What is activation energy short definition
activation energy, in chemistry, the minimum amount of energy that is required to activate atoms or molecules to a condition in which they can undergo chemical transformation or physical transport.
What is activation energy * Your answer?
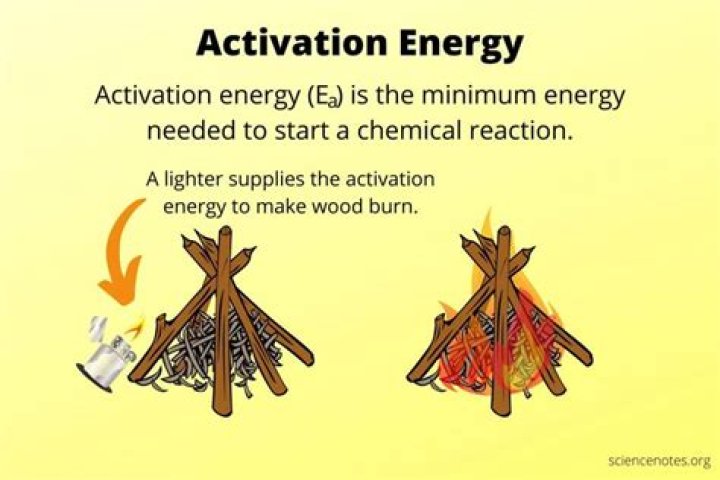
Activation energy is the minimum energy required to cause a process (such as a chemical reaction) to occur.
What is activation energy with example?
They require a certain amount of energy just to get started. This energy is called activation energy. For example, activation energy is needed to start a car engine. Turning the key causes a spark that activates the burning of gasoline in the engine.
What is the definition of activation energy in biology?
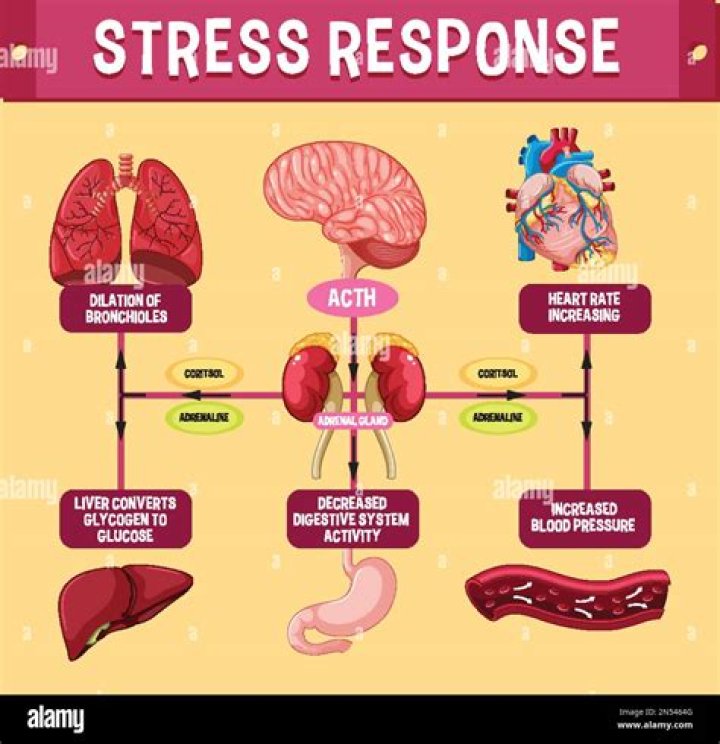
Reactions require an input of energy to initiate the reaction; this is called the activation energy (EA). Activation energy is the amount of energy required to reach the transition state. The source of the activation energy needed to push reactions forward is typically heat energy from the surroundings.What is activation energy called?
Key Points. Reactions require an input of energy to initiate the reaction; this is called the activation energy (EA). Activation energy is the amount of energy required to reach the transition state. The source of the activation energy needed to push reactions forward is typically heat energy from the surroundings.
What is the activation energy of a reaction apex?
Activation energy is the minimum energy required for a chemical reaction to occur, that is, for the bonds within reactants to break and for new bonds to form (the products). This chemical reaction is classified as being either exothermic (thermal energy is released) or endothermic (thermal energy absorbed).
What is the activation energy of a reaction answers?
Correct answer: Activation energy is the difference between the energy of the transition state and the energy of the reactant. Recall that activation energy is the energy barrier that needs to be overcome by a reaction.
What is activation energy Ncert?
The minimum quantitiy of external energy required for the conversion of reactant into product or to produce an unstable intermediate is called activation energy. It is E. Rate of reaction is inversely proportional to the activation energy.What is activation energy class 11?
