What is free energy ap bio
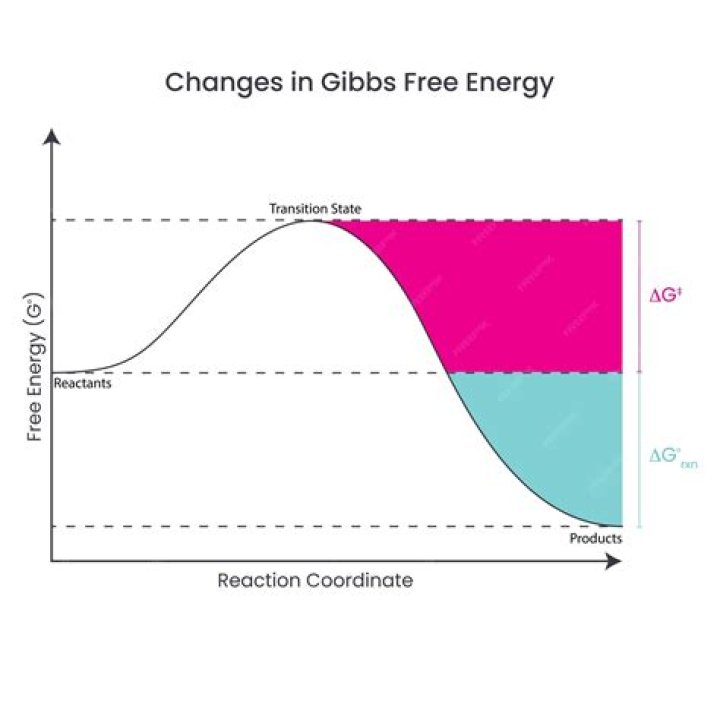
Free energy. the total amount of energy in a system (a cell) that can be tapped to do work. Not all energy transfers are 100% Exergonic Reactions.
What exactly is free energy?
In physics and physical chemistry, free energy refers to the amount of internal energy of a thermodynamic system that is available to perform work. There are different forms of thermodynamic free energy: … Helmholtz free energy is energy that may be converted into work at constant temperature and volume.
Why it is called free energy?
In 1882, the German physicist and physiologist Hermann von Helmholtz coined the phrase ‘free energy’ for the expression E − TS, in which the change in A (or G) determines the amount of energy ‘free’ for work under the given conditions, specifically constant temperature.
What is free energy in ecology?
Thermodynamic free energy refers to the energy available in a physical system (for instance a gas) that can be put to useful work. For example, consider a collection of gas molecules with the same kinetic energy. Only part of this energy is available to do work.What is free energy and free energy change?
The standard free energy change (∆Gº’) of a chemical reaction is the amount of energy released in the conversion of reactants to products under standard conditions.
What is free energy describe its role in bio chemical reactions?
If a chemical reaction requires an input of energy rather than releasing energy, then the ∆G for that reaction will be a positive value. In this case, the products have more free energy than the reactants. Thus, the products of these reactions can be thought of as energy-storing molecules.
What is free energy of a molecule?
5.2 Free energy. Free energy or Gibbs free energy G, is the energy available in a system to do useful work and is different from the total energy change of a chemical reaction.
Why is free energy necessary to living things?
ATP is an essential molecule – without it, livings things cease to live. Organisms use free energy for many other things, such as growth and reproduction. For example, your body’s temperature and metabolism depend on you taking in free energy and matter. It also takes a lot of energy to produce and rear offspring.What is free energy quizlet?
every energy transfer increases the entropy of the universe (disorder) Free energy. the total amount of energy in a system (a cell) that can be tapped to do work. Not all energy transfers are 100% Exergonic Reactions.
What are the conditions of free energy?As with standard heats of formation, the standard free energy of a substance represents the free energy change associated with the formation of the substance from the elements in their most stable forms as they exist under the standard conditions of 1 atm pressure and 298K.
Article first time published onWhat is Gibbs free energy and its significance?
The Gibbs free energy is the available energy of a substance that can be used in a chemical transformation or reaction. Substances tend to transform into other substances that have less Gibbs free energy. The change of Gibbs free energy predicts whether a chemical reaction will occur spontaneously.
What does higher free energy mean?
The balance between reactants and products in a reaction will be determined by the free energy difference between the two sides of the reaction. The greater the free energy difference, the more the reaction will favor one side or the other.
What does Gibbs free energy tell us?
Gibbs free energy measures the useful work obtainable from a thermodynamic system at a constant temperature and pressure. Just as in mechanics, where potential energy is defined as capacity to do work, similarly different potentials have different meanings.
What is free energy Slideshare?
