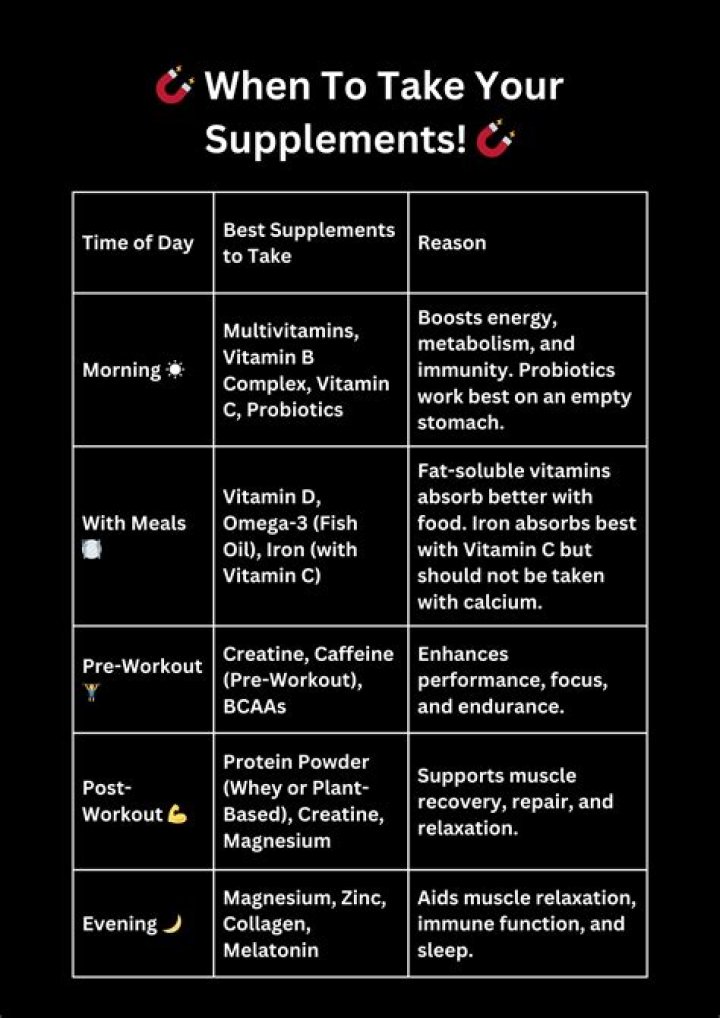
What does freezing point of water mean
: the temperature at which a liquid solidifies specifically : the temperature at which the liquid and solid states of the substance are in equilibrium at atmospheric pressure : melting point the freezing point of water is 0° Celsius or 32° Fahrenheit.
What does freezing point tell you?
The freezing point is the temperature at which the liquid solvent and solid solvent are at equilibrium, so that their vapour pressures are equal. … As a result, the solid will reach equilibrium with the solution at a lower temperature than with the pure solvent.
What does it mean to have a low freezing point?
Changes in pressure can raise or lower the freezing point of a substance. Generally, pressures lower than 1 atmosphere lower the temperature at which a substance freezes, but for water, a higher pressure gives a lower freezing point. … For water at low pressures, vapor directly turns to ice without becoming a liquid.
Why is it important to know the freezing point of water?
Additional Info. Melting points can be determined by visual inspection or by monitoring the temperature of the liquid with time. When the freezing point is reached, the temperature of the solution will no longer drop until all the liquid has frozen.How can you lower the freezing point of water?
Freezing point depression is what occurs when you add a salt to a liquid. A common life example of this is throwing salt on ice or snow in the winter to melt the ice or snow. The action of putting salt on ice lowers the freezing point, which means that the ice will melt at lower temperatures.
What is difference between freezing point and melting point?
The melting point is the temperature at which a solid is changed into a liquid by applying heat and pressure while the freezing point is the temperature at which a liquid is changed into a solid. 2. While most substances, especially pure substances, have the same melting and freezing points.
What lowers the freezing point of water the most?
Sugar lowers the freezing point of water, which makes frozen desserts fair game for changes in freezing point. Most desserts freeze between 29.5 to 26.6 degrees F (-1.4 to -3.0 C) depending on the concentration of sugar. To balance their higher proportion of acids (which give flavors such as lemon, raspberry, etc.
Can freezing point be elevated?
As with the melting point, increased pressure usually raises the freezing point. … Some liquids can be supercooled—i.e., cooled below the freezing point—without solid crystals forming.What is high freezing point?
If the intermolecular forces between its molecules are strong then there is a high freezing point. If the forces are weak, the freezing point is relatively low. The freezing point of a liquid or melting point of solid occurs at the temperature in which the solid and liquid phases are in equilibrium.
What affects freezing point?Most importantly, changes in the pressure can increase or decrease the freezing point of a substance. Typically, pressure lower than 1atm reduces the temperature at which the substance freezes. But in the case of water, the higher the pressure, lower is the freezing point.
