Is ch3nh2 a strong or weak acid or base
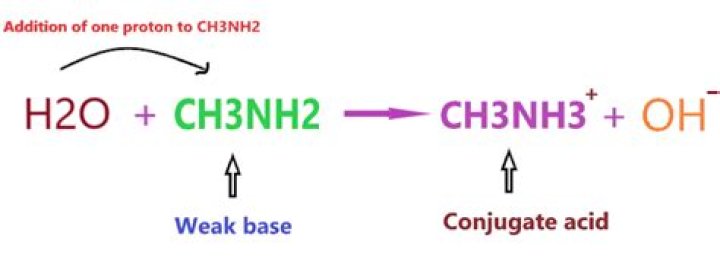
As we discussed earlier, CH3NH2 is a weak base, hence, it will form a conjugate acid by adding one proton to itself. As you see in the above reaction, CH3NH2 is a weak base and we know a weak base always forms a conjugate acid(not necessarily the strong one).
Is CH3NH2 weak or strong acid?
Answer: weak base The formula shown corresponds to methylamine.
Is CH3 2NH a strong or weak base?
Dimethylamine ((CH3)2NH) is a weak base. In aqueous solution at 25°C, it reacts with water to produce its conjugate acid, the dimethylammonium ion ((CH3)2NH2+), and the hydroxide ion (OH-).
Why is CH3NH2 a weak base?
Ammonia has the formula NH3, and has a lone pair of electrons, :NH3. If you see a nitrogen in a compound and it has a lone pair, the compound is a weak base. CH3NH2 is a weak base. By the definition of a Bronsted base, it is a proton acceptor.Is CH3NH3 strong or weak?
CH3NH3+ acts as a acid because Brønsted-Lowry acids give away a proton. Dimethylamine and trimethylamine are co-produced; the reaction kinetics and reactant ratios determine the ratio of the three products. CH3NH2 is not a strong base.
Is CH3NH2 polar or nonpolar?
Methyl amine ie CH3NH2 is a polar molecule. Polarity is the result of significant electronegativity difference between atoms.
What is the role of CH3NH2?
It has a role as a mouse metabolite. It is a primary aliphatic amine, a one-carbon compound and a member of methylamines. It is a conjugate base of a methylammonium.
What is the pH of CH3NH2?
The pH that creates CH3NH2 with a 0.1M molarity is 10^-10.What does CH3NH2 dissociate into?
When CH3NH2 is in water, CH3NH2 will become conjugate acid CH3NH2+ H20 > CH3NH3+ + OH- .
Is CH3NH2 a conjugate base?CH3NH3^ + – CH3NH2 is a conjugate acid base pair.
Article first time published onIs CH3NH2 a weak base in water?
CH3NH2 is considered a weak base because when it is dissolved in an aqueous solution then not all the molecules of it react with water to yield OH– ions, very few molecules of CH3NH2 react with water molecule ions and produce OH– ion in the solution.
Is CH3 3N a strong base?
The compound trimethylamine, (CH3)3N, is a weak base when dissolved in water.
What is the conjugate acid of CH3NH2?
The conjugate acid of ch3nh2 is ch3nh3+.
Why is CH3NH3 a strong acid?
CH3NH3Cl is an ionic compound, consisting of CH3NH3+ and Cl- ions. The overall salt does not donate protons, the CH3NH3+ ion does (to form H3O+) when the salt is dissociated in water. Cl- is a very weak conjugate base so its basicity is negligible. Therefore the salt is acidic because of CH3NH3+, a Bronsted acid.
Is CH3NH3 a strong acid?
In aqueous solution, the methylammonium ion acts as a weak acid. The water molecule acts as bases.
Is Hcch a strong acid?
Hybridization affects electronegativity of an atom. Example: HCCH is more acidic than H2C=CH2, which is more acidic than CH3CH3.
Can CH3NH2 act as an acid?
Methylamine is a Bronsted base, as it can accept a proton from water.
Is CH3NH2 a acid base conjugate acid or conjugate base?
CH3NH3^ + – CH3NH2 is a conjugate acid base pair.
Can CH3NH2 be acidic?
CH3NH2 is a base. It is a base by the same principle as NH3 is a base which is ; A free electron pair on N atom atracts a watter protone to form NH4+ and OH- .
What type of bond is CH3NH2?
Therefore, this is classified as a covalent compound. The compound’s name is methylamine. Each molecule contains 1 carbon atom, 5 hydrogen atoms and 1 nitrogen atom.
Is CH3NH2 ionic or covalent?
(b) CH3NH2 and CH3F are both covalent compounds and have polar bonds.
What shape is CH3NH2?
As a result, the methylamine molecule has a tetrahedral molecular shape considering carbon as a central atom.
How do you make CH3NH2?
Methylamine is prepared commercially by the reaction of ammonia with methanol in the presence of an aluminosilicate catalyst. Dimethylamine and trimethylamine are co-produced; the reaction kinetics and reactant ratios determine the ratio of the three products.
Is CH3NH2 dissolved in water?
The compound methylamine, CH_3NH_2, is a weak base when dissolved in water.
What is aniline pH?
pH lectures Aniline pKb=9.4. Aniline in solution of its hydrochloride is in form of conjugate acid. To calculate pH of such solution we have to find pKa for aniline and treat it like weak acid. … This time [H+]=4.89×10-4 and pH=3.31.
What is the pH of LiOH?
The LiOH concentrations and room temperature pH values are, respectively: 0.06M LiOH, pH 12.8; 0.1M LiOH, pH 13.1; 0.5M LiOH, pH 13.6; 0.85M LiOH, pH 13.8; 1.0M LiOH, pH 14.0 [8]. causing a large increase in the anion defect concentration and hence an increase in oxygen diffusion rate through the oxide film [8].
What is the pH of hydrochloric acid solution?
Hydrochloric acid is an essential component of gastric acid, which has a normal pH of 1.5 to 3.5. A weak acid or base does not ionize completely in aqueous solution. Ionization of a weak acid (HA) is characterized by its dissociation constant (Ka).
Is CH3NH3+ and CH3NH2 a conjugate acid base pair?
CH3NH3^ + – CH3NH2 is a conjugate acid base pair.
Which of the following is a weak acid?
Examples of weak acids include acetic acid (CH3COOH), which is found in vinegar, and oxalic acid (H2C2O4), which is found in some vegetables.
How many valence electrons does CH3NH2 have?
This is the CH3NH2 Lewis structure: Methylamine. For Methylamine we we have 14 valence electrons. For organic compounds, they are often written the way that you should place the atoms on your Lewis structure.
Is CH3NH2 amphoteric?
Ethanol, methylamine, and acetic acid are all amphoteric, reacting as either acids or bases depending on the conditions. ethanol CH3CH2OH methylamine CH3NH2 acetic acid CH3 C OH O (a) Rank ethanol, methylamine, and acetic acid in decreasing order of acidity.