What are allosteric effectors
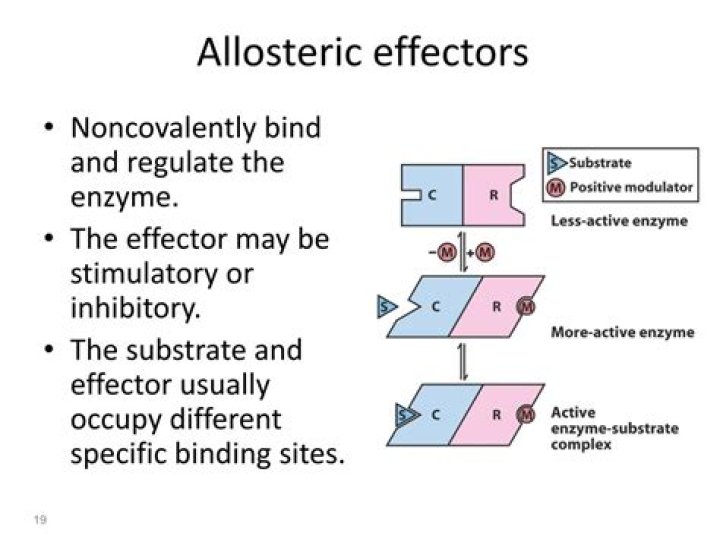
An allosteric effector is a molecule that binds to the site of an allosteric enzyme, causing a change in configuration resulting in an increase (positive effector) or reduction (negative effector) in enzyme activity. It is usually an intermediary in a metabolic pathway.
What is an example of an allosteric effector?
A good example of a homotropic allosteric effector is oxygen (O2) – it acts as an effector of haemoglobin in the human body. A heterotropic allosteric effector is a regulatory molecule which is not also the substrate for the enzyme. It can either activate or inhibit the enzyme it binds to.
What are 2 examples of allosteric effectors of hemoglobin?
Allosteric effectors such as inositol hexaphosphate (IHP) bind to both deoxy-Hb and HbCO, albeit at different sites, leading to a lowered oxygen affinity. The manner in which these effectors impact oxygen binding is unclear and may involve changes in structure, dynamics or both.
What are the two types of allosteric effectors?
Allosteric sites allow effectors to bind to the protein, often resulting in a conformational change involving protein dynamics. Effectors that enhance the protein’s activity are referred to as allosteric activators, whereas those that decrease the protein’s activity are called allosteric inhibitors.What are allosteric enzymes and its examples?
Prominent examples of allosteric enzymes in metabolic pathways are glycogen phosphorylase (41), phosphofructokinase (9, 80), glutamine synthetase (88), and aspartate transcarbamoylase (ATCase) (103).
What is Homotropic and Heterotropic?
When the ligands interacting are all the same compounds, the effect of the allosteric interaction is considered homotropic. When the ligands interacting are different, the effect of the allosteric interaction is considered heterotropic.
Why is Allostery important?
Allosteric regulations are a natural example of control loops, such as feedback from downstream products or feedforward from upstream substrates. Long-range allostery is especially important in cell signaling. Allosteric regulation is also particularly important in the cell’s ability to adjust enzyme activity.
What are Homotropic and Heterotropic allosteric modulators?
It is typically an activator of the enzyme. For example, O2 is a homotropic allosteric modulator of hemoglobin. … It may be either an activator or an inhibitor of the enzyme. For example, H+, CO2, and 2,3-bisphosphoglycerate are heterotropic allosteric modulators of hemoglobin.Is Oxygen an allosteric effector of hemoglobin?
Haemoglobin is an allosteric protein. This means that the binding of oxygen to one of the subunits is affected by its interactions with the other subunits. … For example in blood capillaries (partial pressure of oxygen is approx 20 mmHg) haemoglobin will release its oxygen to myoglobin for storage there.
What are Apoenzymes and Holoenzymes?An apoenzyme is an inactive enzyme, activation of the enzyme occurs upon binding of an organic or inorganic cofactor. Holoenzyme- An apoenzyme together with its cofactor. A holoenzyme is complete and catalytically active. Most cofactors are not covalently bound but instead are tightly bound.
Article first time published onIs O2 binding to myoglobin allosteric?
A) O2 binding to myoglobin is allosteric but not cooperative.
What is role of hemoglobin and myoglobin?
Hemoglobin is essential for transferring oxygen in your blood from the lungs to the tissues. Myoglobin, in muscle cells, accepts, stores, transports and releases oxygen.
What role do allosteric effectors play in hemoglobin's Cooperativity?
Allosteric effectors such as inositol hexaphosphate (IHP) bind to both deoxy‐Hb and HbCO, albeit at different sites, leading to a lowered oxygen affinity. The manner in which these effectors impact oxygen binding is unclear and may involve changes in structure, dynamics or both.