ΔGº = ΔHº – TΔSº Gibbs free energy (J or kJ) enthalpy Temperature (K) entropy Gibbs free energy is the energy that is available to do useful work. A reaction will spontaneously occur if ΔG<0 (exergonic reaction) A reaction will NOT spontaneously occur if ΔG>0 (endergonic reaction)
Is free energy the same as potential energy?
Although cells and organisms require free energy to survive, they cannot spontaneously create energy, as stated in the Law of Conservation of Energy. … The chemical energy in molecules, such as glucose, is potential energy because when bonds break in chemical reactions, free energy is released.
Why free energy is not possible?
Free energy machines do not work. No machine can create energy out of nothing, as this would violate the law of mass-energy conservation, which is fundamental and universal. … Mass can be converted to energy, and energy can be converted to mass, but together they must be conserved.
How does Gibbs free energy relate to equilibrium?
Both K and ΔG° can be used to predict the ratio of products to reactants at equilibrium for a given reaction. ΔG° is related to K by the equation ΔG°=−RTlnK. If ΔG° < 0, then K > 1, and products are favored over reactants at equilibrium. If ΔG° > 0, then K < 1, and reactants are favored over products at equilibrium.
Which has more free energy reactants or products?
In a mixture of reactants and products, the chemical reaction and its reverse occur until chemical equilibrium is achieved. If we begin with a large concentration of reactants, the free energy of reactants is much greater than products and the reaction proceeds.
What does a positive change in the Gibbs free energy value represent quizlet?
What the ΔH tell us in the Gibbs free energy change equation? A negative ΔH means heat is released in going from reactants to products; a positive ΔH means heat is absorbed.
How do humans use free energy?
The breaking down of food particles we consume and derive energy from is called metabolism. … Thus, ATP production in cells stores Gibbs free energy which can then be used for other processes with the help of the reverse reaction which converts ATP to ADP and, in the process, releases 30.5 kJ of energy per ATP molecule.
What do plants do with free energy?
Glucose that is consumed is used to make energy in the form of ATP, which is used to perform work and power chemical reactions in the cell. During photosynthesis, plants convert light energy into chemical energy that is used to build molecules of glucose.
What is Gibbs energy biology?
Gibbs free energy specifically refers to the energy associated with a chemical reaction that is available after accounting for entropy. In other words, Gibbs free energy is usable energy or energy that is available to do work.
What is Gibbs energy class 11?
Gibbs Energy is the maximum (or reversible) work that a thermodynamic system can perform at a constant temperature and pressure. The reversible work in thermodynamics implies a special method in which work is carried out such that the system remains in perfect equilibrium with all its surroundings.
What is meant by Gibbs free energy class 11?
Gibbs free energy is that thermodynamic quantity of a system the decrease in whose value during a process is equal to the maximum possible useful work that can be obtained from the system. … Under constant pressure conditions , the expansion work is given by PΔV.
What is an example of free energy?
For example, the energy for the maximum electrical work done by a battery as it discharges comes both from the decrease in its internal energy due to chemical reactions and from the heat TΔS it absorbs in order to keep its temperature constant, which is the ideal maximum heat that can be absorbed.
How do you find the free energy difference?
- ΔGchange in free energy=ΔHchange in enthalpy−TΔS(temperature) change in entropy.
- aA+bB→cC+dD.
- ΔrGo=cΔfGo(C)+dΔfGo(D)−aΔfGo(A)−bΔfGo(B)
- ΔfG0=∑vΔfG0(products)−∑vΔfG0(reactants)
- ΔGo=ΔHo−TΔSo.
What is the difference between Gibbs free energy and enthalpy?
The key difference between free energy and enthalpy is that free energy gives the total energy available to perform thermodynamic work whereas enthalpy gives the total energy of a thermodynamic system that can be converted to heat.
Where does Gibbs free energy come from?
The Gibbs free energy of a system at any moment in time is defined as the enthalpy of the system minus the product of the temperature times the entropy of the system. The Gibbs free energy of the system is a state function because it is defined in terms of thermodynamic properties that are state functions.
What is Gibbs free energy PDF?
Gibbs energy is the capacity of a system to do non-mechanical work and ΔG measures the non- mechanical work done on it. The Gibbs free energy is the maximum amount of non-expansion work that. can be extracted from a closed system; this maximum can be attained only in a completely reversible.
What is free energy Wikipedia?
Free-energy relationship, a relationship in physical organic chemistry. Principle of minimum energy, a thermodynamic formulation based on the second law. Thermodynamic free energy, the energy in a physical system that can be converted to do work, including: Gibbs free energy.
What is entropy Slideshare?
Conclusion Entropy is the thermodynamic property which is the measure of disorder in a system. It can be expresses by ‘S’=q/t The term is coined by Rudolf Clausius. Entropy is mainly associated with heat and temperature.



