Is hypo osmotic the same as hypotonic
Hyperosmotic solutions are not always hypertonic. But hyposmotic solutions are always hypotonic. … If the solution has a lower concentration of nonpenetrating solutes than the cell does, then there will be net movement of water into the cell at equilibrium and the solution is hypotonic.
Is hypertonic and Hyperosmotic the same thing?
As adjectives the difference between hypertonic and hyperosmotic. is that hypertonic is (of a solution) having a greater osmotic pressure than another while hyperosmotic is hypertonic.
What is hypo osmotic solution?
1. Of, relating to, or characterized by having a lower osmotic pressure than a surrounding fluid under comparison. 2. A condition in which the total amount of solutes (both permeable and impermeable) in a solution is lower than that of another solution.
Is deionized water hypotonic or hypertonic?
Distilled water represents a hypotonic solution, yet the cells do not burst because of the cell wall.What osmolarity is hypotonic?
Osmolality is a measurement of a fluids concentration which is represented by the number of solutes per volume of fluid. The osmolality of the blood and body fluids is approximately 290 mOsmol/L. Hypotonic solutions have an osmolality lower than the body (< 280 mOsmol/l) which actively promotes fluid absorption.
What's the difference between tonicity and osmolarity?
The key difference between tonicity and osmolarity is that the tonicity measures only the concentration of non-penetrating solutes through a semipermeable membrane while the osmolarity measures the total concentration of penetrating and non-penetrating solutes.
What is the difference between Hyperosmotic and Hypoosmotic?
The key difference between isosmotic hyperosmotic and hypoosmotic is that isosmotic refers to the property of having equal osmotic pressures, but hyperosmotic refers to the property of having a high osmotic pressure. Meanwhile, hypoosmotic refers to the property of having a low osmotic pressure.
Is Coke hypertonic or hypotonic?
Regular cola is a hypertonic solution (493 mOsm/L) due to its high carbohydrate content, while both tap water (3 mOsm/L) and light cola (38 mOsm/L) are hypotonic solutions compared with plasma (285-295 mOsm/L).Is vinegar hypotonic?
So after soaking in vinegar you should have also noticed that the egg increases a little in size. This is because the water in the vinegar can enter the egg through the membrane, moving from the higher water concentration in vinegar to the lower concentration in the egg. … Water is known as hypotonic, ie.
What is the difference between osmosis and diffusion?In diffusion, particles move from an area of higher concentration to one of lower concentration until equilibrium is reached. In osmosis, a semipermeable membrane is present, so only the solvent molecules are free to move to equalize concentration.
Article first time published onWhat is hypotonic condition?
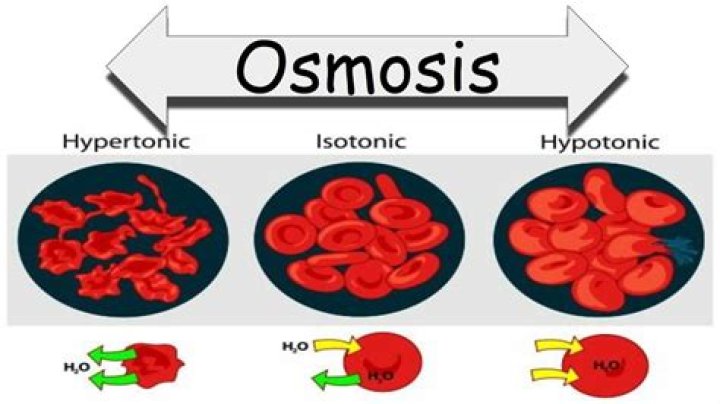
A hypotonic solution has a lower concentration of solutes than another solution. In biology, a solution outside of a cell is called hypotonic if it has a lower concentration of solutes relative to the cytosol. Due to osmotic pressure, water diffuses into the cell, and the cell often appears turgid, or bloated.
What is hypertonic and hypotonic solution?
A hypertonic solution contains a higher concentration of solutes compared to another solution. The opposite solution with a lower concentration is known as the hypotonic solution. … If a cell is placed in a hypertonic solution, the cell is considered hypotonic.
Can a solution be Hyperosmotic and hypotonic?
When a cell is placed in a hyperosmotic but hypotonic solution like 10% dextran, water movement will occur. Therefore, a solution can be hyperosmotic and hypotonic.
What are the examples of hypotonic solution?
