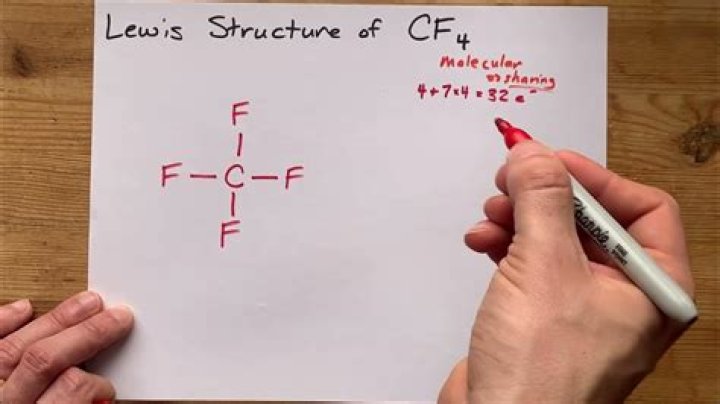
What intermolecular forces are present in carbon tetrafluoride
Carbon tetrachloride has dipole-dipole forces, whereas carbon tetrafluoride only has London dispersion forces.
What intermolecular forces are in carbon tetrafluoride?
Explanation: And only dispersion forces operate in carbon tetrachloride.
Is CF4 dipole-dipole?
Hence the primary inter molecular forces would be dipole – dipole and hydrogen bond which is a special type of dipole – dipole interaction between the hydrogen atom and electronegative F atom. ##CF_4## has a tetrahedral structure. It is non-polar molecule.
Which intermolecular forces does carbon tetrabromide participate in?
Going down the list from weakest to strongest (generally) forces, we know firstly that CBr4 has dispersion forces, which are possessed by all molecules.What is the intramolecular force of CF4?
Step 4: The electronegativity difference between C and F is 1.5 so the C–F bond is polar. The dipole arrows cancel out, making CF4 a nonpolar compound. Since CF4 is nonpolar, it exhibits dispersion forces.
Does carbon tetrafluoride have polar bonds?
Carbon Tetrafluoride is a nonpolar covalent compound. If we look at the bonds individually, Carbon has an electronegativity of 2.5 and fluorine has an electronegativity of 4.0.
What intermolecular forces are present in sulfur dioxide?
SO2 is a polar molecule. Generally dipole-dipole forces are stronger than LDF forces. However, SO3 is significantly larger than SO2. SO3 is also planar.
What type of intermolecular force is carbon disulfide?
Since CS2 is nonpolar, it exhibits dispersion forces. Both compounds only have dispersion forces. The only intermolecular interactions between Bromine and Carbon disulfide are dispersion forces.Does carbon tetrafluoride have dispersion forces?
Carbon tetrachloride has dipole-dipole forces, whereas carbon tetrafluoride only has London dispersion forces.
What type of intermolecular force is carbon dioxide?CO has two C-O bonds. The dipoles point in opposite directions, so they cancel each other out. Thus, although CO₂ has polar bonds, it is a nonpolar molecule. Therefore, the only intermolecular forces are London dispersion forces.
Article first time published onWhat is the dipole moment of CF4?
Carbon (2.55) is less electronegative than fluorine (3.98), which means the C–F bond has a dipole moment. The dipole arrows in CF4 would look like this: The C–F dipole moments just cancel out since they point at opposite directions. This means CF4 has no molecular dipole.
Why is carbon tetrafluoride nonpolar?
CF4 is in a tetrahedral molecular shape, so all the dipoles of the 4 polar C-F bonds cancel each other out, resulting in an overall nonpolar molecule.
What is the strongest type of intermolecular force present in chbr3?
Therefore, the strongest type of IMF present in CHBr3 is dipole-dipole interaction.
Why is chf3 not hydrogen bonding?
601-607. ions. (d) CH3F(l) – Dipole – dipole forces: CH3F is a polar molecule, it has a permanent dipole. In this case hydrogen bonding does NOT occur, since the F atom is bonded to the central C atom (F must be bonded to H in order for hydrogen bonding to occur).
What intermolecular forces are present in HCl?
