Is pH above 14 possible
It describes how many hydrogen ions (protons) are present in a solution: the higher the pH, the lower the hydrogen ion concentration, and vice versa. But the scale does not have fixed limits, so it is indeed possible to have a pH above 14 or below zero.
What is the highest level of pH?
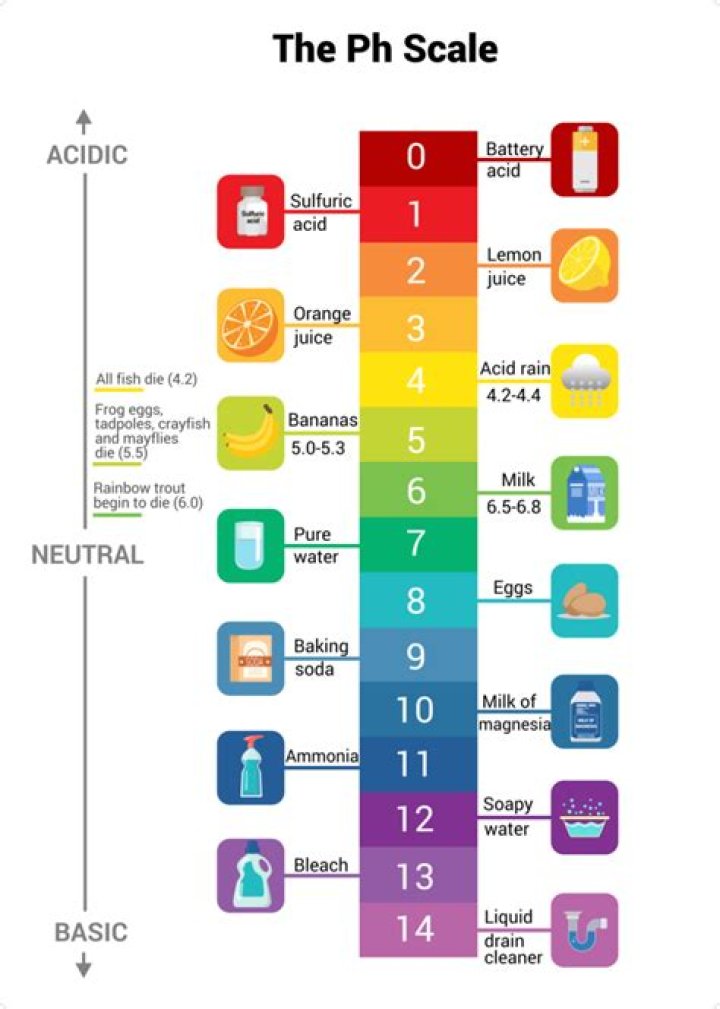
The pH scale ranges from 0 to 14, with a pH of 7 being neutral. Buffers are solutions that moderate pH changes when an acid or base is added to the buffer system.
Why pH range is not more than 14?
One far end is not more than 1M of hydrogen ions, which results in a pH value of not more than 0. While on the other end is not more than 1M of hydroxide ions which results in a pH value of not more than 14. … The pH value goes out of the 0-14 range when the concentration of the solution exceeds 1M.
Can you get pH 16?
To obtain a solution of pH=16, one has to dissolve 100- Equivalents of NaoH in 1- litre of water. But the SOLUBILITY of NaoH= 1100- g/ LITRE at 25°C. Even at 100°C, it’s solubility is 3700- g/ LITRE only. So it is not possible to obtain a solution with pH= 16- practically.What has a pH of 13?
pH ValueH+ Concentration Relative to Pure WaterExample110.000 1ammonia solution120.000 01soapy water130.000 001bleach, oven cleaner140.000 000 1liquid drain cleaner
What foods have a pH above 7?
Examples of acid-forming foods (pH0 to 7)are meats, poultry, eggs, dairy products, sugary drinks, snacks, wheat products, coffee, mayonnaise, milk, alcohol and ketchup; examples of neutral foods (ph7) are natural fats, sugars and starches and examples of alkaline foods (pH 7 to 14) are vegetables, legumes, fruits and …
What chemical has the highest pH?
Sodium hydroxide has a pH that sits around 14, the top of the pH scale.
What is the lowest possible pH?
Since pH is defined as minus the logarithm of a number whose value is between 0 and 1, theoretically, the lowest value of pH is zero (0), which means a hydrogen ion activity of 1, which means basically that hydrogen ions (i.e. protons) are all that is present.Is a pH of 15 possible?
But the scale does not have fixed limits, so it is indeed possible to have a pH above 14 or below zero. For example, concentrated hydrochloric acid can have a pH of around -1, while sodium hydroxide solution can have a pH as high as 15.
