What do you mean by Hydrohalogenation of alkenes
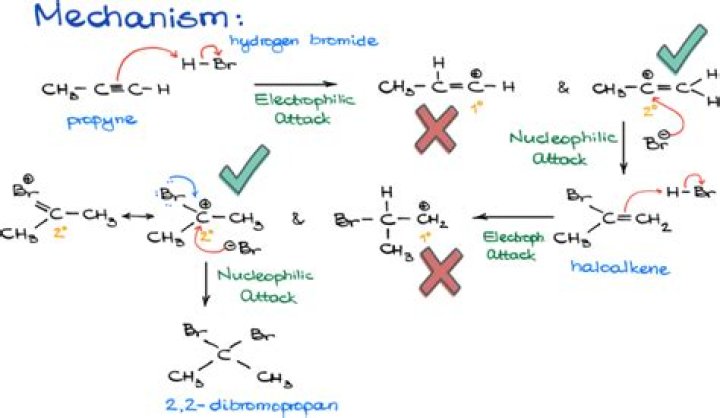
Reaction Overview: The hydrohalogenation of alkenes involves breaking a carbon to carbon double bond, followed by the electrophilic addition of a hydrogen atom and halogen. The halide will add to the more substituted carbon following Markovnikov’s rule. The product is a haloalkane also called an alkyl halide.
What is hydrohalogenation of an alkene?
Dehydrohalogenation is an elimination reaction that eliminates (removes) a hydrogen halide from a substrate. The reaction is usually associated with the synthesis of alkenes, but it has wider applications.
How many products are in hydrohalogenation?
In the alkene hydrohalogenation reaction, two different products are possible, depending on which alkene carbon the halogen atom is added to. These two products are regioisomers, which are structural isomers that differ based on the position of a substituent or functional group (in this case, the halogen atom).
What do you mean by hydrohalogenation?
From Wikipedia, the free encyclopedia. A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes.What is the importance of hydrohalogenation?
Radical halogenation (or hydrohalogenation of olefins) allows the introduction of an halide group at a predictable position on the carbon chain. This C−X can be hydrolyzed to give alcohols, the which can be further oxidized to give ketones and aldehydes and acids.
What is Zaitsev product?
In organic chemistry, Zaitsev’s rule (or Saytzeff’s rule, Saytzev’s rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. … While effective at predicting the favored product for many elimination reactions, Zaitsev’s rule is subject to many exceptions.
What type of reaction is Hydrohalogenation?
Hydrohalogenation, an electrophilic alkene addition reaction, is highly useful as a precursor reaction in multi-step organic chemistry synthesis. Understanding the Molecules: H-X molecules such as H-I, H-Br and H-Cl are highly polar molecules.
What is the order of reactivity of alkenes towards Hydrohalogenation electrophilic?
When treated with HX alkenes form alkyl halides. Hydrogen halide reactivity order : HI > HBr > HCl > HF (paralleling acidity order).Is Hydrohalogenation a symmetrical addition reaction?
Hydrohalogenation of Symmetrical Alkenes In this reaction, the hydrogen atom of HBr adds to one of the carbon atoms and the bromine atom of HBr adds to the other carbon atom. Bromoethane is the only product possible for this reaction.
What do you mean by addition reaction?addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. … An addition reaction may be visualized as a process by which the double or triple bonds are fully or partially broken in order to accommodate additional atoms or groups of atoms in the molecule.
Article first time published onWhat is the major product of the following Hydrohalogenation reaction?
The major product is generated from the more stable carbocation, while the minor product forms from the less stable one. Thus, the major product is 2‐bromopropane. Hydrogen bromide can also be added to an alkene in an anti‐Markovnikov fashion.
What is the name of HX?
The Full form of HX is Medical History, or HX stands for Medical History, or the full name of given abbreviation is Medical History.
What is HX in chemistry?
From Wikipedia, the free encyclopedia. Hydrogen halides (hydrohalic acids when in the aqueous phase) are diatomic, inorganic compounds that function as Arrhenius acids. The formula is HX where X is one of the halogens: fluorine, chlorine, bromine, iodine, or astatine.
