Is f2 a element or compound
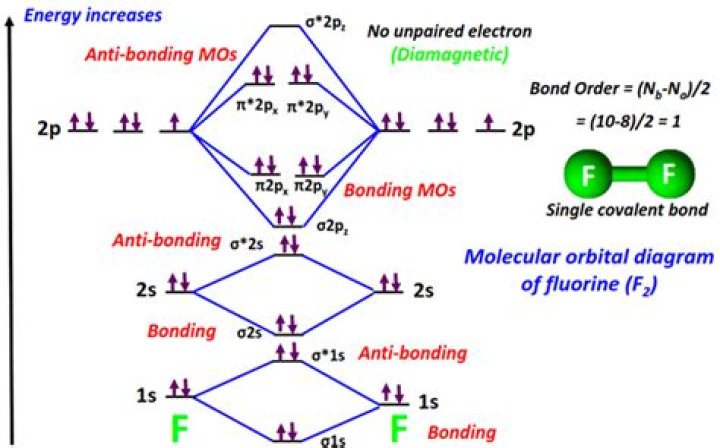
Fluorine is the chemical element in the periodic table that has the symbol F and atomic number 9. Atomic fluorine is univalent and is the most chemically reactive and electronegative of all the elements. In its pure form, it is a poisonous, pale, yellow-green gas, with chemical formula F2.
Is fluorine an element or compound?
fluorine (F), most reactive chemical element and the lightest member of the halogen elements, or Group 17 (Group VIIa) of the periodic table. Its chemical activity can be attributed to its extreme ability to attract electrons (it is the most electronegative element) and to the small size of its atoms.
Is F2 a pure substance or mixture?
Fluorine gas is a diatomic element which formed by only one component. So,its a pure substance not mixture. A pure substance is a form of matter that has a constant composition and properties that are constant throughout the sample.
What is F2 in chemistry compound?
Fluorine | F2 – PubChem.What is F in chemistry periodic table?
Fluorine – Element information, properties and uses | Periodic Table.
Is F2 an ionic compound?
Is fluorine a molecular element?
What is Fluorine on the periodic table?
Fluorine: a small yet very adaptable element The most reactive, electronegative and oxidizing element, fluorine (F) is the first halogen in the periodic table. Fluorine is naturally present in the Earth’s crust where it is chiefly found in mineral form.
Elements and compounds are both examples of pure substances. A substance that cannot be broken down into chemically simpler components is an element. ... For example, water is a compound composed of the elements hydrogen and oxygen. Today, th
How many atoms are there in F2?FluorineStandard atomic weight Ar, std(F)18.998403163(6)Fluorine in the periodic table
Is a compound a pure substance?
One molecule of F2 contains 2 atoms of fluorine.What is a compound or a molecule?
Article first time published onA compound is a molecule made of atoms from different elements. All compounds are molecules, but not all molecules are compounds. ... There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds.
What are the examples of elements?
Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen. At present, 94 are natural elements whereas 24 are synthetic. Thus, a total of 118 elements have been identified so far. Elements having an at
What are the 10 example of elements?
H - Hydrogen.He - Helium.Li - Lithium.Be - Beryllium.B - Boron.C - Carbon.N - Nitrogen.O - Oxygen.
What is called element?
An element is a substance that cannot be broken down into any other substance. There are about 100 elements, each with its own type of atom. Everything in the universe contains the atoms of at least one or more elements. The periodic table li
How do you write fluorine?
Fluorine is a chemical element with symbol F and atomic number 9.
How many isotopes does f have?
Fluorine (9F) has 18 known isotopes ranging from 13F to 31F (with the exception of 30F) and two isomers (18mF and 26mF). Only fluorine-19 is stable and naturally occurring; therefore,
What is the electronegativity of F?
The concept of electronegativity was introduced by Linus Pauling in 1932; on the Pauling scale, fluorine is assigned an electronegativity of 3.98, and the other elements are scaled relative to that value.
What number is chromium?
Group61907°C, 3465°F, 2180 KAtomic number2451.996State at 20°CSolid52CrElectron configuration[Ar]
What is fluorine a metal nonmetal or metalloid?
Its atomic number is 9 and its atomic weight is 19, and it's a gas at room temperature. It is the most electronegative element, given that it is the top element in the Halogen Group, and therefore is very reactive. It is a nonmetal, and is on
How do you make fluorine?
Fluorine is produced commercially by electrolyzing anhydrous hydrogen fluoride containing dissolved potassium fluoride to achieve adequate conductivity (Jaccaud and Faron 1988; Shia 1994). Potassium fluoride and hydrogen fluoride form potassi
Is F and F ionic or covalent?
Explanation: In order to find how ionic a bond is, you need to look at the differences in their electronegativity. ... F-F is the most covalent because the electronegativity values are the same so the difference would be zero.
What is the ionic charge of F2?
We have removed two electrons, so the molecule becomes the ion F2+2 with a charge of +2.
What type of solid is formed by F2?
Small symmetrical molecules (nonpolar molecules), such as H2, N2, O2, and F2, have weak attractive forces and form molecular solids with very low melting points (below −200 °C).
Is babr2 a molecular compound?
Barium bromide is an ionic compound.
What’s a molecular element?
Molecular elements are the chemical species that has at least two atoms of the same chemical element bonded to each other via chemical bonding. These are different from chemical compounds because a chemical compound contains two or more atoms
What are the 9 molecular elements?
hydrogen.oxygen.nitrogen.fluorine.chlorine.bromine.iodine.
Is F2 a complete octet?
Complete step by step solution: Thus it has an incomplete octet. ... Whereas, ${F_2}$ completes the octet rule because ${F_2}$ contains a single pair of electrons. Each Fluorine atom also has three pairs of electrons that are not shar
What is the polarity of F2?
H2 and F2 is non-polar as they are made of two same atoms which means both atoms should have the same electronegativity. Thus, an equal number of electrons exist in the orbital overlap, and thus H2 and F2 is non-polar.
Is a compound a mixture?
Compounds are pure substances. ... Each molecule of a compound is made from two or more different kinds of atoms that are chemically bonded. Mixtures are made of two or more substances — elements or compounds — that are mixed physically but n
What is the pure element?
An element is a pure substance that cannot be broken down into different types of substances. Examples of elements include carbon, oxygen, hydrogen, gold, silver and iron. ... Each element is made up of just one type of atom. An atom is the s
Which of the following is not an element?
Answer: (3)silica Graphite is a carbon element. Silicon(Si) and germanium(Ge) are also elements, but silica (SiO2) is not an element.
How many molecules does F2 have?