What happens when a protein is denatured
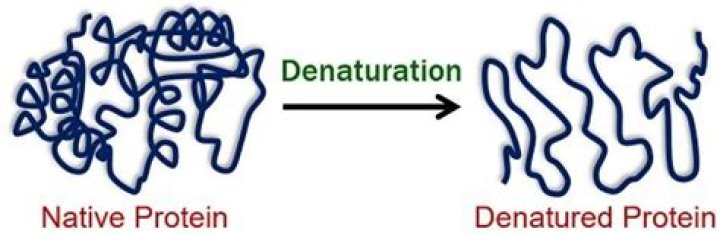
Denaturation involves the breaking of many of the weak linkages, or bonds (e.g., hydrogen bonds), within a protein molecule that are responsible for the highly ordered structure of the protein in its natural (native) state. Denatured proteins have a looser, more random structure; most are insoluble.
What can happen to a protein if its is denatured?
When a protein is denatured, secondary and tertiary structures are altered but the peptide bonds of the primary structure between the amino acids are left intact. Since all structural levels of the protein determine its function, the protein can no longer perform its function once it has been denatured.
What is denaturation of protein and what is its effect?
When a native protein is subjected to change in pH, temperature or chemicals, the tertiary structure of protein gets unfolded, the protein gets denatured. This causes the protein to change biological activity.
What happens when a protein is denatured quizlet?
When a protein is denatured, it disrupts the hydrogen, ionic, and disulfide bridges within it, as well as affecting its temperature, pH (hydrogen structure) and salinity. … Other chemicals that can break the bonds inside the protein that help it keep its shape.When a protein is denatured why does it lose functionality?
Because many of the proteins original bonds have been broken, those parts of the molecule are now available to make new bonds with other substances. This changes the way the molecule reacts. Since a protein’s function is dependent on its shape, a denatured protein is no longer functional.
What causes denaturation of protein quizlet?
How does heat cause denaturing of proteins? Proteins are heat sensitive thus it disrupts the weaker intermolecular linkages (ie. hydrogen bonds). Temperature required for denaturation depends on the protein.
What causes a protein to denature what happens to a protein if it is denatured quizlet?
What happens to the protein when it is denatured? The breakdown or alteration of a protein from the unfolding of polypeptide chains. The bonds in the protein break down into smaller peptide bonds. Proteins are denatured for digestions and are also denatured under intense amounts of heat.
When a protein is denatured which type of bonding is affected?
Protein function depends absolutely on its structure.. In denaturation, the peptide bonds are not affected, but the H-bonds, disulfide bonds, salt bridges and hydrophobic interactions can all be disrupted, leading to the consecutive alteration of 4o, 3o and 2o structure.What happens to protein molecules when you heat them?
When protein is heated, it can ‘denature’– this means the protein molecules unfold or break apart. This is what your body does to protein anyway, breaking down the amino acids and digesting protein. Much like when you cook meat, the protein you gain is not altered by cooking.
How does heat affect protein structure?Effect on Protein Structure Heat or UV radiation supplies kinetic energy to protein molecules, causing their atoms to vibrate more rapidly and disrupting relatively weak hydrogen bonding and dispersion forces.
Article first time published onHow did the proteins change when they were denatured?
The denatured protein has the same primary structure as the original, or native, protein. The weak forces between charged groups and the weaker forces of mutual attraction of nonpolar groups are disrupted at elevated temperatures, however; as a result, the tertiary structure of the protein is lost.
What happens when a protein is denatured chegg?
When a protein undergoes structural changes during denaturation, it leads to the permanent loss of biological properties. The structure of denatured proteins is more random and mostly are insoluble. In protein denaturation, protein often becomes insoluble when it is boiled.
What is protein denaturation?
Denaturation involves the breaking of many of the weak linkages, or bonds (e.g., hydrogen bonds), within a protein molecule that are responsible for the highly ordered structure of the protein in its natural (native) state. Denatured proteins have a looser, more random structure; most are insoluble.
