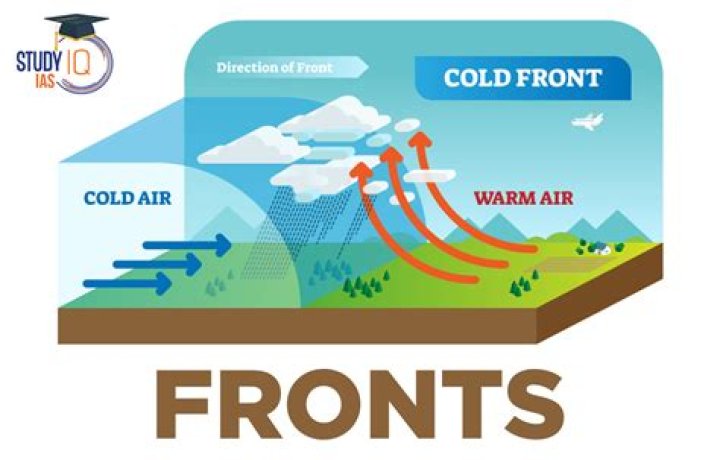
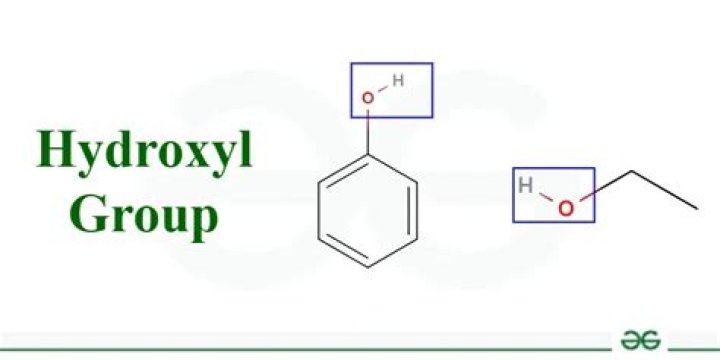
Is a hydroxyl group polar or nonpolar
Functional GroupPropertiesHydroxylPolarMethylNonpolarCarbonylPolar
Why are hydroxyl groups polar?
Hydroxyl groups are polar. … Because polarity causes attraction between molecules. We know that opposites attract, so the positive side of a molecule will be attracted to the negative side of another molecule. Water is a polar molecule as well, so it will be attracted to molecules containing hydroxyl groups.
Is hydroxyl group positive or negative?
In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion HO−, called hydroxide, and the neutral radical HO•, known as the hydroxyl radical, consist of an unbonded hydroxyl group.
Is hydroxyl group hydrophobic or hydrophilic?
Hydroxyl groups (-OH), found in alcohols, are polar and therefore hydrophilic (water liking) but their carbon chain portion is non-polar which make them hydrophobic.Are hydroxyl groups polar and water soluble?
One of the most common groups is the -OH (hydroxyl) group. Its presence will enable a molecule to be water soluble. Isomers are molecules with identical molecular formulas but differ in arrangement of their atoms (e.g., glyceraldehyde and dihydroxyacetone).
Is oh a polar covalent bond?
A molecule which has one or more polar covalent bonds may have a dipole moment as a result of the accumulated bond dipoles. In the case of water, we know that the O-H covalent bond is polar, due to the different electronegativities of hydrogen and oxygen.
What does a hydroxyl group do?
2.5 Hydroxyl Groups They readily participate in hydrogen bonding, generating either a net positively or negatively charged ion. This group can also participate in chemical reactions to link molecules together, forming chains of sugars or fatty acids.
Is starch hydrophobic?
Starch is an inherently hydrophilic polymer that can easily absorb moisture due to the presence of hydroxyl groups; but this feature weakens its mechanical properties.Are amides hydrophobic or hydrophilic?
SolubilityEdit Larger amides have trouble dissolving because of their long hydrophobic carbon chains. Amides are typically less soluble than amines and carboxylic acids because they can both donate and accept hydrogen bonds.
Are amines hydrophilic or hydrophobic?Physical Properties of Amines Amines also display some solubility in water. However, the solubility decreases with an increase in carbon atoms, due to the increased hydrophobicity of the compound as the chain length increases.
Article first time published onWhy are hydroxyl groups reactive?
The functional group of the alcohols is the hydroxyl group, –OH. Unlike the alkyl halides, this group has two reactive covalent bonds, the C–O bond and the O–H bond. … Consequently, the covalent bonds of this functional group are polarized so that oxygen is electron rich and both carbon and hydrogen are electrophilic.
Is hydroxyl a functional group?
A functional group may participate in a variety of chemical reactions. Some of the important functional groups in biological molecules are shown above; they include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl (R-S-H).
Is hydroxyl group a base?
