Is Hydrosulfuric acid a strong acid
Hydrosulfuric acid is classified as a weak acid. Its chemical symbol is H2S H 2 S .
Is Hydrosulfuric acid an acid or base?
KaAcidBase4.4 * 10-7Carbonic acidHydrogen carbonate ion1.1 * 10-7Hydrosulfuric acidHydrogen sulfide ion6.3 * 10-8Dihydrogen phosphate ionHydrogen phosphate ion6.2 * 10-8Hydrogen sulfite ionSulfite ion
Is perchloric acid a weak acid?
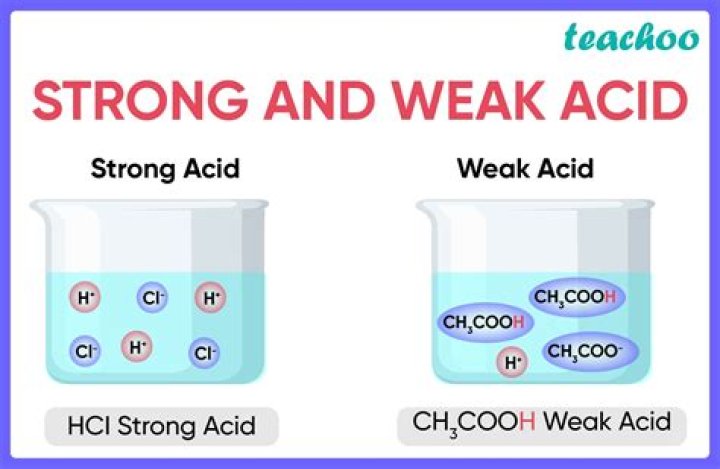
All the other acids are weak. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).
Is Sulphuric acid strong or weak?
AcidConjugate BaseStrong AcidsHCl (hydrochloric acid) (strongest)Cl− (chloride ion) (weakest)H2SO4 (sulfuric acid)HSO4− (hydrogen sulfate ion)HNO3 (nitric acid)NO3− (nitrate ion)Is nitrous acid strong or weak?
Nitrous acid (HNO2), a weak acid, is very unstable and exists only in aqueous solution.
Is h3so4 an acid?
AcidH2CO3 (carbonic acid)HCO−3 (hydrogen carbonate ion)HCN (hydrocyanic acid) (weakest)CN− (cyanide ion) (strongest)
Is chloric acid a strong acid?
Chloric acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is a strong acid (pKa ≈ −2.7) and oxidizing agent.
Why H2SO4 is called strong acid?
Sulphuric acid is called a strong acid because it dissociates completely into its component ions in water. This also makes it a good electrolyte.Why is Sulphuric acid a weak acid?
AcidConjugate BaseH2SO4 (sulfuric acid)HSO4− (hydrogen sulfate ion)HNO3 (nitric acid)NO3− (nitrate ion)Weak Acids
Is hydrofluoric acid a strong acid?Acidity. ) and protons, thus greatly increasing the acidity. This leads to protonation of very strong acids like hydrochloric, sulfuric, or nitric when using concentrated hydrofluoric acid solutions. Although hydrofluoric acid is regarded as a weak acid, it is very corrosive, even attacking glass when hydrated.
Article first time published onIs perchloric acid strong or weak?
5 Perchloric acid. Perchloric acid (HClO4) is one of the strongest mineral acids. Hot and concentrated perchloric acid has powerful oxidizing and dehydrating properties, and it will react explosively with organic compounds.
Is perchloric acid a binary acid?
Simple acids, known as binary acids, have only one anion and one hydrogen. … Therefore, HClO4 is called perchloric acid. With one fewer oxygen than the “-ate” ion, the acid will have the suffix “-ous.” For example, chlorous acid is HClO2.
Why is perchloric acid strong?
The Hydrogen atom in HClO4 will experience less attraction to Oxygen than in HIO4 (the electron is further). That’s why HClO4 is stronger than HIO4.
Why nitrous acid is a weak acid?
