What does Benedicts solution detect
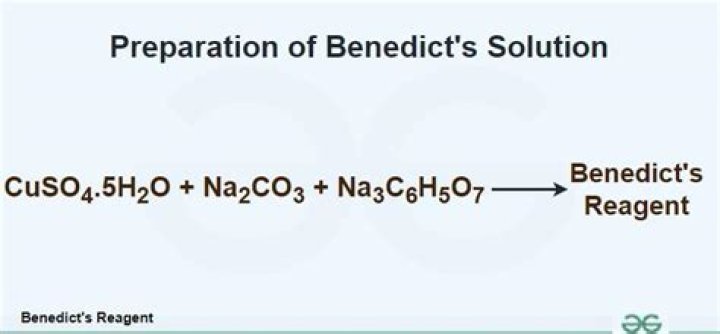
Benedict’s Test is used to test for simple carbohydrates. The Benedict’s test identifies reducing sugars (monosaccharide’s and some disaccharides), which have free ketone or aldehyde functional groups. Benedict’s solution can be used to test for the presence of glucose in urine.
What does the Benedicts solution test for?
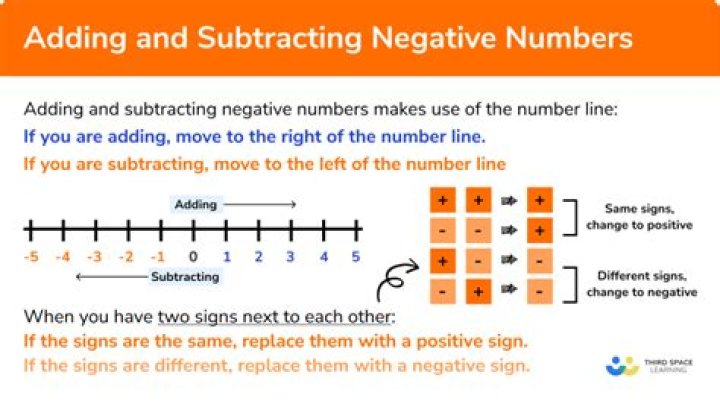
We can use a special reagent called Benedict’s solution to test for simple carbohydrates like glucose. Benedict’s solution is blue but, if simple carbohydrates are present, it will change colour – green/yellow if the amount is low and red if it is high.
How does Benedict's test detect reducing sugars?
In lab, we used Benedict’s reagent to test for one particular reducing sugar: glucose. Benedict’s reagent starts out aqua-blue. As it is heated in the presence of reducing sugars, it turns yellow to orange. The “hotter” the final color of the reagent, the higher the concentration of reducing sugar.
How does Benedict's solution react with starch?
Benedict’s solution turns orange when it reacts with individual glucose molecules. It does not react with the bonded glucose molecules in sucrose or starch. In the experiment, you added amylase to the starch in test tube 4. … This reaction is indicated by the color change in the Benedict’s solution.What solution is used in testing for proteins?
Proteins are detected using Biuret reagent . This turns a mauve or purple colour when mixed with protein.
Why does the Benedict's test not detect starch?
Starches do not react or react very poorly with Benedict’s reagent due to the relatively small number of reducing sugar moieties which occur only at the ends of carbohydrate chains. Other carbohydrates which produce a negative result include inositol.
What does the iodine test detect?
Iodine Test Using iodine to test for the presence of starch is a common experiment. A solution of iodine (I2) and potassium iodide (KI) in water has a light orange-brown color. If it is added to a sample that contains starch, such as the bread pictured above, the color changes to a deep blue.
What are the tests for detecting carbohydrates that uses the oxidation reduction principle?
Most commonly used tests for detection of reducing sugars are Fehling’s Test, Benedict’s Test and Barfoed’s Test. a) Fehling’s Test: Fehling’s Solution (deep blue colored) is used to determine the presence of reducing sugars and aldehydes. Perform this test with fructose, glucose, maltose and sucrose.What test is used to detect the presence of starch?
An iodine test can be used for the detection of starch in a given sample. The iodine test can help to distinguish starch from monosaccharides, disaccharides, and other polysaccharides. The iodine test is used for distinguishing between starch, glycogen, and carbohydrates.
How do you identify reducing sugars?A reducing sugar is one that reduces another compound and is itself oxidized; that is, the carbonyl carbon of the sugar is oxidized to a carboxyl group. A sugar is classified as a reducing sugar only if it has an open-chain form with an aldehyde group or a free hemiacetal group.
Article first time published onWhat is reducing sugar in biology?
Reducing Sugar (biology definition): A sugar that serves as a reducing agent due to its free aldehyde or ketone functional groups in its molecular structure. … Examples are glucose, fructose, glyceraldehydes, lactose, arabinose and maltose, except for sucrose.
How does biuret reagent detect the presence of protein?
The biuret reaction can be used to assess the concentration of proteins because peptide bonds occur with the same frequency per amino acid in the peptide. … The test is named so because it also gives a positive reaction to the peptide-like bonds in the biuret molecule.
