Is sodium perchlorate a strong acid
Perchloric acid is a mineral acid with the formula HClO4. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid.
Is sodium perchlorate a strong acid? - Google Search
Perchloric acid is a mineral acid with the formula HClO4. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid.
Is sodium chlorate strong or weak?
Corrosionpedia Explains Sodium Chlorate Sodium chlorate is an inorganic compound. It is a highly powerful oxidizing agent due to its high oxidizing potential.
Is perchlorate a base or acid?
Perchlorate is a monovalent inorganic anion obtained by deprotonation of perchloric acid. It is a monovalent inorganic anion and a chlorine oxoanion. It is a conjugate base of a perchloric acid.Is NaClO an acid or base?
NaClO is weakly basic because ClO− is the conjugate base of a weak acid, HClO (hypochlorous acid).
Is MgSO4 acidic or basic?
3 Answers. Looking at the following references, it seems that most agree that a water solution of MgSO4 is slightly acidic. “The pH of an aqueous magnesium sulfate solution is related to the molarity of the MgSO4. Typically, the pH is between 5.5 and 6.5 due to magnesium’s affinity for hydroxide ion (OH-).
What makes a weak acid?
A weak acid is an acid that partially dissociates into its ions in an aqueous solution or water. In contrast, a strong acid fully dissociates into its ions in water. … At the same concentration, weak acids have a higher pH value than strong acids.
Is perchlorate ionic or covalent?
Silver perchlorate is an ionic compound formed between a silver ion and the polyatomic ion perchlorate. A polyatomic ion is a molecule with an electrical charge. Perchlorate has three oxygen atoms double bonded with a chlorine atom, and one oxygen atom singly bonded with the same chlorine atom.What is the pH of sodium perchlorate?
The pH is often adjusted to 2.5 or 3.0.
Is acetic acid a strong acid?A strong acid is an acid which is completely ionized in an aqueous solution. Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water. A weak acid is an acid that ionizes only slightly in an aqueous solution. Acetic acid (found in vinegar) is a very common weak acid.
Article first time published onIs hydrobromic acid strong or weak?
All the other acids are weak. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).
Why has sodium chlorate been banned?
In the past, the main use of sodium chlorate was as a pesticide, largely to kill weeds and undesirable foliage. Sodium chlorate is harmful to both humans and the environment. Therefore, the sale and use of sodium chlorate in plant protection products and pesticides in the EU is banned.
Is perchloric acid a strong acid?
Perchloric acid is a corrosive inorganic liquid that is colorless, odorless, and oily in nature. … A cold 70% aqueous perchloric acid solution is considered to be a strong acid or superacid (stronger than sulfuric and nitric acids) but is not necessarily a strong oxidizing agent.
Is sodium sulfite acidic or basic?
Sodium sulphite, Na2SO3, is a salt made from the neutralization reaction between a strong base (sodium hydroxide) and a weak acid (sulphurous acid). Its aqueous solution is, therefore, distinctly basic in nature, with a pH value slightly greater than 7.
What is NaClO used for?
Sodium hypochlorite, commonly known as bleach, is most frequently used as a disinfecting agent. It is a broad-spectrum disinfectant that is effective for the disinfection of viruses, bacteria, fungi, and mycobacterium.
Is Hydrochloric acid is a strong acid?
HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule.
Which is the strongest acid?
For instance, hydrochloric acid comes in at about pH 1.6, nitric acid at 1.08 and pure sulfuric acid at a whopping pH -12. That makes sulfuric acid the strongest ‘normal’ acid you’ll find. Anything more powerful is defined as a superacid.
Is Methanoic acid a strong acid?
acidKa (mol dm-3)hydrofluoric acid5.6 x 10-4methanoic acid1.6 x 10-4ethanoic acid1.7 x 10-5hydrogen sulphide8.9 x 10-8
Is sodium hydroxide a strong acid?
NaOH is the strong base because it completely dissociates in an aqueous solution and produces a higher amount of OH– ions.
What makes a stronger acid?
The bond strength of an acid generally depends on the size of the ‘A’ atom: the smaller the ‘A’ atom, the stronger the H-A bond. When going down a row in the Periodic Table (see figure below), the atoms get larger so the strength of the bonds get weaker, which means the acids get stronger.
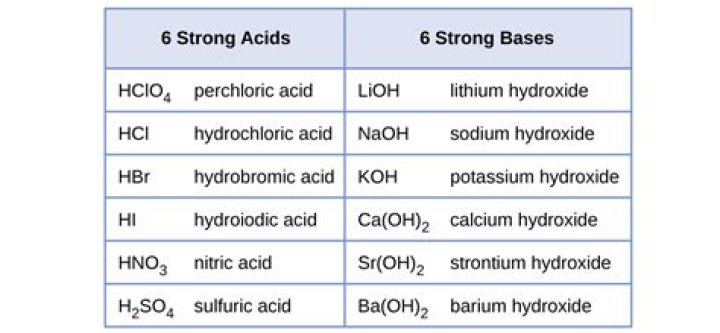
What are the 7 strong acids?
- Chloric acid: HClO3.
- Hydrobromic acid: HBr.
- Hydrochloric acid: HCl.
- Hydroiodic acid: HI.
- Nitric acid: HNO3.
- Perchloric acid: HClO4.
- Sulfuric acid: H2SO4.
Is MgSO4 a Lewis acid?
* MgSo4 is a neutral ionic salt. * It does not have/ is not an acid or a base. * Acid : when dissolved in water dissociates to form H+ ions.
What is the acidic radical of MgSO4?
Acidic radicalBasic radicale. Mg(OH)2OH-Mg2+
Is MgSO4 a strong electrolyte?
The compound magnesium sulfate is a strong electrolyte.
What is the pH of perchloric acid?
The pH of the perchloric acid solution is 1.55. The pH of an acid can be calculated by taking the negative log of the hydrogen ion concentration in…
Is sodium a peroxide?
Sodium peroxide is the inorganic compound with the formula Na2O2. This yellowish solid is the product of sodium ignited in excess oxygen. It is a strong base.
Is CH3NH3NO3 an acidic salt?
CH3NH2 is a weak base (Kb = 5.0 * 10-4) and so the salt, CH3NH3NO3, acts as a weak acid.
Is sodium perchlorate ionic?
An inorganic sodium salt comprising equal numbers of sodium and perchlorate ions. Sodium perchlorate is the inorganic compound with the chemical formula NaClO4. It is a white crystalline, hygroscopic solid that is highly soluble in water and in alcohol. It is usually encountered as the monohydrate.
Is sodium hydroxide ionic or covalent?
In other words, NaOH is an ionic molecule. It dissolves in water because of the charge-dipole interactions between the ions and the water. Figure 2 shows a sodium cation and a hydroxide anion each surrounded by a cluster of water molecules.
Is sodium acetate a strong acid?
Sodium acetate is a salt of weak acid & strong Base. So, it is not categorized as acid or base. But it gives basic solution on hydrolysis.
Is sodium hydroxide a strong base?
Because sodium hydroxide is a strong base that dissociates completely in solution to form hydroxide ions, if the concentration of hydroxide ions in solution is . 10 molar, so is the initial concentration of sodium hydroxide.