Article first time published onDoes water freeze under pressure?
Yes, pressure affects the freezing point of water but not as much as the boiling point. increasing pressure, lowers the melting point of ice.
Does water freeze faster under pressure?
The more molecules dissolved in the water, the lower the freezing point. … One is that at higher pressures, the air will have a higher heat capacity per unit volume than at lower pressures, and so flowing high-pressure cold air past the water can freeze the water faster.
Why does salt lower the freezing point of water more than sugar?
The salt solute is able to depress the freezing point more than the sugar solute because the salt is ionically bonded while the sugar solute is covalently bonded. Because salt is ionically bonded, its ions are able to fully dissociate in solution.
What is the difference between the freezing point and boiling point water?
The freezing point of water on the Kelvin scale is 273.15 K, while the boiling point is 373.15 K. … As can be seen by the 100 kelvin difference between the two, a change of one degree on the Celsius scale is equivalent to the change of one kelvin on the Kelvin scale.
What is the difference between freezing and freezing point?
The key difference between freezing point and freezing point depression is that freezing point is the temperature at which a liquid becomes a solid, whereas freezing point depression is the decrease of the freezing point of a solvent due to the addition of a solute into the solvent.
Why is the freezing and melting point of water the same?
Upon cooling, the particles in a liquid lose energy, stop moving about and settle into a stable arrangement, forming a solid. Freezing occurs at the same temperature as melting, hence, the melting point and freezing point of a substance are the same temperature.
What determines the lowest freezing point?
Remember, the greater the concentration of particles, the lower the freezing point will be. 0.1mCaI2 will have the lowest freezing point, followed by 0.1mNaCl, and the highest of the three solutions will be 0.1mC6H12O6, but all three of them will have a lower freezing point than pure water.
What is the freezing point of water at standard pressure?
Ordinarily, the freezing point of water and melting point is 0 °C or 32 °F. The temperature may be lower if supercooling occurs or if there are impurities present in the water which could cause freezing point depression to occur. Under certain conditions, water may remain a liquid as cold as -40 to -42°F!
Why does freezing point of water decrease with pressure?
We know that with increasing atmospheric pressure, the freezing point of water slightly decreases. Because of increasing the pressure, which prevents water molecules properly arranging them. If water freezes, the mean forms a crystalline solid phase with a proper arrangement. … That means water freezes at below 273K.
Why does water not freeze at 0 degrees?
When dissolved in water, the water molecules tend to stick to the salt ions instead of to each other, and they therefore don’t freeze as readily. As you add more salt to water, its freezing point continues to drop until the water reaches saturation and cannot hold any more salt.
Can water freeze above 0 degrees?
Yes, however only in special circumstances. One good example is black ice. As long as the asphalt is 0 degrees Celsius or below, water can freeze regardless even if the air temperature is above 0 degrees Celsius. Otherwise, it impossible for water to freeze above 0 degrees Celsius.
Is liquid water colder than 32?
After water changes from a liquid to a gas (at 212 degrees Fahrenheit) it can actually heat up much hotter than that. … And water freezes at 32 degrees Fahrenheit. But it can actually get colder than that, all the way toward what we call absolute zero. This value is equivalent to about -459 degrees Fahrenheit.
What is the hardest form of ice?
- Diamond. Its hardness is legendary. …
- Methane. At least it might be harder than nitrogen.
How does water freeze in a pipe?
Instead, it has to do with pressure inside the pipes. When water freezes, its molecules crystalize into an open hexagonal form, which takes up more space than when the molecules are in their liquid form — that is, the water molecules expand as they freeze. As the ice expands, it pushes water toward the closed faucet.
What is the freezing point of water at 1 atm pressure?
The freezing point of water at 1 atmospheric pressure is O Celsius, 273.15K. If the pressure is lowered to the triple point the temperature is +0.01C defined as 273.16K. Increasing P above the triple point decreases the melting point.
What effect did salt have on the freezing point of water?
Salt molecules block water molecules from packing together when temperature is lowered. It then prevents them from becoming ice. More water molecules leave the solid phase than the ones entering the solid phase. Freezing point depression occurs when the freezing point of the liquid is lowered by addition of solute.
How does salt affect water freezing point?
When salt molecules displace water molecules, the freezing rate slows down. … This lowers the freezing point of ocean water to about -1.8° C or 28.8° F. So ocean water will freeze. It just needs to reach a lower temperature.
How does salt affect the freezing point and boiling point of water?
The more salt (or any solute) added to water, the more you raise the boiling point. … Freezing point depression is another colligative property that works the same way: If you add salt to water, you lower its freezing point as well as raise its boiling point.
What is the difference between the boiling point and the freezing point of pure water at standard pressure?
Freezing and Boiling Points: The freezing and boiling points are the temperatures at which the respective changes in the phases occur. For water at standard pressure, we know that its freezing point is 0∘C 0 ∘ C , and its boiling point is 100∘C 100 ∘ C .