What do electrons in the same shell have in common
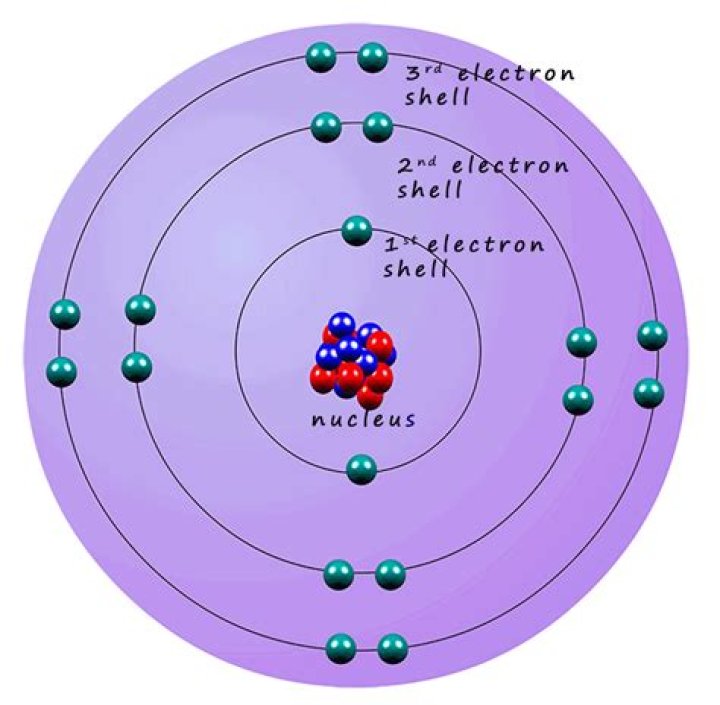
What do electrons in the same shell have in common? They all have the same amount of energy.
What do electrons in the same electron shell have in common?
Each electron shell consists of one or more subshells, and each subshell consists of one or more orbitals. It is often stated that electrons in the same electron shell have the same energy levels. This is at best a generalisation. All electrons in the same subshell will have exactly the same energy level.
What do all the number of electrons have in common?
All atoms have the same number of electrons as protons, so the positive and negative charges “cancel out”, making atoms electrically neutral. Unlike protons and neutrons, which are located inside the nucleus at the center of the atom, electrons are found outside the nucleus.
What do all atoms of the same electron have in common?
Ernest Z. The common feature is that the atoms of all elements consist of electrons, protons, and neutrons.What do atoms in the same column have in common?
Explanation: For the elements in column 1,2 and 13-18 the atoms in the same column have the same amount of outermost electrons, called valence electrons. … An atom can have at a maximum 8 valence electrons (this becomes a bit more complicated after the third row).
Are electron shells and orbitals the same thing?
The main difference between shell subshell and orbital is that shells are composed of electrons that share the same principal quantum number and subshells are composed of electrons that share the same angular momentum quantum number whereas orbitals are composed of electrons that are in the same energy level but have …
How does n relate to the number of electrons?
How does n relate to the number or orbitals per main energy level (shell)? The number of orbitals in shell (n) is equal to n^2. … The number of allowed electrons in shell (n) is equal to 2n^2.
Are electrons of different elements the same?
No. Two atoms of the same chemical element are typically not identical. First of all, there is a range of possible states that the electrons of an atom can occupy. Two atoms of the same element can be different if their electrons are in different states.What do all elements of the same element have in common?
All atoms of the same element have the same number of protons, but some may have different numbers of neutrons. … Atoms of the same element that differ in their numbers of neutrons are called isotopes. Many isotopes occur naturally. Usually one or two isotopes of an element are the most stable and common.
What do all atoms of the same element have in common quizlet?Atoms of the same element all have the same number of protons but they can have different numbers of neutrons. … The atomic number is the number of protons in the nucleus of the atoms in that element. The atomic mass is the average mass of the different isotopes of that element.
Article first time published onHow many electrons are in a shell?
Energy shellMaximum number of electronsFirst2Second8Third8
Does the third shell have 8 or 18 electrons?