Activation energy is defined as the minimum amount of extra energy required by a reacting molecule to get converted into product. It can also be described as the minimum amount of energy needed to activate or energize molecules or atoms so that they can undergo a chemical reaction or transformation.
What is activation energy in microbiology?Activation energy is the energy needed to form or break chemical bonds and convert reactants to products (Figure 4). Enzymes lower the activation energy by binding to the reactant molecules and holding them in such a way as to speed up the reaction.
Article first time published onWhat is activation energy and why is it important?
Activation energy is the minimum energy required to cause a reaction to occur. To understand activation energy, we must first think about how a chemical reaction occurs. Anyone who has ever lit a fire will have an intuitive understanding of the process, even if they have not connected it to chemistry.
What do you mean by activation energy and why it is referred in the definition of enzymes?
The activation energy is the energy required to start a reaction. Enzymes are proteins that bind to a molecule, or substrate, to modify it and lower the energy required to make it react. The rate of reaction is given by the Arrhenius equation. The rate of reaction increases if the activation energy decreases.
What is activation in chemistry?
Chemistry. In chemistry, “activation” refers to the reversible transition of a molecule into a nearly identical chemical or physical state, with the defining characteristic being that this resultant state exhibits an increased propensity to undergo a specified chemical reaction.
What is activation energy in thermodynamics?
Activation energy can be thought of as the magnitude of the potential barrier (sometimes called the energy barrier) separating minima of the potential energy surface pertaining to the initial and final thermodynamic state. … The term Activation Energy was introduced in 1889 by the Swedish scientist Svante Arrhenius.
What is the activation energy quizlet?
Activation energy is the energy required to break existing bonds, and form new bonds. If a collision occurs with more energy than the activation energy, the reaction will occur. … The higher the activation energy, the more energy is required for a collision to be effective.
What is activation energy a level?
The activation energy is the minimum energy required for a reaction to occur. This means that the reactant molecules have enough kinetic energy to collide successfully and overcome the repulsion caused by outer electrons.
What is the activation energy in a graph?
In a diagram, activation energy is graphed as the height of an energy barrier between two minimum points of potential energy. The minimum points are the energies of the stable reactants and products. Even exothermic reactions, such as burning a candle, require energy input.
How is activation energy determined?
Determining Activation Energy. Notice that when the Arrhenius equation is rearranged as above it is a linear equation with the form y = mx + b; y is ln(k), x is 1/T, and m is -Ea/R. The activation energy for the reaction can be determined by finding the slope of the line.
What does the activation energy Ea represent group of answer choices?
The Activation Energy (Ea) – is the energy level that the reactant molecules must overcome before a reaction can occur.
What is the purpose of a catalyst apex?
A catalyst speeds up a chemical reaction by lowering the amount of energy required for the reaction to take place.
How do enzymes affect the activation energy of a reaction apex?
Enzymes affect the activation energy by lowering it. Since the apex of the energy curve describes the energy of the transition state, we shall see shortly that enzymes actually stabilize the transition state and lower its energy.
What is activated complex class 12?
When reactant molecules absorb energy, their bonds loosened and new loose bonds are formed between them. The intermediate thus formed is called an activated complex or transition state. It is unstable and immediately dissociates to form the stable products.
What is activation energy and threshold energy?
The minimum energy that molecules need to have in order for a reaction to take place is called the threshold energy. … The difference between the threshold energy and whatever internal energy molecules have is called the activation energy.
What is activation energy of a reaction Class 12?
Activation energy: The activation energy (Ea) is defined as the minimum kinetic energy required for a molecular collision to lead to reaction.
Why is activation energy high?
If the molecules in the reactants collide with enough kinetic energy and this energy is higher than the transition state energy, then the reaction occurs and products form. In other words, the higher the activation energy, the harder it is for a reaction to occur and vice versa.
What is activation energy and how do enzymes affect if?
Enzymes are biological catalysts. Catalysts lower the activation energy for reactions. The lower the activation energy for a reaction, the faster the rate. Thus enzymes speed up reactions by lowering activation energy.
What is the activation energy of an exothermic reaction?
Figure 12.4: The energy changes that take place during an exothermic reaction. The activation energy is the difference between the energy of the reactants and the maximum energy (i.e. the energy of the activated complex).
What is activation process?
In the activation process, the functional groups present on the surface of polymeric material are replaced with atoms or chemical groups from the plasma. In this process, upon plasma treatment the plasma breaks the polymer’s backbone or groups from the backbone, creating free radicals on the surface.
What is activation energy in collision theory?
In order to effectively initiate a reaction, collisions must be sufficiently energetic (kinetic energy) to break chemical bonds; this energy is known as the activation energy. As the temperature rises, molecules move faster and collide more vigorously, greatly increasing the likelihood of bond breakage upon collision.