All molecules and atoms have London dispersion (i.e. van der Waals) forces. With HCl , a polar molecule, all we say is that the predominant intermolecular force is dipole dipole interaction. Even though HCl has dispersion forces, they are overshadowed by dipole-dipole by far.
What intermolecular forces are present in CS2?
Sample Response: CS2 and COS both have London Dispersion Forces, but since COS is a polar molecule, it also exhibits dipole-dipole forces. However, the London Dispersion Forces in CS2 are so strong that they overpower the strength of both the LDFs and the dipole-dipole forces in COS.
Is sulfur dioxide dipole-dipole?
Yes, sulfur dioxide (SO2) is a polar molecule that feature dipole dipole interactions in its intermolecular forces.
What kind of compound is sulfur dioxide?
sulfur dioxide, (SO2), inorganic compound, a heavy, colourless, poisonous gas. It is produced in huge quantities in intermediate steps of sulfuric acid manufacture.
What type of intermolecular force will act in sulfur dioxide and why?
The molecule has a bent shape due to the presence of lone pair of electron. So due to the bent shape of sulfur dioxide it can be inferred that it is a polar molecule. Hence, being a polar molecule sulfur dioxide has dipole-dipole intermolecular forces.
How would you describe a molecule of carbon tetrafluoride CF4?
Name of MoleculeCarbon tetrafluorideChemical formulaCF4
Is NO2 polar or nonpolar?
NO2 polar or nonpolar You know that, nitrogen dioxide is created bonds between nitrogen and oxygen. And other think of nitrogen dioxide is, the different in Electronegativity in Both atom. … result it make polar, so, it is called polar molecules.
What is the strongest intermolecular force in SiF4?
Since SiF4 has a greater molecular mass than SiH4, therefore SiF4 has a greater London dispersion force and a greater boiling point.
Is carbon disulfide made up of atoms or molecules?
Since the molecules consist of atoms, it is not right to assume that carbon disulfide is made of atoms. Know, a molecule has special properties. And no atom has the carbon disulfide properties. Only molecules of carbon disulfide have the properties of carbon disulfide, and it is made of molecules.
Is carbon disulfide ionic or covalent compound?
Answer: CS2 ( Carbon disulfide ) is a covalent bond What is chemical bond, ionic bond, covalent bond? Top Answer. A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms.
What is the strongest intermolecular force in carbon dioxide?
CO2 has polar bonds (O is much more electronegative than C) but the polar bonds ARE symmetrically opposite to one another so CO2 is not a polar molecule and does not have permanent dipole-dipole interactions. The strongest type of intermolecular forces are called hydrogen bonds.
What is the predominant intermolecular forces present in carbon dioxide and methane?
Dispersion forces are the only type of intermolecular force operating between non-polar molecules, for example, dispersion forces operate between hydrogen (H2) molecules, chlorine (Cl2) molecules, carbon dioxide (CO2) molecules, dinitrogen tetroxide (N2O4) molecules and methane (CH4) molecules.
Why is carbon dioxide nonpolar?
Carbon dioxide, which has the chemical formula CO2, is non-polar. It contains two polar bonds that are arranged symmetrically. … But the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment. Thus, the molecule is non-polar.
What is the dipole moment of sf6?
Sulfur hexafluoride is made up of six fluorine atoms and a single sulfur atom. The electronegativity of fluorine is greater than sulfur due to which the S-F bond becomes polar. What is this? Therefore, the S-F bond also gives a non zero dipole moment.
In which cases do you expect deviations from the idealized bond angle?
Since PF3 and SBr2 both have lone pairs of electrons on the central atom, their bond angles should deviate from the idealized bond angle as lone pairs are more repulsive than bonding pairs of electrons.
Is the carbon hydrogen bond polar?
Hydrogen and carbon have similar electronegativity values, so the C—H bond is not normally considered a polar covalent bond. Thus ethane, ethylene, and acetylene have nonpolar covalent bonds, and the compounds are nonpolar.
Is carbon tetrafluoride soluble in water?
NamesSolubility in water0.005%V at 20 °C 0.0038%V at 25 °CSolubilitysoluble in benzene, chloroformVapor pressure3.65 MPa at 15 °C 106.5 kPa at −127 °CHenry’s law constant (kH)5.15 atm-cu m/mole