What is the general formula for alkenes?
Alkenes are defined as either branched or unbranched hydrocarbons that possess at least one carbon–carbon double bond (CC) and have a general formula of CnH2n [1].
Why do alkenes undergo addition reaction?
Alkenes are unsaturated molecules, which means they do not have all the hydrogen they could have. This is because there is at least one double bond between carbons. This is a stable structure, but not the most stable, so when certain compounds or elements are added, like fluorine, they undergo an addition reaction.
What is the main reaction of alkenes?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
Is Hydrohalogenation syn addition?
2. The Hydrohalogenation Reaction Provides A Mixture of Syn and Anti Products. Stereochemistry: as we saw in the stereochemistry post, this reaction provides a mixture of “syn” and “anti” products (when the reactant makes this possible).
What type of stereochemistry is involved in hydrogenation?
Ernest Z. The stereochemistry arises because both the hydrogen and the alkene must be adsorbed on the surface of a solid catalyst. The catalytic hydrogenation of an alkene results in a syn addition.
Why is Markovnikov's rule followed?
Markovnikov’s rule is an empirical rule used to predict regioselectivity of electrophilic addition reactions of alkenes and alkynes. … To afford the observed product, the net reaction is addition of the hydrogen atom in HBr to the doubly bonded carbon atom in the alkene, bearing the greater number of hydrogen atoms.
What is Zaitsev and Hoffman?
The Hofmann Elimination is an elimination reaction of alkylammonium salts that forms C-C double bonds [pi bonds]. … In contrast with most elimination reactions that yield alkenes, which follow the Zaitsev (Saytzeff) rule, the Hofmann elimination tends to provide the less substituted alkene.
What does Zaitsev rule say?
Today, we refer to this as Zaitsev’s rule, which states that the more highly substituted alkene is the more likely product of an elimination reaction. Therefore, tetra-substituted alkenes are more stable compared to tri-substituted, compared to di-substituted, compared to mono-substituted alkenes.
What is Markovnikov rule explain with example?
Let us explain Markovnikov’s rule with the help of a simple example. When a protic acid HC (X = Cl, Br, I) is added to an asymmetrically substituted alkene, addition of acidic hydrogen takes place at the less substituted carbon atom of the double bond, while X is added to the more alkyl substituted carbon atom.
Why is Bromopropane a minor product?
Carbon-1 is bonded to 2 hydrogen, while carbon-2 is bonded to 1 hydrogen only. … Conversely when hydrogen is added to carbon-2, which has less hydrogen, and bromine is added to carbon-1, the product 1-bromopropane will be the minor product.
What is Markovnikov rule in chemistry?
Markovnikov rule, in organic chemistry, a generalization, formulated by Vladimir Vasilyevich Markovnikov in 1869, stating that in addition reactions to unsymmetrical alkenes, the electron-rich component of the reagent adds to the carbon atom with fewer hydrogen atoms bonded to it, while the electron-deficient component …
What is correct order of electrophilic addition of following alkenes?
I > II > III > IV.
Which order is correct about electrophilic addition on these alkenes?
I > II > III.
What is addition and substitution reaction?
An addition reaction occurs when two or more reactants combine to form a single product. This product will contain all the atoms that were present in the reactants. … A substitution reaction occurs when an exchange of elements in the reactants takes place.
What is addition reaction Class 10?
An addition reaction occurs when two or more reactants combine to form a single product. Addition reactions occur with unsaturated compounds. The general equation for an addition reaction: A+BA+B →→ C.
What is meant by nucleophilic addition reaction?
In organic chemistry, a nucleophilic addition reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile, such that the double or triple bond is broken.
What is addition reaction Class 11?
An Additional reaction is defined as the reaction where two compounds combine to form a new compound without losing any of its atoms. In this reaction weaker pi-bond is converted to form two new sigma bonds.
Why is 2 Bromopropane the major product?
Here we see that in principle, propene can protonate to give two different carbocations, one 2o and the other 1o. Formation of the more stable 2o carbocation is preferred. The carbocation then reacts with the nucleophile to give the alkyl bromide and hence 2-bromopropane is the major product.