Which is the strongest acid?Strong AcidsStrong Baseshydrobromic acid (HBr)potassium hydroxide (KOH)hydroiodic acid (Hl)calcium hydroxide (Ca(OH)2)nitric acid (HNO3)strontium hydroxide (Sr(OH)2)sulfuric acid (H2SO4)barium hydroxide (Ba(OH)2)
Article first time published onIs a pH of 0 possible?
The pH value can be less than 0 for very strong acids, or greater than 14 for very strong bases. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.
What is the most powerful base?
The title of strongest base in the world belongs to ortho-diethynylbenzene dianion. This superbase has the strongest proton affinity ever calculated (1843 kJ mol−1), beating out a long-standing contender known as lithium monoxide anion.
What is the pH of blood?
The acidity and alkalinity of your blood are measured using the pH scale. The pH scale ranges from 0 (very acidic) to 14 (very alkaline). Blood is usually between 7.35 to 7.45.
What is the pH of a Coke?
How acidic is Coke? Its pH is reported to be 2.6 to 2.7, mainly due to H3PO4, phosphoric acid. As a fizzy drink, it contains plenty of dissolved carbon dioxide, but this makes very little contribution to the acidity.
What pH is toxic?
The danger of more acidic water – when the pH is less than 6.5 – is that it can leach metals from the well and from the pipes that bring you water. These metals include lead, manganese, copper and iron, and they can be toxic in large amounts. So acidic water obviously poses a health risk.
What base has a pH of 14?
Increasing pH (Decreasing Acidity)Substances14 (most basic)Sodium hydroxide (NaOH)
What does a pH of 6.8 mean?
The more acidic a solution the greater the hydrogen ion concentration and the lower the pH; a pH of 7.0 indicates neutrality, a pH of less than 7 indicates acidity, and a pH of more than 7 indicates alkalinity. … A pH above 7.8 or below 6.8 is generally fatal.
Is soap an alkaline?
Soap, of necessity, and by definition is alkaline. … Without getting crazy technical and losing your interest, sodium hydroxide breaks up the fatty acids or triglycerides of our oil – neutralises the acid, and leaves us a hard bar that is alkaline enough to remove dirt and oils from our skin.
Is honey an alkaline?
The pH scale ranges from 0 to 14, where anything with a pH of less than 7 is considered acidic. A pH of 7 is neutral. A pH greater than 7 is considered akaline. … Scientists have recorded a pH level of between 3.3 to 6.5 for different types of honey, so honey is therefore acidic.
What is the pH of a Apple?
ItemApproximate pHApplesApples Delicious3.90Apples Golden Delicious3.60Apples Jonathan3.33
What pH is a lemon?
Lemon juice contains citric acid and has a pH of around 3.
Why pH scale is up to 14 only?
However, in reality, most solutions you would find in a standard laboratory have a pH value between 0-14. This is because, in order to reach pH levels below 0 or above 14, one would require extraordinarily acidic or basic solutions, respectively.
Who is the Queen of acid?
Nitric Acid (HNO3) is known as Queen of acids.
Who is the king of chemicals?
Sulfuric acid is sometimes referred to as the “king of chemicals” because it is produced worldwide in…
Is ha a weak acid?
What is a Weak Acid? A weak acid is any acid that reacts with water (donates H+ ions) to a very small extent, usually less than 5 – 10%. … The equation representing the ionization of any weak acid, HA, and the equilibrium expression, Ka, are shown below.
Is there a ph0?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. … As this diagram shows, pH ranges from 0 to 14, with 7 being neutral. pHs less than 7 are acidic while pHs greater than 7 are alkaline (basic).
Can you have a molarity over 1?
If the density of the solution is 1.072 g mL(raise to power -1) then what shall be the molarity of the solution? Yes. As molality = no of moles of solute/ weight of solvet in kg. If no of mole is greater than 1 in 1kg of solvent then molality is grater than 1.
Do Superbases exist?
A superbase is a compound that has a particularly high affinity for protons. Superbases are of theoretical interest and potentially valuable in organic synthesis. Superbases have been described and used since the 1850s.
Do bases burn you?
Bases with a pH greater than 10 can cause chemical burns. Strong bases include, calcium hydroxide, sodium hydroxide and potassium hydroxide. … Bases also react with oils on skin and fatty tissue, which can lead to extensive damage to skin and subcutaneous tissue.
What is the weakest acid in the world?
Hydrofluoric acid is the only weak acid produced by a reaction between hydrogen and halogen (HF). Acetic acid (CH3COOH), which is contained in vinegar, and oxalic acid (H2C2O4), which is present in some vegetables, are examples of weak acids.
What is the pH of human saliva?
Saliva has a pH normal range of 6.2-7.6 with 6.7 being the average pH. Resting pH of mouth does not fall below 6.3. In the oral cavity, the pH is maintained near neutrality (6.7-7.3) by saliva.