Is fractional distillation an open or closed system
This is an open system; the vacuum adapter is open to the atmosphere. You should never heat a closed system, as it may explode or fly apart. Distillation is complete when either the upper limit of the expected boiling range is reached or there is little distilland remaining in the flask.
What type of process is fractional distillation?
Fractional distillation is the process of separating crude oil into groups of hydrocarbons with similar numbers of carbon atoms. We call these groups of hydrocarbons “fractions”.
What are the differences between fractional distillation and simple distillation?
Fractional distillation is the process for separating the mixture into its parts. Simple distillation is the process in which two liquids with a different boiling point can be separated.
Why can't distillation occur in a closed system?
If you heat an organic compound in a closed-system distillation apparatus that is not properly vented, it can build pressure and potentially burst. The resulting explosion can cause dangerous flying glass, hot, corrosive chemicals to spatter and potentially hazardous fumes to be released.Is fractional distillation the same as distillation?
Summary: 1. Simple distillation is the method used to separate substances in mixtures with significantly different boiling points, while fractional distillation is used for mixtures containing chemicals with boiling points close to each other.
What is fractional distillation?
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to fractionate.
What is fractional distillation of air?
Fractional distillation is the technique that is used for obtaining different components from the air. Fractional distillation is a separation method where the difference in boiling points of components is used to separate the liquid mixture into fractions through distillation.
Why is an ice bath used in distillation?
Since the vapor pressure of water at 20 °C is roughly 2.3 kPa, then 2.3/101.325 or 0.023 would be the fraction of water that would not condense and would pass out of the condenser. This is why the distillate is frequently chilled in an ice bath during the distillation.Should you close a distillation system explain why?
If any vapor escapes at the connection points, it may come into direct contact with the heat source and ignite. Never heat a closed system, the increasing pressure will cause the glass to explode.
What is the role of the condenser in the distillation?In chemistry, a condenser is laboratory apparatus used to condense vapors — that is, turn them into liquids — by cooling them down. … In distillation, a mixture is heated until the more volatile components boil off, the vapors are condensed, and collected in a separate container.
Article first time published onHow does distillation differ from fractional distillation even though both involve evaporation and condensation?
Explaining the Difference The difference between simple and fractional distillation is the number of times that the liquid is vaporized and condensed. Simple distillation condenses the liquid once, so the boiling points of the two liquids must be far apart to make it efficient.
How distillation differs from fractional distillation even though both are evaporation following by condensation?
Simple distillation will only have one distillation (vaporization-condensation) cycle in the whole process, while the fractional cycle will have at least two cycles. 3. Fractional distillation will use additional equipment called the ‘fractionating column’, while the simple distillation will not need that equipment.
How does fractional distillation work in terms of evaporation and condensation?
Fractional distillation works because the different liquids have different boiling points. When the mixture is heated: … vapours condense when they reach a part of the column that is below the temperature of their boiling point. the liquid flows out of the column.
How is fractional distillation used to separate gases?
How Separation of Gases by Fractional Distillation Works. Each gas has a characteristic boiling point, defined as the temperature at which it transforms from a liquid into a gas. If you have a random sample of gases, you can separate them by gradually cooling the sample until each component gas liquifies.
Can distillation be used to separate air?
Air can be separated into its components by means of distillation in special units. So-called air fractionating plants employ a thermal process known as cryogenic rectification to separate the individual components from one another in order to produce high-purity nitrogen, oxygen and argon in liquid and gaseous form.
Which gas is separated first in fractional distillation of air?
Liquid nitrogen has a boiling point of −190 degree Celsius. It turns into a gas first and separates from air.
Is fractional distillation a physical process?
Fractional distillation is a strictly physical process. The idea is that part of the mixed solution boils off leaving the higher boiling point component behind.
Is fractional distillation a chemical or physical method?
Fractional distillation is the process of taking a chemical mixture and using heat to separate out the various components in that mixture.
Which of the following is not separated through distillation process?