In this sense the third shell can hold 8 electrons. 4s2 not the third shell, but the next 10 electrons go into the 3d orbitals that are part of the third shell but shown on the fourth shell level. … So the third shell can be considered to hold 8 or 18 electrons but in total the third shell can hold 18 electrons.
Are all protons the same?
All protons are identical. For example, hydrogen protons are exactly the same as protons of helium and all other elements, or pure substances. However, atoms of different elements have different numbers of protons. … The number of protons in an atom determines the electrical charge of the nucleus.
What do elements in the same group have in common examples?
All the elements in one group have the same number of valence electrons. … Since elements in a group have the same number of valence electrons, they behave similarly in chemistry. An example would be the alkali metals (excepting hydrogen. Hydrogen is in this group only because it has one valence electron.
What are the electrons in the outermost shell called?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.
What do elements in the same column in the periodic table have in common quizlet?
The elements in the same column of the periodic table are all part of the same chemical family or group. Each member of the chemical family has the same number of valence electrons.
How many electrons are in the N 5 shell?
Energy Level (Principal Quantum Number)Shell LetterElectron Capacity3M184N325O506P72
How many electrons can n 2 have?
(a) When n = 2, there are four orbitals (a single 2s orbital, and three orbitals labeled 2p). These four orbitals can contain eight electrons. Again, each orbital holds two electrons, so 50 electrons can fit in this shell.
What is the relationship between the value of N and the total number of orbitals in a main energy level?
For n = 2, there are 22 or four orbitals. For n = 3 there are nine orbitals, for n = 4 there are 16 orbitals, for n = 5 there are 52 = 25 orbitals, and so on. To calculate the maximum number of electrons in each energy level, the formula 2n2 can be used, where n is the principal energy level (first quantum number).
What are electron orbital shells?
An electron shell is the outside part of an atom around the atomic nucleus. It is a group of atomic orbitals with the same value of the principal quantum number n. Electron shells have one or more electron subshells, or sublevels.
Do all of the electrons in a given shell have the same energy?
All orbitals within the same shell have the same energy. … The two conclusions were that a maximum of 2 electrons can occupy a single orbital. And the 2 electrons in the same orbital must have opposite spins.
Which element has two electron shells?
Atomic NumberElementEnergy Levels or “shells”2Helium (He)23Lithium (Li)24Beryllium (Be)25Boron (B)2
What is similar about the electrons on the outside of each element?
Each column is called a group. The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. They are the electrons involved in chemical bonds with other elements.
Are the number of protons and electrons the same?
An atom contains equal numbers of protons and electrons . Since protons and electrons have equal and opposite charges , this means that atoms are neutral overall.
What do all compounds have in common?
Two things are true of all compounds: A compound always has the same elements in the same proportions. For example, carbon dioxide always has two atoms of oxygen for each atom of carbon, and water always has two atoms of hydrogen for each atom of oxygen. A compound always has the same composition throughout.
Why are electrons the same?
Every electron in the universe has exactly the same mass, exactly the same charge, and if you think about it, there’s no reason why they’d have to be. … According to him, the reason that every electron is the same is because every electron is the same electron.
How do electrons in the same atom differ?
The central structure of an atom is the nucleus, which contains protons and neutrons. This nucleus is surrounded by electrons. Although these electrons all have the same charge and the same mass, each electron in an atom has a different amount of energy. … Electrons that have higher energy are found further away.
How are atoms of the same element similar and different?
ISOTOPES AND ATOMIC MASSES For many of the chemical elements there are several known isotopes. Isotopes are atoms with different atomic masses which have the same atomic number. The atoms of different isotopes are atoms of the same chemical element; they differ in the number of neutrons in the nucleus.
What do all atoms of the same element have?
All atoms of an element have the same number of protons, and every element has a different number of protons in its atoms. For example, all helium atoms have two protons, and no other elements have atoms with two protons.
Why do all atoms of an element have the same atomic number although they may have different mass numbers quizlet?
Atoms of each element have the same number of protons, but they can have different numbers of neutrons. Why do isotopes of the same element have different atomic masses? Isotopes of an element vary in mass because their numbers of neutrons differ.
Do all atoms of a given element have the same?
– Atoms of a given element are identical (meaning they have the same mass and properties). – Atoms retain their identity in all chemical reactions.



