Is co2 a diatomic molecule
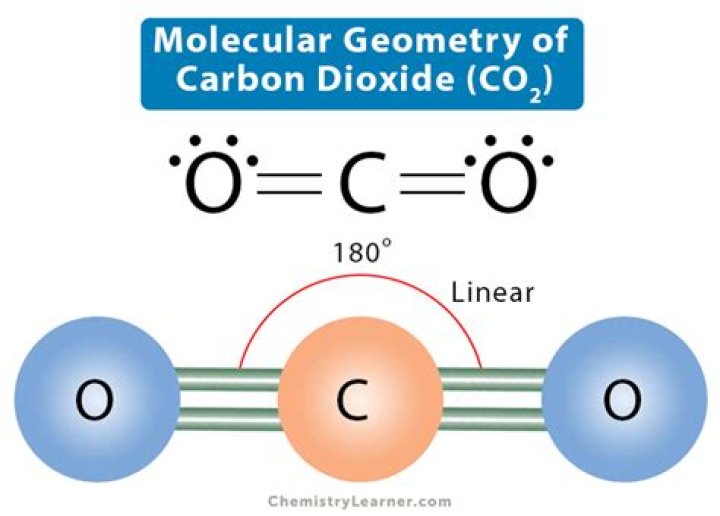
No. CO2 is triatomic. As the name suggests it has three atoms. A molecule is said to be diatomic, If there is only two atoms present in the molecule.
Is co2 a diatomic molecule? - Google Search
For example, carbon monoxide (diatomic), carbon dioxide, ozone (O3), nitrogen monoxide (diatomic), nitrogen dioxide, sulfur dioxide, sulfur trioxide, etc..
Is CO2 a molecular compound?
Carbon Dioxide (CO2) is a gas with a molecular structure composed of two oxygen atoms and one carbon atom. It is an important greenhouse gas because of its ability to absorb infrared wavelengths. Carbon dioxide is a chemical compound that is found in Earth’s atmosphere as a gas.
What are the 8 diatomic molecules?
- Hydrogen.
- Nitrogen.
- Oxygen.
- Fluorine.
- Chlorine.
- Bromine.
- Iodine.
Is CO2 paramagnetic or diamagnetic?
The magnetism of CO2 is diamagnetism and a magnetic force directs CO2 gas towards lower magnetic fields. However, oxygen gas, which is paramagnetic, has a magnetic force that is more than two 160 times greater than that of CO2.
What are examples of diatomic molecules?
Terms. Diatomic molecules are composed of only two atoms, of either the same or different chemical elements. Common diatomic molecules include hydrogen (H2), nitrogen (N2), oxygen (O2), and carbon monoxide (CO).
What is not a diatomic molecule?
The common elements that exist as diatomic molecules at room temperature are hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine. Therefore, the element that does not exist as a diatomic molecule is neon.
Is CO2 ionic or molecule?
No, CO2 is not an ionic compound. As per the definition, an ionic compound is a compound that is mostly formed between a metal atom and a non-metal atom. Meanwhile, CO2 is a compound that is formed between two non-metal atoms (carbon and oxygen) thus giving it a covalent nature.What 7 elements are diatomic?
The 7 diatomic elements are hydrogen (H), nitrogen (N), oxygen (O), fluorine (F), chlorine (Cl), bromine (Br), and iodine (I).
Why CO2 is a molecule?A molecule of carbon dioxide (CO2) is made up of one carbon atom and two oxygen atoms. Carbon dioxide is an important greenhouse gas that helps to trap heat in our atmosphere. Without it, our planet would be inhospitably cold.
Article first time published onWhat makes CO2 a molecule?
The carbon dioxide molecule is made up of one carbon atom joined to two oxygen atoms. This means it has a chemical formula is CO₂.
How is co paramagnetic?
c. O2 is paramagnetic because it has two unpaired electrons, one in each of its p* orbitals. … CO is diamagnetic because all of its electrons are paired.
What is the nature of CO molecule?
Carbon monoxide is a colorless gas and is represented as CO. It has one carbon atom which is covalently bonded to one oxygen atom. It is also known as Carbonic oxide or Carbon oxide. It is a toxic gas.