Which enzymes are termed as allosteric enzymes?
- Aspartate Transcarbamoylase (ATCase) ATCase catalyses the biosynthesis of pyrimidine. …
- Glucokinase. It plays an important role in glucose homeostasis. …
- Acetyl-CoA Carboxylase. Acetyl-CoA carboxylase regulates the process of lipogenesis.
What is the meaning of allosteric protein?
A protein whose shape is changed when it binds a particular molecule. In the new shape the protein’s ability to react to a second molecule is altered.
What are allosteric enzymes Class 11?
An allosteric enzyme is an enzyme that binds to the allosteric modulator that changes the conformational structure and when binds to the ligands then it results in the change in the structure of the ligand. … The point at which the effector molecule binds is called the allosteric site.
What medications are allosteric inhibitors?
The vast majority of the currently available allosteric drugs are noncovalent. Examples include valium and the benzodiazepines, which target the ionotropic GABA receptor, positive allosteric modulators of mGluRs (Wood et al., 2011), and positive and negative modulators of GPCRs (Conn et al., 2009).
What is the difference between active site and allosteric site?
Active site binds substrate and catalyzes the reaction resulting in the production of a particular product. Allosteric site is a specific part of an enzyme formed by several amino acids that provide the modulation of enzymatic activity.
What is an allosteric drug?
In pharmacology and biochemistry, allosteric modulators are a group of substances that bind to a receptor to change that receptor’s response to stimulus. Some of them, like benzodiazepines, are drugs.
Is co an allosteric effector?
Carbon monoxide (CO) poisoning causes between 5,000−6,000 deaths per year in the US alone. The development of small molecule allosteric effectors of CO binding to hemoglobin (Hb) represents an important step toward making effective therapies for CO poisoning.
What is the difference between Cooperativity and Allostery?
The term cooperativity is used to describe folding of macromolecules and the formation of molecular structures and macromolecular ensembles while allostery is often referenced to illustrate ligand-induced conformational transitions that impact the function of a biological molecule.
What are allosteric activators?
Allosteric activators bind to locations on an enzyme away from the active site, inducing a conformational change that increases the affinity of the enzyme’s active site(s) for its substrate(s). Allosteric inhibitors modify the active site of the enzyme so that substrate binding is reduced or prevented.
Is carbon dioxide an allosteric effector of hemoglobin?
Carbon dioxide and hydrogen ions are two allosteric effectors of hemoglobin. They bind to different sites on the hemoglobin molecule, stabilize the T-state of hemoglobin and lower its affinity for oxygen. … Carbon dioxide on the other hand binds onto the terminal amino residue to form a carbamate ion.
How do allosteric effectors affect hemoglobin binding to oxygen?
These are allosteric effectors of Hb binding of O 2. Some of these molecules are also transported by Hb. … O 2 – positively affects binding of O 2 (positive cooperativity). This results from the breakage of salt bridges between Hb subunits that help stabilize the “tense” deoxy forms of the subunits.
What type of allosteric effector is oxygen?
-binding capacity of hemoglobin are called effectors (allosteric regulation). Effectors may be positive or negative; homotropic or heterotropic effectors. Oxygen is a homotropic positive effector. -binding curve to the left, negative effectors shift the curve to the right.
Which is a property of allosteric enzyme?
Allosteric enzymes have active and inactive shapes differing in 3D structure. Allosteric enzymes often have multiple inhibitor or activator binding sites involved in switching between active and inactive shapes. Allosteric enzymes have characteristic “S”-shaped curve for reaction rate vs. substrate concentration.
Is phosphorylation allosteric regulation?
The kinase that catalyses the phosphorylation of pyruvate decarboxylase is subject to allosteric regulation by a number of small molecules, including acetyl CoA, pyruvate and ADP, as indicated in Figure 34 .
What is the difference between positive and negative Allosterism?
A positive allosterism indicates that the binding of an effector molecule to an enzyme causes the enzyme to change its configuration into an active form. In contrast, a negative allosteirsm indicates that an effector molecule binding causes the enzyme to change its configuration from an active form to an inactive form.
What is apoenzyme BYJU's?
Apoenzyme or apoprotein is an enzymatically inactive protein part of an enzyme, which requires a cofactor for its activity. Apart from catalytic RNA, most of the enzymes are proteins. Not all the enzymes require a cofactor.
Are cofactors organic?
Organic cofactors are small organic molecules (typically a molecular mass less than 1000 Da) that can be either loosely or tightly bound to the enzyme and directly participate in the reaction. In the latter case, when it is difficult to remove without denaturing the enzyme, it can be called a prosthetic group.
What are the two main types of cofactors?
Cofactors can be divided into two types: inorganic ions and complex organic molecules called coenzymes. Coenzymes are mostly derived from vitamins and other organic essential nutrients in small amounts.