that passes into or out of the system during a reaction is the enthalpy change. Whether the enthalpy of the system increases (i.e. when energy is added) or decreases (because energy is given off) is a crucial factor that determines whether a reaction can happen.
Are the products of an exergonic reaction more or less stable than the reactants?
In exergonic reaction the free energy of the products is lower than that of the reactants; meanwhile in endergonic the free energy of the products is higher than that of the reactants. The building of complex molecules, such as sugars, from simpler ones is an anabolic process and is endergonic.
How does the enthalpy of the products of a reaction system compared with the enthalpy of the reactants when the reaction is?
How does the enthalpy of the products of a reaction system compare with the enthalpy of reactants when the reaction is endothermic and exothermic? … Overall enthalpy change in a reaction is equal to the sum of enthalpy changes for the individual steps in he process. Used to calculate enthalpies of reactions.
Why does temperature decrease in endothermic reactions?
In an endothermic change, temperature is absorbed from surrounding molecules to continue reacting. If these molecules are losing heat, that means their temperature will drop, resulting in a temperature decrease.



