Is aubagio a chemotherapy drug
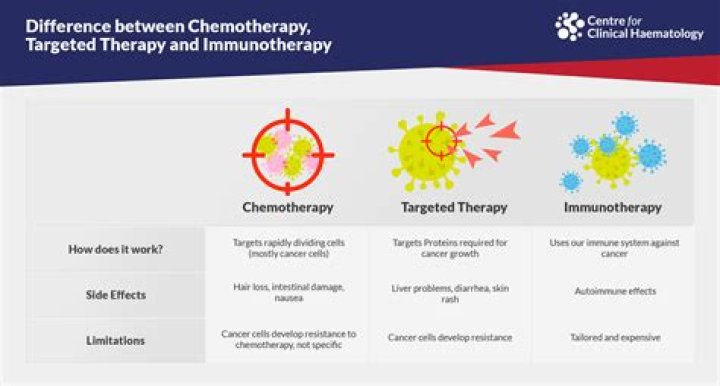
One doc called Aubagio a “mild chemotherapy” type drug and a “cousin” to the drug Leflunomide which is used for rheumatoid arthritis (another condition in which the body’s immune system attacks healthy tissue, like MS).
Are MS drugs chemotherapy?
This may slow down or stop disease activity in MS. Lemtrada (alemtuzumab), which is used to treat relapsing remitting MS, is a chemotherapy drug. Other drugs that have been used to treat MS include azathioprine, cyclophosphamide, methotrexate and mitoxantrone.
Is Aubagio an immunosuppressant drug?
Yes, Aubagio (teriflunomide) may lower your white blood cell (WBC) count and possibly suppress your immune system. White blood cells are found in your blood and are used to fight infection.
What drug class is Aubagio?
Aubagio contains the active ingredient teriflunomide. It belongs to the pyrimidine synthesis inhibitor drug class. Tecfidera contains a different active ingredient, dimethyl fumarate. It belongs to the disease-modifying therapy drug class.Are MS drugs immunosuppressants?
Immunosuppressive therapy has been used to treat multiple sclerosis (MS) for over 30 years based on the hypothesis that MS is a T cell-mediated autoimmune disease. The most commonly used immunosuppressive agents in MS are azathioprine, cyclophosphamide, methotrexate, and mitoxantrone.
Does MS reduce life expectancy?
Average life expectancy is slightly reduced for people with MS. It’s most commonly diagnosed in people in their 20s and 30s, although it can develop at any age. It’s about 2 to 3 times more common in women than men. MS is 1 of the most common causes of disability in younger adults.
Is Mavenclad a chemotherapy?
Is Mavenclad a form of chemotherapy? Yes, Mavenclad’s active drug, cladribine, belongs to a group of drugs called antimetabolites. Antimetabolites are a type of chemotherapy that kills certain cells. But Mavenclad isn’t used for cancer treatment.
How safe is aubagio?
Aubagio, with the same RxScore as Gilenya, had the highest number of reports of diarrhea, but that’s the only side effect for which it scored the highest, making it among the safest of the MS therapies.How long can you take aubagio?
If you’re not pregnant and are using effective birth control, you can take AUBAGIO. Always talk to your healthcare provider about all your medications if you are planning to become pregnant. If you stop taking AUBAGIO, it stays in your system for an average of 8 months although it may remain for up to 2 years.
Which is better Tecfidera or aubagio?Tecfidera was more effective at reducing ARRs, with patients showing an adjusted 42% lower relapse rate compared to Aubagio. In agreement with these results, the risk of having a first relapse also was lower in patients treated with Tecfidera.
Article first time published onIs aubagio an immune modulator?
AUBAGIO IS AN IMMUNOMODULATOR. *The exact mechanism by which AUBAGIO exerts its therapeutic effect in MS is not fully understood, but may involve a reduction in the number of activated T and B cells in the CNS.
Is aubagio an anti inflammatory?
Aubagio, also known as teriflunomide, is an immunomodulator with anti-inflammatory properties. It is a once-daily oral medication for the treatment of relapsing-remitting multiple sclerosis (RRMS).
Does aubagio help with fatigue?
Official Answer. Aubagio is unlikely to reduce fatigue associated with Multiple Sclerosis (MS). 85% of patients with MS report fatigue and 76.6% report that fatigue has a significant impact on their lives.
What is the best drug for multiple sclerosis?
For primary-progressive MS , ocrelizumab (Ocrevus) is the only FDA-approved disease-modifying therapy (DMT). Those who receive this treatment are slightly less likely to progress than those who are untreated. For relapsing-remitting MS , several disease-modifying therapies are available.