How does biuret solution work?
The biuret test measures peptide bonds in a sample. … In an alkaline solution, copper II is able to form a complex with the peptide bonds. Once this complex has been formed, the solution turns from a blue color to a purple color. The deeper the purple color, the more peptide-copper complexes that have been formed.
What does biuret reagent detect?
Biuret reagent is an alkaline solution of 1% CuSO4, copper sulfate. The violet color is a positive test for the presence of protein, and the intensity of the color is proportional to the number of peptide bonds in the solution.
What would happen if you did an experiment where the iodine solution was placed in the dialysis bag and the starch solution was in the beaker?
If we did an experiment in which the iodine solution was placed in the baggie and the starch solution was in the breaker the iodine would move out the baggie. Once the iodine makes it way out the baggie the starch would change color.
Why does iodine only test for starch?
Chemical Test for Starch or Iodine Iodine – KI Reagent: Iodine is not very soluble in water, therefore the iodine reagent is made by dissolving iodine in water in the presence of potassium iodide. … Starch amylopectin does not give the color, nor does cellulose, nor do disaccharides such as sucrose in sugar.
What macromolecule does iodine test for?
Iodine solution is used to test for starch. The solution is brownish-yellow, but changes to dark blue/black when it reacts with starch. Biuret solution is used to test for protein.
Why does Benedict's solution react with glucose?
This reaction is caused by the reducing property of simple carbohydrates. The copper (II) ions in the Benedict’s solution are reduced to Copper (I) ions, which causes the color change. … As the concentration of reducing sugar increases, the nearer the final colour is to brick-red and the greater the precipitate formed.
Why is iodine solution used?
Iodine test : Iodine solution is used to test the presence of carbohydrate because carbohydrates constitute of starch and this starch complexes with iodine to form starch iodine complex which is of blue black colour. … Thus Iodine test is usually carried out to test the presence of carbohydrate.
Why does lactose react with Benedict's solution?
Because the aglycone is a hemiacetal, lactose undergoes mutarotation. For the same reason lactose is a reducing sugar. The free aldehyde formed by ring opening can react with Benedict’s solution. Thus, a solution of lactose contains both the α and β anomer at the “reducing end” of the disaccharide.
What solution is used to detect presence of starch in the leaves?
Iodine solution is used to test leaves for the presence of starch. You need to: heat a plant leaf in boiling water for 30 seconds (this stops its chemical reactions)
What is the importance of detecting the presence of carbohydrates?
It is important to determine the type and concentration of carbohydrates in foods for a number of reasons. Food Quality – physicochemical properties of foods such as sweetness, appearance, stability and texture depend on the type and concentration of carbohydrates present.
What tests detect carbohydrates?
Molisch’s test is a general test for carbohydrates. This test is given by almost all of the carbohydrates. In this test, concentrated sulfuric acid converts the given carbohydrate into furfural or its derivatives, which react with α-naphthol to form a purple coloured product.
Which solution is used test for carbohydrates?
Test for Carbohydrates: Tollen’s test – Given sample food + Tollen’s reagent → Silver mirror confirms the presence of carbohydrates. Iodine test – Given sample food + Iodine solution → Blue colour solution confirms the presence of starch.
What is the importance of reducing sugars?
Reducing sugar intake lowers specifically the risk of developing overweight and obesity, and in turn in developing diabetes. It also has a significant effect on lowering dental caries.
How do you identify a reducing agent?
A reducing agent is something that causes another substance to reduce. If the oxidation number is greater in the product than the reactant, then the substance lost electrons and the substance was oxidized. If the oxidation number is less, then it gained electrons and the substance was reduced.
Why is glucose called a reducing sugar?
Glucose is a reducing sugar because it belongs to the category of an aldose meaning its open-chain form contains an aldehyde group. Generally, an aldehyde is quite easily oxidized to carboxylic acids. … Glucose is also a monosaccharide and thus is reducing in nature.
What is reducing sugar answer the following?
A reducing sugar is a carbohydrate that is oxidized by a weak oxidizing agent in basic aqueous solution. A reducing sugar is any sugar that is capable of acting as a reducing agent because it has a free aldehyde group or a free ketone group.
What is reducing sugar class 11 biology?
-Reducing sugar is any sugar that’s capable of acting as a reducer because it’s a free aldehyde radical or a free group. -All monosaccharides are reducing sugars, alongside some disaccharides, some oligosaccharides, and a few polysaccharides.
What is meant by reducing sugar and non reducing sugar?
Lesson Summary. Reducing sugars are sugars where the anomeric carbon has an OH group attached that can reduce other compounds. Non-reducing sugars do not have an OH group attached to the anomeric carbon so they cannot reduce other compounds. All monosaccharides such as glucose are reducing sugars.
Which of the following tests is used to detect the presence of lipids?
The Sudan III test is used to test detect lipids .