Is ammonium an acid or base
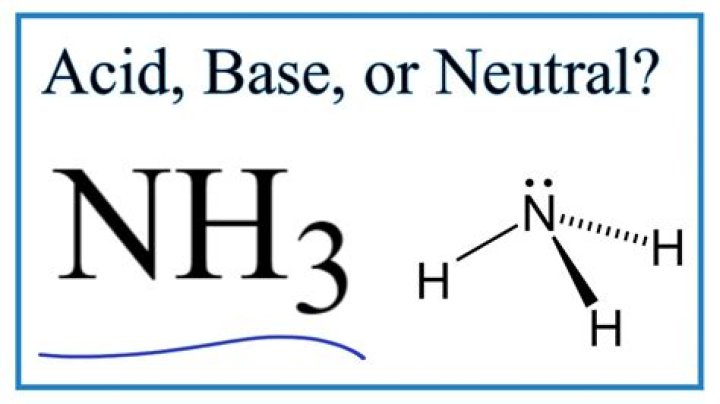
Ammonia is a weak base because its nitrogen atom has an electron pair that readily accepts a proton. Also, when dissolved in water, ammonia acquires hydrogen ions from water to produce hydroxide and ammonium ions. It is the production of these hydroxide ions that imparts ammonia its characteristic basicity.
Is ammonium a base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. … Only about 1% has actually produced hydroxide ions. A weak base is one which doesn’t convert fully into hydroxide ions in solution.
Is ammonium ion an acid?
Name of MoleculeAmmonium ionAcidity (pKa)9.25
Is NH4+ an acid or base?
NH3 is a weak base and dissociates partially in solution (as indicated with reversible arrow) to form OH- and NH4+ ions. Since this is a reversible process, NH4+ can donate H+ to form back NH3. Therefore the nature of NH4+ is acidic and we call NH4+ the conjugate acid of NH3.Is ammonium a strong acid?
Under certain conditions, the ammonium ion IS a strong acid. Saturated aqueous ammonium chloride solutions are routinely used in the workup of organic reactions, where mineral acids were used in the past. Well, ammonium ion is the conjugate acid of a moderately strong base…
Is ammonium a conjugate acid?
Ammonium, NH4+, is the conjugate acid of ammonia, NH3.
Can ammonia act as an acid?
Ammonia is normally a base, but in some reactions it can act like an acid. Ammonia acts as a base. It accepts a proton to form ammonium. Ammonia also acts as an acid.
Why is NH4+ a lewis acid?
Ammonium ion that is NH4+ has a positive charge on it so it has a tendency to accept a pair of electrons from some lewis bases which are electron pair donor, to change into a more stable form that is without any charge. So ammonium ion are considered as lewis acid.Why is NH4+ an acid?
NH4+ is acidic according to Bronsted-Lowry Theory because it can give away one proton to other species (like water or hydroxide ion).
Is ammonium a metal?Under normal conditions, ammonium does not exist as a pure metal, but does as an amalgam (alloy with mercury).
Article first time published onIs nh4no3 an acid?
Is ammonium nitrate a base or acid? Ammonium nitrate is not acid but a salt but the solution is acidic since it is a salt with a weak base (ammonium hydroxide) and a heavy nitric acid.
Is ammonium ionic or covalent?
NH3 (ammonia) is a covalent compound because the bond is formed between one nitrogen and three hydrogen atoms through the sharing of electrons with each other.
Is ammonia a base or alkali?
Ammonia is the hydrogen ion acceptor, and is therefore a base, and water is the hydrogen ion donor, and is therefore an acid.
Is ammonia alkaline or basic?
Ammonia is moderately basic; a 1.0 M aqueous solution has a pH of 11.6, and if a strong acid is added to such a solution until the solution is neutral (pH = 7), 99.4% of the ammonia molecules are protonated.
How does ammonia act as a base?
In the reaction of ammonia with water to give ammonium ions and hydroxide ions, ammonia acts as a base by accepting a proton from a water molecule, which in this case means that water is acting as an acid.
Is NH3 a base in water?
A substance that accepts and H+ from water is considered a base. Both NH3 and H2O are amphoteric (they have H atoms that can be donated as H+ ions and thus act as acids and lone-pair electrons that can accept an H+ and thus act as bases). Thus, either NH3 or H2O can act as an acid or a base.
Why does ammonia behave as a base when it reacts with an acid?
Why water acts as an acid when mixed with ammonia? Water acts like a acid with NH3. NH3 then acts like a base because it gets H+ proton. Bronsted Lowry explains this.
Is ammonium a conjugate base?
AcidConjugate baseNH + 4 Ammonium ionNH3 AmmoniaH2O Water (pH=7)OH− Hydroxide ion
Is ammonia a conjugate base or acid?
In this reaction, ammonia is the conjugate base and accepts a proton from water, where it becomes the conjugate acid ammonium. You can show that the lone pair of electrons on the nitrogen atom reacts with the hydrogen on water molecules, giving an ammonium ion and a hydroxide ion.
What is conjugate base of ammonia?
Ammonia less a proton is the amide ion, NH−2. NH3 can donate a proton & act as an acid. NH2− is the conjugate base of NH3.
Is NH4+ Arrhenius acid or base?
NH4+ is acidic according to Bronsted-Lowry Theory because it can give away one proton to other species (like water or hydroxide ion). However, it is neither acidic nor basic according to Lewis Theory because it doesn’t have any room to receive electron pairs and doesn’t have any lone electron pairs ti donate.
Is ammonium chloride a base?
Ammonium chloride, a water-soluble salt of ammonia, is slightly acidic as a water-based solution. The acidic component of ammonium chloride (Cl-) produces hydrogen (H+) ions when dissolved in water. The basic component (NH4+) produces hydroxide (OH-) ions when dissolved in water.
Is ammonium a Lewis base?
Ammonium ion is Lewis base.
Can NH4+ act as lewis acid?
No. A lewis acid is a chemical specie in which one of the atom requires a lone pair of electron in order to get a stable electronic configuration. In NH4+, all atoms are in stable electronic configuration and hence it isn’t a lewis acid.
Is ammonium a gas?
At room temperature, ammonia is a colorless, highly irritating gas with a pungent, suffocating odor. In pure form, it is known as anhydrous ammonia and is hygroscopic (readily absorbs moisture). … Ammonia gas dissolves easily in water to form ammonium hydroxide, a caustic solution and weak base.
Is NH4NO3 ionic or molecular?
Ionic bond. NH4NO3 is a nitrate salt of the ammonium cation. Since ammonium is a cation and bonds with the anion nitrate, hence the compound is bonded by an ionic bond.
Is ammonium an element?
Ammonium is not found on the periodic table of elements because it is a compound rather than an element.
Is NH4NO3 acidic or basic or neutral?
NH4NO3 is acidic salt, because it consists of NH3 and HNO3. NH3 is a weak base and HNO3 is a strong acid. So, overall the salt is acidic.
Is NH4NO3 a strong or weak acid or base?
Name of MoleculeAmmonium nitrateNatureAcidic salt
Is NH4NO3 a compound?
Ammonium nitrate is a chemical compound with the chemical formula NH4NO3. It is a white crystalline solid consisting of ions of ammonium and nitrate. It is highly soluble in water and hygroscopic as a solid, although it does not form hydrates.
Is ammonium ionic compound?
Ammonium Sulfate is an ionic compound formed by two polyatomic ions, Ammonium NH+4 and Sulfate SO−4 . In order for these two polyatomic ions to bond the charges must be equal and opposite.