What is PES in chemistry
Photoelectron spectroscopy (PES) is an experimental technique used to determine the relative energies of electrons in atoms and molecules.
What is PES in AP chemistry?
Photoelectron spectroscopy (PES) is a method to compare the relative energies of atoms, ions, and molecules. PES uses energy from electrons emitted through the photoelectric effect to provide insight about the electronic configuration of a sample.
What are two types of photons generally used in PES?
The light source in a PES experiment is a UV or X-ray source. UV light can be generated using a helium lamp, but the intensity of such light sources isn’t great for most modern experiments, which turn to laser sources for UV light, or to light generated by synchrotron sources for high-energy UV (VUV) or X-ray light.
What is PES used for?
Price elasticity of supply (PES or Es) is a measure used in economics to show the responsiveness, or elasticity, of the quantity supplied of a good or service to a change in its price or cost.What is the PES for most goods?
The PES for perfectly elastic supply is infinite, where the quantity supplied is unlimited at a given price, but no quantity can be supplied at any other price.
Why is PES positive?
The Price Elasticity of Supply is always positive because the Law of Supply says that quantity supplied increases with an increase in price. … If the supply is inelastic, firms find it hard to change production in a given time period.
What are the determinants of PES?
Factors that Influence the PES There are numerous factors that impact the price elasticity of supply including the number of producers, spare capacity, ease of switching, ease of storage, length of production period, time period of training, factor mobility, and how costs react.
What is an orbital diagram in chemistry?
Orbital diagrams are pictorial descriptions of the electrons in an atom. … According to the Auf Bau Principle, each electron occupies the lowest energy orbital. The Pauli Exclusion Principle says that only two electrons can fit into an single orbital.What are the applications of XPS?
XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, coatings, viscous oils, glues, ion-modified materials and many others.
What is coulombic attraction?Coulombic attraction is a fancy name for the attraction that occurs between oppositely charged particles. In chemistry, we talk about the attraction between protons (+) in the nucleus to the electrons (-) surrounding the nucleus.
Article first time published onWhat is a PES chart?
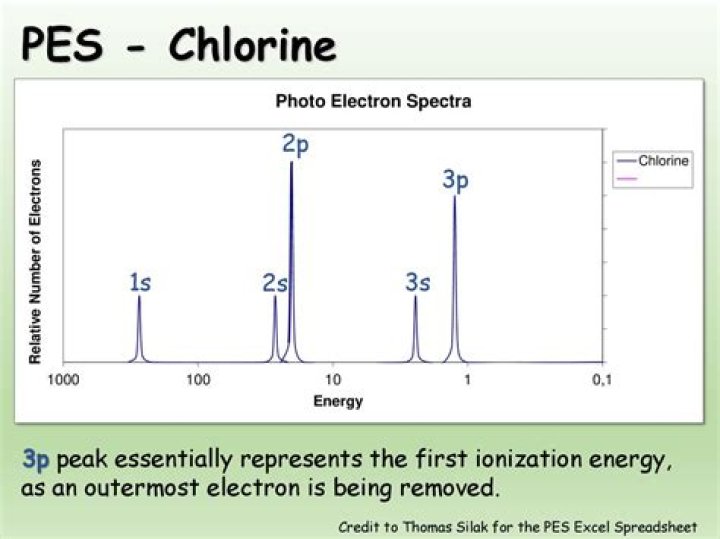
A PES spectrum is a graph of photoelectron count vs. binding energy. The peaks in a PES spectrum correspond to electrons in different subshells of an atom. The peaks with the lowest binding energies correspond to valence electrons, while the peaks with higher binding energies correspond to core electrons.
What is called binding energy?
binding energy, amount of energy required to separate a particle from a system of particles or to disperse all the particles of the system. Binding energy is especially applicable to subatomic particles in atomic nuclei, to electrons bound to nuclei in atoms, and to atoms and ions bound together in crystals.
What is binding energy in XPS?
Atoms present in compound being tested by XPS are determined according to the equation: Here, binding energy is the energy of an electron attracted to a nucleus; photon energy is the energy of X-ray photons being used by the spectrometer, and the kinetic energy is the energy of the ejected electrons from the sample.