Would the molecule be diamagnetic or paramagnetic?
On the basis of no of unpaired electrons available in the molecule ,if there are no unpaired electrons it is diamagnetic and if there are one or more unpaired electron(s) it is paramagnetic . This is simplest way one can know about the magnetic behavior of any molecule.
What are diatomic atoms?
Diatomic molecules contain two atoms that are chemically bonded. If the two atoms are identical, as in, for example, the oxygen molecule (O2), they compose a homonuclear diatomic molecule, while if the atoms are different, as in the carbon monoxide molecule (CO), they make up…
Are molecules Triatomic?
Homonuclear triatomic molecules contain three of the same kind of atom. That molecule will be an allotrope of that element. Ozone, O3 is an example of a triatomic molecule with all atoms the same. Triatomic hydrogen, H3, is unstable and breaks up spontaneously.
Which of the following elements is diatomic?
The seven diatomic elements are Hydrogen, Nitrogen, Oxygen, Fluorine, Chlorine, Iodine, Bromine.
What is meant by diatomic molecule?
Diatomic molecules are molecules composed of only two atoms, of the same or different chemical elements. The prefix di- is of Greek origin, meaning “two”. … The bond in a homonuclear diatomic molecule is non-polar.
How do you know if an element is a diatomic molecule?
Diatomic Elements: Diatomic molecules are molecules composed of only two atoms, of the same or different chemical elements. The prefix di- is of Greek origin, meaning “two”. If a diatomic molecule consists of two atoms of the same element, such as hydrogen (H2) or oxygen (O2), then it is said to be homonuclear.
Is a molecule a diatomic?
Astatine (atomic number 85, symbol At) and tennessine (atomic number 117, symbol Ts) are also in the halogen group and may form diatomic molecules. … However, diatomic molecules formed by other elements are not very stable, so their bonds are easily broken.
Why do some elements form diatomic or triatomic molecules?
Answer: Diatomic elements played an important role in the elucidation of the concepts of element, atom, and molecule in the 19th century, because some of the most common elements, such as hydrogen, oxygen, and nitrogen, occur as diatomic molecules.
Which of the following is not a diatomic element?
Carbon does not exist as a diatomic molecule. The seven elements that do are hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and…
Is CO2 covalent or molecular?
Carbon dioxide is made up of one carbon atom, two oxygen atoms. There are four covalent bonds in one molecule of carbon dioxide. Carbon and oxygen are non-metals, thus we know carbon dioxide is a covalent compound.
Is CO2 polar or nonpolar?
Polar molecules have a non-zero net dipole moment. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar.
What kind of compound is CO2?
Carbon dioxide is a one-carbon compound with formula CO2 in which the carbon is attached to each oxygen atom by a double bond.
Is CO2 a covalent bond?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, which is shown here as two lines and referred to as a double bond.
Is carbon a compound or molecule?
Carbon is the only element that can form so many different compounds because each carbon atom can form four chemical bonds to other atoms, and because the carbon atom is just the right, small size to fit in comfortably as parts of very large molecules.
Is CO2 an inert gas?
It is an inert gas that cannot usually be sensed by odor, color or taste although some people claim it has a slightly pungent odor. It is difficult to know when carbon dioxide is present and when it may be depleting oxygen needed for breathing.
How do you make CO2 in chemistry?
Carbon dioxide is produced whenever an acid reacts with a carbonate. This makes carbon dioxide easy to make in the laboratory. Calcium carbonate and hydrochloric acid are usually used because they are cheap and easy to obtain. Carbon dioxide can be collected over water, as shown in the diagram.
Why is carbon dioxide classified as compound not a mixture?
Carbon dioxide (CO2) is a chemical compound. A molecule of it is composed of a lone carbon atom and two oxygen atoms, each connected to the carbon atom by a covalent bond. … The compound formed cannot be seperated into the constituent elements by any physical method, unlike mixtures.
What is the molar mass of CO2?
The molecular mass of carbon dioxide is 44.01amu. The molar mass of any compound is the mass in grams of one mole of that compound. One mole of carbon dioxide molecules has a mass of 44.01g, while one mole of sodium sulfide formula units has a mass of 78.04g.