Hypotonic Solution Examples Hypotonic saline i.e., 0.45% sodium chloride or 0.25% sodium chloride with or without dextrose, 2.5% dextrose solution, etc are some of the examples of the hypotonic solutions that are hypotonic with respect to blood serum and are used as hypotonic intravenous solutions.
What is the difference between hypertonic hypotonic and isotonic solutions?
The main difference between isotonic hypotonic and hypertonic solutions is that isotonic solutions are solutions having equal osmotic pressures while hypotonic solutions are solutions having a lower osmotic pressure and hypertonic solutions are solutions with a high osmotic pressure.
What is an example of a hypertonic solution?
Hypertonic solutions have a higher concentration of electrolytes than plasma. … Common examples of hypertonic solutions are D5 in 0.9% normal saline and D5 in lactated ringers. The administration of hypertonic solutions should be monitored extremely closely, as they can quickly lead to fluid overload.
What is the difference between Isosmotic and isotonic?
Isotonic refers to a solution having the same solute concentration as in a cell or a body fluid. Isosmotic refers to the situation of two solutions having the same osmotic pressure. Isosmotic solutions cause cells to absorb water from surrounding or to lose water from cells.
How do you find hypotonic?
If the solute concentration outside the cell is lower than inside the cell, and the solutes cannot cross the membrane, then that solution is hypotonic to the cell. If a cell is placed in an isotonic solution, there will be no net flow of water into or out of the cell, and the cell’s volume will remain stable.
Is a marine fish Hyperosmotic or Hypoosmotic?
Saltwater fish are hypoosmotic to the sea, their blood has a lower solute content and, therefore, a lower osmotic pressure (about 400 mOsmol) than sea water (about 1000 mOsmol).
What is the difference between tonicity and osmotic pressure?
Osmotic pressure is the pressure of a solution against a semipermeable membrane to prevent water from flowing inward across the membrane. Tonicity is the measure of this pressure. … Usually, there is a higher concentration of solutes on one side of the membrane than the other.
What fluids have the same tonicity as plasma?
Isotonic IV Fluids Most IV fluids are isotonic, meaning, they have the same concentration of solutes as blood plasma. When infused, isotonic solutions expand both the intracellular fluid and extracellular fluid spaces, equally.
What is tonicity of a solution?
Tonicity is defined as the ability of a solution surrounding a cell to cause that cell to gain or lose water (Urry et al., 2017). While osmolarity is an absolute quantity, tonicity is relative.
Why does an egg swell in water?
This process is called osmosis. In osmosis, the water molecules move from the area with more water molecules to the area with fewer water molecules. … When you put the egg into pure water, some of the water molecules from the solution move into the egg and the egg swells up.
Is an egg hypertonic or hypotonic?
Compared to distilled water, the solution within the egg membrane is hypertonic. Therefore when the decalcified egg is placed in water, the water flows through the membrane into the egg. A concentrated sugar solution is hypertonic compared to the solution inside the egg.
What happens when we put egg in vinegar?
If you soak an egg in vinegar the eggshell will absorb the acid and break down, or dissolve. The calcium carbonate will become carbon dioxide gas, which will go into the air. … Soak one egg in vinegar for 24 hours (1 day), one egg for 48 hours (2 days) and one egg for 72 hours (3 days).
Is coffee hypotonic?
Coffee cup analogies (remember coffee is mostly water): OSMOLARITY is a number referring to the number of moles of solute in a solution. —-For a cup of coffee with no solutes, osmolarity = 0. … And therefore, your coffee would be hypotonic to your friend’s, or your friend’s would be hypertonic to yours.
Is Red Bull hypertonic?
Hypertonic Drinks have a high osmolality than body fluids are absorbed slowly and are best taken as recovery drink after a workout. Hypertonic drinks have a carbohydrate concentration of over 10%. Energy drinks including Redbull and soft drinks like coke or lemonade come into this category as well as Lucozade energy.
Why is alcohol hypertonic?
Drinking alcohol can cause dehydration, which makes the blood hypertonic.
What are three differences between osmosis and diffusion?
Diffusion Versus OsmosisDiffusionOsmosis
What is the main difference between osmosis and diffusion quizlet?
The main difference between the two is osmosis is the diffusion of water and has to do with water and diffusion is the process of movement. Diffusion can be air and osmosis is water.
What are the 3 types of osmosis?
The three types of osmotic conditions include- hypertonic, isotonic, and hypotonic.