Non-acidic Functional Groups The hydroxyl, which is simply an OH attached to the molecule, could conceivably lose it’s hydrogen, making it acidic, but that is not how the molecule normally interacts.
Is a hydroxyl group water soluble?
Hydroxyl groups are also able to form hydrogen bonds with water, a property that increases the hydrophilicity and solubility of molecules containing them. The carbohydrates are an example of a group of molecules that are extremely soluble due to hydroxyl functional groups.
Why are hydroxyl groups soluble in water?
Due to the high electronegativity of the oxygen atom, the bond between the oxygen and the hydrogen is highly polar. Consequently, the hydroxyl group strongly attracts water molecules, forming hydrogen bonds. Hydroxyl groups are responsible for the solubility of some molecules such as sugars.
What is a hydroxyl group quizlet?
hydroxyl group: structure. —OH : A hyrdrogen atom bonded to an oxygen atom, bonded to the carbon skeleton of the organic molecule. hydroxyl group: compound name. Alcohols – names usually end in “-ol”
What is a hydroxyl group in DNA?
While DNA contains deoxyribose, RNA contains ribose, characterised by the presence of the 2′-hydroxyl group on the pentose ring (Figure 5). This hydroxyl group make RNA less stable than DNA because it is more susceptible to hydrolysis.
What macromolecule is hydroxyl in?
Hydroxyl groups appear on carbohydrates (A glucose), on some amino acids (B Threonine), and on nucleic acids (C Adenine).
What is a hydroxyl biology?
Hydroxyl. (Science: chemistry) a compound radical, or unsaturated group, ho, consisting of one atom of hydrogen and one of oxygen. It is a characteristic part of the hydrates, the alcohols, the oxygen acids, etc.
What type of bonding is OH?
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile, and a catalyst.
Is the C OH bond polar or nonpolar?
Each C – O bond is polar since oxygen (3.5) is more electronegative than carbon (2.5), however, these bonds are arranged symmetrically (all angles are 180°) so that the two dipoles cancel out resulting in no net dipole for the molecule.
Is OH nonpolar covalent polar covalent or ionic?
The bond between oxygen and hydrogen in the O-H is polar covalent because the electronegativity of oxygen is 3.44, while the EN for hydrogen is 2.20, a difference of 1.24. The oxygen pulls the electron pair closer so it has a partial negative charge, leaving the hydrogen with a partial positive charge.
Are amides polar?
Due to the tendency of the amide’s NH2 group to have more possibilities for hydrogen bonding compared to the carboxylic OH group, amides are more polar than carboxylic acids.
Do amides have strong intermolecular forces?
This arrangement results in a dipole-dipole force that is a strong attractive intermolecular force. Of all acid derivatives, amides have the highest boiling points. This is not only due to the dipole-dipole interaction; primary and secondary amides also experience hydrogen bonding.
What is an amide group?
amide group, a functional group –C(=O)N= consisting of a carbonyl adjacent to a nitrogen atom. cyclic amide or lactam, a cyclic compound with the amide group –C(=O)N– in the ring. metal amide, an ionic compound (“salt”) with the azanide anion H2N− (the conjugate base of ammonia) or to a derivative thereof R2N−.
What is a hydrophilic group?
Hydrophilic means “water-loving.” Chemical groups that tend to make substances hydrophilic include ionic (charged) groups and groups that contain oxygen or nitrogen atoms. … Cellulose fibers are hydrophilic due to the presence of -OH groups at their surfaces. The opposite of hydrophilic is hydrophobic, or water-hating.
Is glucose hydrophobic?
Glucose, a monosaccharide The presence of all this oxygen in the structure of the glucose molecule ensures that it is strongly hydrophilic (‘loves’ water).
What is hydrophilic molecule?
A hydrophilic molecule or portion of a molecule is typically charge-polarized and capable of hydrogen bonding, enabling it to dissolve more readily in water than in oil or other hydrophobic solvents. Hydrophilic substances can seem to attract water out of the air. The hydrophilic concept is used in many industries.
Are amines polar or nonpolar?
The N-H bonds in amines are somewhat polar. As we might guess from considering electronegativities (estimated from positions in the periodic table), the N-H bond is more polar than the C-H bond and less polar than the O-H bond. This polarity shows up in a comparision of physical properties of amines and alcohols.
Is the amino group polar?
An organic compound that contains an amino group is called an amine. Like oxygen, nitrogen is also more electronegative than both carbon and hydrogen, which results in the amino group displaying some polar character.
Which is an amine group?
In organic chemistry, amines (/əˈmiːn, ˈæmiːn/, UK also /ˈeɪmiːn/) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. … Important amines include amino acids, biogenic amines, trimethylamine, and aniline; see Category:Amines for a list of amines.