What is electronic spectroscopy?
electron spectroscopy, method of determining the energy with which electrons are bound in chemical species by measuring the kinetic energies of the electrons emitted upon bombardment of the species with X-ray or ultraviolet radiation.
Why is AES surface sensitive?
Surface sensitivity in AES arises from the fact that emitted electrons usually have energies ranging from 50 eV to 3 keV and at these values, electrons have a short mean free path in a solid.
What is the first ionization energy?
By definition, the first ionization energy of an element is the energy needed to remove the outermost, or highest energy, electron from a neutral atom in the gas phase.
Why are there only two peaks and not three in the lithium spectrum?
24. Why are there only two peaks and not three in the lithium spectrum in Model 4? Two of the electrons in lithium have the same ionization energy, so there are only two peaks.
Why are photoelectrons called photoelectrons?
When light shines on a metal, electrons can be ejected from the surface of the metal in a phenomenon known as the photoelectric effect. This process is also often referred to as photoemission, and the electrons that are ejected from the metal are called photoelectrons.
How does adding one more proton affect a PES?
Adding or removing protons from the nucleus changes the charge of the nucleus and changes that atom’s atomic number. So, adding or removing protons from the nucleus changes what element that atom is! For example, adding a proton to the nucleus of an atom of hydrogen creates an atom of helium.
How is PES calculated?
Price elasticity of supply measures the responsiveness of quantity supplied to a change in price. The price elasticity of supply (PES) is measured by % change in Q.S divided by % change in price. If the price of a cappuccino increases by 10%, and the supply increases by 20%. We say the PES is 2.0.
Why is PES important for government?
Price elasticity of supply is important for government because it can tell the government something about what the incidence of taxes will be. When governments impose taxes, they can be felt more or less by different groups of people.
How is demand derived?
Derived demand is related solely to the demand placed on a good or service for its ability to acquire or produce another good or service. Derived demand can be spurred by what is required to complete the production of a particular good, including the capital, land, labor, and necessary raw materials.
What are the 4 types of elasticity?
Four types of elasticity are demand elasticity, income elasticity, cross elasticity, and price elasticity.
Why is ped always positive?
The price elasticity of supply is generally positive because the supply curve slopes upward. The income elasticity of demand is the percentage change in the quantity demanded divided by the percentage change in income. … If the income elasticity of demand is positive, it is a normal good.
Why is PES inelastic?
Supply is price inelastic if a change in price causes a smaller percentage change in supply. In this case, an increase in price from £30 to £40 has led to an increase in quantity supplied from 15 to 16. With a PES of 0.2, it is inelastic because PES is less than one.
How can I increase my PES?
- Creating spare capacity.
- Using the latest technology.
- Keeping sufficient stocks.
- Developing better storage systems.
- Prolonging the shelf life of products.
- Developing better distribution systems.
- Providing training for workers.
- Having flexible workers who can do a range of jobs.
What is equilibrium price?
The equilibrium price is the only price where the plans of consumers and the plans of producers agree—that is, where the amount consumers want to buy of the product, quantity demanded, is equal to the amount producers want to sell, quantity supplied. This common quantity is called the equilibrium quantity.
How do you calculate PED?
The price elasticity of demand (PED) is calculated by dividing the percentage change in quantity demanded by the percentage change in price.
Why XPS does not detect hydrogen and helium?
Hydrogen and helium are essentially impossible to detect by a lab-based XPS. … Hydrogen also has an extremely small photoelectron cross-section and suffers from having to share its only electron in forming compounds, which then resides in a valence-like orbital.
What XPS means?
XPS is the abbreviation of XML Paper Specification. It is a page description language which can describe a single page or a document containing multiple pages. The description includes all the text and graphics that appear on the page(s).
What does XPS show?
X-ray photoelectron spectroscopy (XPS), also known as electron spectroscopy for chemical analysis (ESCA), is a technique for analyzing a material’s surface chemistry. XPS can measure elemental composition as well as the chemical and electronic state of the atoms within a material.