What is the covalency of n2o4
So oxidation state of N in N2O4 is +4 and covalency is also +4.
What Covalency is N2O4?
So oxidation state of N in N2O4 is +4 and covalency is also +4.
What is the covalency of nitrogen in n2o5 and N2O4?
The covalence of nitrogen in N2O5 is 4.
What is the covalency of n2o5?
Covalency is 4. Therefore, the covalency of nitrogen in nitrogen pentoxide is 4.How do you calculate Covalency?
Covalency is the number of bonds an atom forms within a molecule. To determine the covalency, you draw the Lewis structure of the molecule and count the number of shared electron pairs.
What is the covalency of N2O 3?
Nitrogen has a maximum covalency 4.
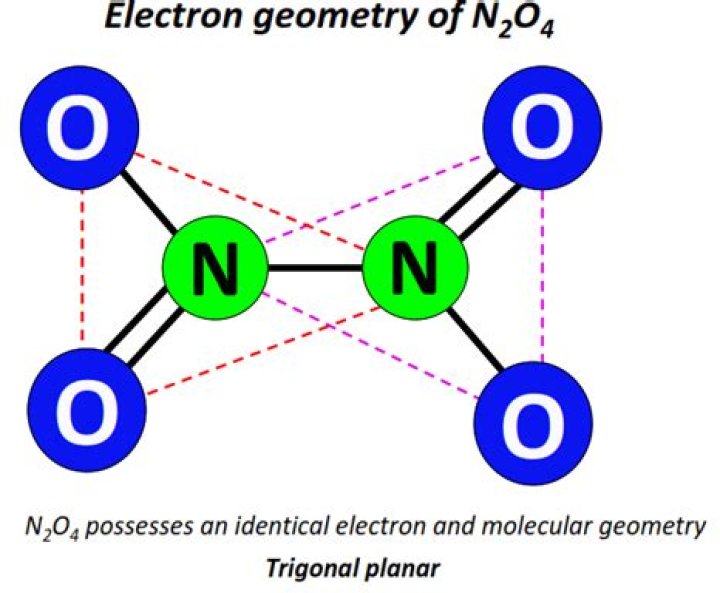
What is N2O4 structure?
Dinitrogen tetroxide is a one of the oxide of nitrogen and are two nitrogen atoms are located at center of the molecule. In Lewis Structure of N2O4, two oxygen atoms have connected to one nitrogen atom. There are charges on atoms in Lewis Structure of N2O4.
What is the covalency of NO2?
Let the valency of nitrogen is X. We know that the valency of oxygen is +2. Therefore, individually we can find the valency of N. Therefore, the Oxidation state of nitrogen in NO is +2,in NO2 is +4 and in N2O is +1 .What is Covalency of nitrogen?
Covalency of Nitrogen – Nitrogen has 5 valence electrons. It needs 3 electrons to complete its octet. It can share three valence electrons with nitrogen atom to acquire stable electronic configuration to form N2. The covalency of nitrogen is 3.
How do you calculate the covalency of nitrogen?Nitrogen has tendency to form pπ-pπ multiple bonds, which decides the structure of oxides. N2O5 has following structure: From the above structure, it is clear that Nitrogen atom is sharing its electrons with oxygen atom. Nitrogen shares it four pair of electrons with oxygen, therefore nitrogen covalency is four (4).
Article first time published onWhy nitrogen Covalency is 4?
It will be 4 only because N can accommodate a maximum of 8 electrons in its outermost shell. When its three 2p electrons bond with H the octet becomes completely filled. So, neither can any more covalent bonds be formed nor can the lone pair be broken and thus one coordinate bond can be formed.
What is the Valency of nitrogen in n2o 5?
It contains 2 nitrogen atom combined with 5 oxygen atom. Here in N2O5, nitrogen atom shares its electrons with 5 oxygen atom and the valency of nitrogen in N2O5 will be 5+.
How do you find the covalency of the central atom?
The central atom is usually the atom with the lowest subscript in the molecular formula and the atom that can form the most bonds. If all of the atoms usually form the same number of bonds, the least electronegative atom is usually the central atom.
What is the covalency of chlorine?
Total number of valence electrons of chlorine is 7, so its maximum covalency is also 7. -Hence, the maximum covalency of chlorine is 7.
What is the covalency of sodium?
The number of electrons lost or gained by an atom to get stable electronic configuration in forming ionic bond is called its electrovalency. Ex: In getting Neon gas configuration Sodium (Na) loses one electron to Chlorine. Thus electrovalency of Sodium is 1.
Is N2O4 empirical formula?
“The chemical formula of a compound written with the smallest possible integers. (ex. H(4)O(2) is a chemical formula for water ; H(2)O is the empirical formula).” While N2O4 And N2H6 are not empirical formulas because the formula integers are not smallest or not simple that can be divided by two to become simple.
What is the oxidation number of Cl2?
However, for Chlorine gas, the oxidation number (Cl2) is zero.
Is 2no2 n2o4 a redox reaction?
It is a redox reaction. Balanced equation: Cu + 4 HNO3 → CU(NO3)2 + 2 NO2 + 2H2O.
What is the valency of nitrogen in n2o4?
You’re answer – We know the valency of oxygen is (-2). Therefore, Oxidation state of nitrogen in NO is +2 ,in NO2 is +4 and in N2O is +1 .
What is the valency of copper in cu2o?
Cuprous oxide or Cu2O is one of the principle oxides of copper. Its valency is 1.
What is the covalency of nitrogen in hno3?
Nitrogen having a covalency of three has a non-bonding or lone pair of electrons with it. Hence, option C is the right answer.
What is Covalency of Aluminium?
The maximum covalency of Aluminium is 6. Since Aluminium belongs to the 3rd period, it possesses a 3d subshell as well. As a cation it can use sp3d2 hybrid orbitals for a covalency of 6.
What nitrogen has highest Covalency?
Covalency of an atom in a covalent compound is the number of covalent bonds formed by an atom. Maximum covalency of Nitrogen is 4 (example – ammonium ions).
Why does r3p O exist?
Nitrogen does not contains d-orbitals. As a result, it cannot expand its covalency beyond four and cannot form pπ – dπ multiple bonds. In constrast, P contains the d-orbitals, and can expand its covalency beyond 4 and can form pπ-dπ multiple bonds. Hence R3P = O exist but R3N = O does not.
Can nitrogen have 5 bonds?
So if you are following the rules, you might well assume that nitrogen would be able to form five bonds (after all, it has five valence electrons). But when we look carefully, we never see a nitrogen atom making five bonds, and in all stable compounds it makes only three bonds.
Why nitrogen Valency is 3?
The nitrogen atom has 5 electrons in the outermost shell, so it can accept 3 electrons to fulfil the octet structure. Therefore the valency of nitrogen in NH3 is 3.
What is the covalency of phosphorus?
Its maximum covalency will be 15-10, that is 5. But in ground state, it can only lose three electrons of 3p orbitals as the two electrons of 3s are intact due to high nuclear charge. So, we can say that the covalency of phosphorus atoms in ground state is 3.
What is the Valency of phosphorus 3?
the valency is the combining capacity of the atom. therefore the valency of phosphorus is 3 and 5.
What is the central atom in HCN?
Central atom:CN contibutes:3 e−Total VSE:8Total VSEP:41 x triple bond:− 2 pairs
What is the central atom of HF?
In the HF molecule, fluorine is a core central atom with one hydrogen atom connected to it. It has three lone pairs of electrons on fluorine.