What factors cause denaturation of proteins?
Various reasons cause denaturation of protein. Some of them are an increased temperature that ruptures the protein molecules’ structure, changes in pH level, adding of heavy metal salts, acids, bases, protonation of amino acid residues, and exposure to UV light and radiation.
What are 3 factors that can denature proteins?
Changes in pH, Increased Temperature, Exposure to UV light/radiation (dissociation of H bonds), Protonation amino acid residues, High salt concentrations are the main factors that cause a protein to denature.
How does heat cause denaturation what happens when a protein is denatured by heat?
Heat can be used to disrupt hydrogen bonds and non-polar hydrophobic interactions. This occurs because heat increases the kinetic energy and causes the molecules to vibrate so rapidly and violently that the bonds are disrupted. The proteins in eggs denature and coagulate during cooking.
Why do proteins break down in heat?
Because proteins’ function is dependent on their shape, denatured proteins are no longer functional. During cooking the applied heat causes proteins to vibrate. This destroys the weak bonds holding proteins in their complex shape (though this does not happen to the stronger peptide bonds).
At what temperature does protein breakdown?
It’s a process called denaturation and it involves the breaking down of protein strands by the application of extreme conditions (i.e time and temperature) that ultimately render meat moist and tender. Denaturation begins at roughly 105°F and continues upwards to temperatures in excess of 200°F.
When proteins are denatured which type of bond is not disturbed?
The primary structure of a proteins is not disturbed, because the denaturation reactions are not strong enough to break the covalent peptide bonds. Branden, C. I., & Tooze, J. (2012). Introduction to protein structure. Garland Science.
When a protein is denatured which level of protein structure is unaffected?
5. 7. Protein denaturation disrupts the secondary, tertiary, and quaternary levels of structure. Only primary structure is unaffected by denaturation.
What happens when you denature a protein with heat or pH changes?
If the protein is subject to changes in temperature, pH, or exposure to chemicals, the internal interactions between the protein’s amino acids can be altered, which in turn may alter the shape of the protein.
What changes can cause a protein to unravel?
Denaturation (“changing the nature”) happens when protein molecules unravel from their naturally coiled state. With eggs, this happens most frequently when they’re heated or beaten, but denaturation can also be prompted by salt, acids (such as vinegar), alkalies (such as baking soda), and freezing.
Why is denaturation of proteins important?
The way proteins change their structure in the presence of certain chemicals, acids or bases – protein denaturation – plays a key role in many important biological processes. And the way proteins interact with various simple molecules is essential to finding new drugs. … Journal link: Protein Sci.
What are the advantages of denaturation?
They are especially beneficial to bodybuilders who use denatured proteins. They help aid in protein absorbance and protein digestion since the protein is already broken down. In essence, a denatured protein is already broken up before entering the body as opposed to being broken up in the stomach by acids.
Why do salts need to be at a high concentration to affect the protein structure?
Why do proteins precipitate at high salt concentrations? If the salt concentration becomes too high the salt ions interact with the water molecules. Eventually there are not enough water molecules to interact with the protein and the protein precipitates.
How can a highly concentrated salt denature a protein?
However, at very high salt concentration, the increased surface tension of water generates a competition between protein and salt ions for hydration. Salts strip off the essential layer of water molecules from the protein surface eventually denaturing the protein.
What maintains the tertiary structure of a protein?
Disulfide bonds make proteins less susceptible to unfolding; typically, they will link -sheets, -helices, and loops, which means that they primarily maintain tertiary structure, not secondary, which refers to local conformations, and is maintained largely by hydrogen bonds.
How does denaturation of a protein affect its function quizlet?
Denaturation causes a protein to lose its shape, which leads to losing its function.
Which of the following describe s a difference between protein degradation and protein denaturation?
A. Protein degradation disrupts the peptide bonds, but protein denaturation does not.