Which of the following is not separated through distillation process? Explanation: Milk and water are not separated through the distillation process. All the other options can be separated through a distillation process. 7.
What is the difference between simple distillation and fractional distillation Brainly?
The main difference between fractional distillation and simple distillation is that simple distillation separate liquids with boiling point gaps of at least 50 degrees whereas fractional distillation separates liquids with closer boiling points.
What happens if you heat a closed system?
A closed system is any assembly which is completely sealed and not open to the air. … Heating the system creates pressure inside it. If too much pressure builds up, it has to release somehow.
Why is a thermometer used in fractional distillation?
The thermometer allows us to check how close to boiling we are and make sure the distillation isn’t going too quickly. Why? If you heat a mixture of two or more different liquids with varied boiling points, obviously one liquid will boil first.
Where should the thermometer bulb be during a fractional distillation?
The bulb part of the thermometer is positioned near the side arm of the Y-adaptor so that it monitors the temperature of the vapors. See the illustrations below. If it is too low, it will be too close to the boiling liquid and will read higher than the true vapor temperature.
Why should you not distill to dryness?
Do not distill to complete dryness. The temperature of a heated, dry flask can rise quickly, and distillations can leave explosive residues. Explosion of a distilling flask heated to dryness can cause serious injury or death!
What is the Lyne arm?
The lyne arm is a copper tube that connects the head of a pot still to the condenser. The lyne arm is a copper tube that connects the head of a pot still to the condenser. As it leaves the head of the still a lyne arm either angles upwards, is horizontal, or angles downwards.
What is reflux in fractional distillation?
Reflux is a technique involving the condensation of vapors and the return of this condensate to the system from which it originated. It is used in industrial and laboratory distillations. It is also used in chemistry to supply energy to reactions over a long period of time.
What is difference between evaporation and condensation?
Condensation is the change from a vapor to a condensed state (solid or liquid). Evaporation is the change of a liquid to a gas.
What is the difference between condensation evaporation and distillation?
EvaporationDistillationEvaporation is a gradual and slow processDistillation is rapid and quickIt occurs at the surfaceIt doesn’t occur at the surface
Where does condensation take place in distillation?
Vapor that passes the thermometer condenses in the condenser, a double-walled tube that is cooled by water flowing through the outer layer, and drips into the receiver.
How does fractional distillation work in terms of evaporation?
Fractional distillation works because the different liquids have different boiling points. When the mixture is heated: vapours rise through a column which is hot at the bottom, and cooler at the top. vapours condense when they reach a part of the column that is below the temperature of their boiling point.
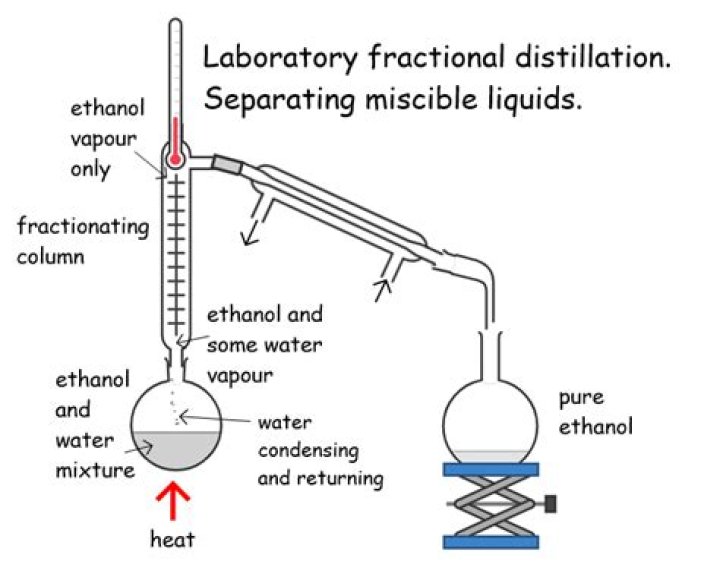
How does fractional distillation separate ethanol and water?
For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points. When the mixture is heated, one liquid evaporates before the other.