How do immunosuppressants work for multiple sclerosis?
Immunosuppressants are used for their ability to suppress immune reactions. Agents such as methotrexate have shown some effectiveness in delaying progression of impairment of the upper extremities in patients with secondary progressive MS.
What is selective immunosuppressant?
Selective immunosuppressive agents are drugs that suppress the immune system due to a selective point of action. They are used to reduce the risk of rejection in organ transplants, in autoimmune diseases and can be use as cancer chemotherapy .
Is MAVENCLAD an immunosuppressant?
MAVENCLAD®, also known as cladribine, is an oral selective Immunosuppressant. It is indicated as monotherapy for the treatment of adult patients with relapsing-remitting multiple sclerosis (RRMS) to reduce the frequency of clinical exacerbations and delay the progression of disability.
What is the newest drug for MS?
A new medication for multiple sclerosis (MS) has been approved by the Food and Drug Administration (FDA). The new medication, Ponvory, is a once-daily oral treatment. It’s been shown to reduce annualized relapse rates and new brain lesions in people with MS. This approval is based on a 2-year phase 3 clinical trial.
How long does MAVENCLAD stay in your system?
For most patients, the active ingredient in MAVENCLAD does not remain in the body beyond 1 week. The number of T and B cells reach their lowest level about 2-3 months after the start of each treatment course and were lower with each additional treatment course.
Does MS get worse with age?
Over time, symptoms stop coming and going and begin getting steadily worse. The change may happen shortly after MS symptoms appear, or it may take years or decades. Primary-progressive MS: In this type, symptoms gradually get worse without any obvious relapses or remissions.
Is MS considered a disability?
MS is considered a disability by the Social Security Administration (SSA). Someone with MS can qualify for disability benefits if it is severe enough to prevent them from being able to work full time.
What should I avoid with multiple sclerosis?
It’s recommended that people with MS avoid certain foods, including processed meats, refined carbs, junk foods, trans fats, and sugar-sweetened beverages.
Can aubagio cause heart problems?
Four cardiovascular deaths, including three sudden deaths, and one myocardial infarction in a patient with a history of hyperlipidemia and hypertension were reported among approximately 2600 patients exposed to AUBAGIO in the premarketing database.
Does aubagio affect blood pressure?
Increased Blood Pressure: Blood pressure increases and hypertension have occurred with AUBAGIO. Measure blood pressure at treatment initiation and manage any elevations during treatment. Respiratory Effects: Interstitial lung disease (ILD), including acute interstitial pneumonitis, has been reported with AUBAGIO.
What are the side effects of Kesimpta?
- upper respiratory tract infection,
- headache,
- injection-related reactions (fever, headache, muscle pain, chills, and fatigue), and.
- local injection site reactions (redness, pain, itching, and swelling)
Is Ocrevus better than aubagio?
As for other efficacy and safety outcomes, Ocrevus revealed superior efficacy in reducing the risk of 24-week confirmed disability progression compared to placebo, Rebif, or Aubagio (7 mg), and to enable relapse-free periods compared to 14 other medications.
When is the best time to take Aubagio?
by Drugs.com If you decide to take it at night, then take it every night. If you experience fatigue, tiredness, or weakness with Aubagio, you may find it better to take it at night. The recommended dosage of Aubagio is one tablet (either the 7mg strength or 14mg strength) taken once daily with a glass of water.
Who makes Aubagio?
Aubagio is made by Sanofi Genzyme which is the specialty care business arm of Sanofi.
Does Aubagio cause diarrhea?
The most common side effects when taking AUBAGIO include: headache; diarrhea; nausea; hair thinning or loss; and abnormal liver test results. These are not all the side effects of AUBAGIO. Tell your healthcare provider about any side effect that bothers you.
Is aubagio cytotoxic?
Teriflunomide is considered cytostatic but not cytotoxic: it does not affect resting or slowly dividing lymphocytes. This article reviews the available pharmacological properties of oral teriflunomide and its clinical efficacy and tolerability in patients with relapsing multiple sclerosis.
Which is better Gilenya or Aubagio?
Gilenya (fingolimod) is linked to significantly lower annualized relapse rates in relapsing-remitting multiple sclerosis (RRMS) patients compared to Tecfidera (dimethyl fumarate) or Aubagio (teriflunomide), a study suggests. All three therapies showed similar effects on disability outcomes.