Every acid reacts with water. When it loses a proton, it transfers that proton to water. … Let’s look at the reaction of a strong acid, nitric acid or HNO3, and a weaker acid, nitrous acid or HNO2, in water. Nitric acid is a stronger acid than nitrous acid because its conjugate base is more stable.
Is nitrous acid a strong or weak electrolyte?
Nitrous acid, HNO2(aq), is a weak electrolyte.
What's the strongest acid in the world?
The world’s strongest superacid is fluoroantimonic acid, HSbF6. It is formed by mixing hydrogen fluoride (HF) and antimony pentafluoride (SbF5). Various mixtures produce the superacid, but mixing equal ratios of the two acids produces the strongest superacid known to man.
Is Cl A strong base?
A strong acid like HCl donates its proton so readily that there is essentially no tendency for the conjugate base Cl– to reaccept a proton. Consequently, Cl– is a very weak base.
Is chloric acid the same as hydrochloric acid?
As adjectives the difference between hydrochloric and chloric. is that hydrochloric is (chemistry) of or relating to hydrochloric acid while chloric is (chemistry) of a compound, containing chlorine with a higher oxidation number than equivalent chlorous compounds; especially having an oxidation number of 5.
What type of compound is chloric acid?
Chloric acid is a chlorine oxoacid. It is a conjugate acid of a chlorate.
What are the 7 strong acids?
- Chloric acid: HClO3.
- Hydrobromic acid: HBr.
- Hydrochloric acid: HCl.
- Hydroiodic acid: HI.
- Nitric acid: HNO3.
- Perchloric acid: HClO4.
- Sulfuric acid: H2SO4.
Is LiOH a strong base?
LiOH is a weak base because it is more covalent due to small size. So, they cannot yield hydroxyl ions easily in aqueous solutions which is the main cause of basicity.
What is the formula for Hydrosulfuric acid?
Hydrosulfuric AcidNameHydrosulfuric AcidAlso Known asHydrogen Sulfide and SulfaneAppearanceColourless GasChemical FormulaH2S
Why concentrated H2SO4 is weaker than dilute Sulphuric acid?
The presence of water in dilute sulphuric acid increases the hydrogen ion concentration. Hence it is a stronger acid than concentrated sulphuric acid which contains comparatively less water.
Which acid is the weakest?
Hydrofluoric acid is the only weak acid produced by a reaction between hydrogen and halogen (HF). Acetic acid (CH3COOH), which is contained in vinegar, and oxalic acid (H2C2O4), which is present in some vegetables, are examples of weak acids.
Is sulphurous acid a weak acid?
Although sulfuric acid is a strong acid, sulfurous acid is weak.
Why H2SO4 is stronger acid than HCl?
However, HCl is stronger than H2SO4. This is mainly due to the difference in the basicity of both acids. Additionally, if we look at the pKa value HCl has a pKa of -6.3 and sulfuric acid has a pKa ~-3. What this means is that HCl is more acidic as it donates a proton easily.
How strong is hydrofluoric acid?
Hydrofluoric acid is extremely corrosive! Aqueous solutions dilute as 0.1 M will pH at approximately 1.0. Unlike other mineral acids, hydrofluoric acid will attack glass, concrete, rubber, quartz and alloys containing silica.
Why is hydrofluoric acid considered a weak acid?
Why Hydrofluoric Acid Is a Weak Acid Because the hydroxonium ion is attached to the fluoride ion, it isn’t free to function as an acid, thus limiting the strength of HF in water. The FHF- bifluoride anion is stabilized by a strong hydrogen bond between hydrogen and fluorine.
Is HBrO3 a strong acid?
The pKa of HBrO3 is -2 and of HBr04 is -4.6. This means that both are strong acids, completely ionized in water.
How strong is perchloric acid?
Perchloric acid is a strong acid used for complete digestions of organic material. It is normally supplied in bottles of up to one gallon in capacity at 70-72% strength. In many respects, its hazards are similar to those of nitric acid, as both are strong oxidants.
Is sroh2 a strong base?
Group IA and Group IIA (Only Sr, Ca, Ba) with hydroxide (OH−) are strong bases.