What is the atomic number equal to
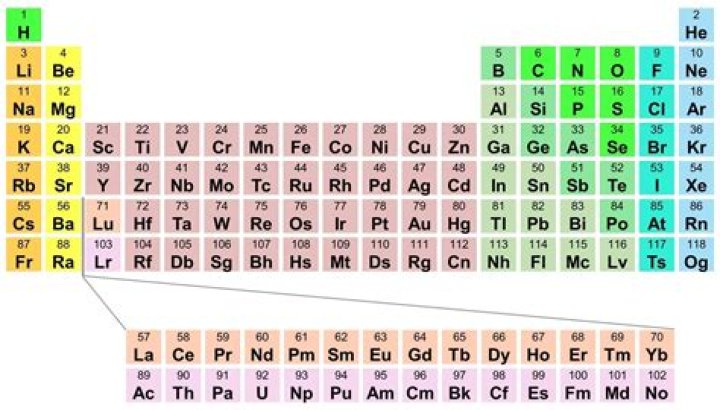
atomic number, the number of a chemical element in the periodic system
What is the atomic number equivalent to?
The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons.
What is the atomic number of this element 2 8 2?
An element has its electron configuration as 2, 8, 2. a. The atomic number of this element is 12.
What is the atomic number equal to quizlet?
The atomic number is equal to the number of protons. The mass number is equal to the sum of the number of protons and the number of neutrons .Is the mass number the same as the atomic weight?
The atomic weight is an actual mass (made relative, i.e., a ratio), while the mass number is a counted number (and so an integer). This weighted average can be quite different from the near-integer values for individual isotopic masses. For instance, there are two main isotopes of chlorine: chlorine-35 and chlorine-37.
Which number is equal to the number of protons in the atom quizlet?
Atomic number is equal to the number of protons in the atom. You just studied 16 terms!
How do you find the atomic number?
The atomic number of an atom is equal to the number of protons in the nucleus of an atom or the number of electrons in an electrically neutral atom. For example, in a sodium atom, there are 11 electrons and 11 protons. Thus the atomic number of Na atom = number of electrons = number of protons = 11.
Why is chlorine CL and not ch?
Answer: Chlorine was discovered by Humphry Davy in the early 19th century and was named after the Greek word ‘chloros’ which meant that it had to be differentiated from carbon with the addition of either h or l after the case of carbon.What is the atomic number of Cl 1?
Chlorine is a chemical element with symbol Cl and atomic number 17.
What is the atomic number of this element 2 8 3?please answer asap. Atomic number of the element = 2+8+3=13, so it is aluminum. It has 3 valence electrons. = 13 .
Article first time published onWhat is the atomic number of 282?
RoentgeniumMass number[282] (unconfirmed: 286)Roentgenium in the periodic table
How is atomic weight calculated?
The atomic weight of any atom can be found by multiplying the abundance of an isotope of an element by the atomic mass of the element and then adding the results together. This equation can be used with elements with two or more isotopes: Carbon-12: 0.9889 x 12.0000 = 11.8668.
What is difference between atomic number and atomic mass?
The major difference between atomic number and mass number is that the atomic number states the number of protons present in an atom whereas, the mass number indicates the total of the number of protons and the number neutrons present in an atom.
How is atomic mass calculated?
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
What is the number of protons equal to?
The number of protons in the nucleus of the atom is equal to the atomic number (Z). The number of electrons in a neutral atom is equal to the number of protons.
Which number is equal to the number of protons in an atom?
The number protons equals the number of electrons in a NEUTRAL atom.
When an atom has an equal number of protons and electrons it is said to quizlet?
When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The total electric charge of the atom is therefore zero and the atom is said to be neutral.
What is the atomic number of chlorine 36?
GeneralNameschlorine-36, Cl-36Protons17Neutrons19Nuclide data
What is CI on the periodic table?
Chlorine – Element information, properties and uses | Periodic Table.
Why is chlorine greenish yellow in Colour?
The halogens darken in colour as the group is descended: thus, while fluorine is a pale yellow gas, chlorine is distinctly yellow-green. This trend occurs because the wavelengths of visible light absorbed by the halogens increase down the group.
Why chlorine is written as Cl2?
Chlorine is called Cl2 because it is a diatomic molecule. Diatomic means there are two atoms of the same elements in the molecule. There are other diatomic molecule, such as hydrogen (H2), nitrogen (N2), oxygen (O2), fluorine (F2), chlorine (Cl2), bromine (Br2), iodine (I2).
What element is atomic number 37?
So there we have rubidium, the explosive red element number 37 in the periodic table.
What is the atomic number of 283?
CoperniciumDensity (near r.t. )14.0 g/cm3 (predicted)Triple point283 K, 25 kPa (predicted)Atomic properties
What is the atomic number of the element 283?
This element is Mg. Therefore, The atomic number of this element is 12.
What is the atomic number of element of Period 3 and group 17?
Answer: it’s chlorine and its atomic number is 17.
What is element 112 on the periodic table?
atomic number112atomic weight285electron config.[Rn]5f146d107s2
How many neutrons does RG have?
Electrons:111Protons:111Neutrons:170Electron Configuration:[Rn] 5f14 6d10 7s1Atomic Radius:114 pm (predicted)