What is the definition of mass number in chemistry
mass number, in nuclear physics, the sum of the numbers of protons and neutrons present in the nucleus of an atom.
What is a mass number in chemistry?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. … The mass number is different for each different isotope of a chemical element.
What is mass number example?
(ii) Mass number: It is the sum of the number of neutrons and the number of protons. For example, the atomic number of Lithium is 4 which is equal to the number of protons, the number of neutrons of magnesium is 4. The mass number is equal to 8(4+4).
What is the mass number number?
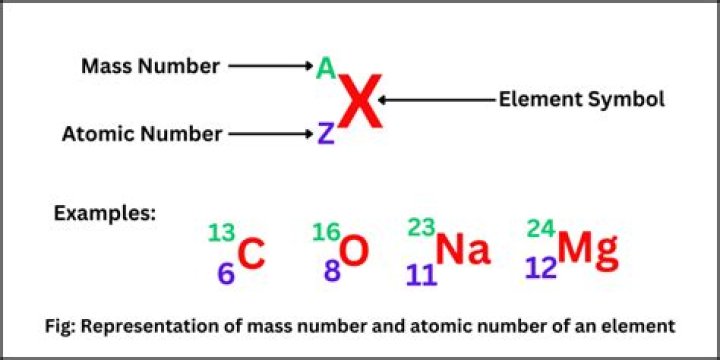
The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom. Consider the table below, which shows data from the first six elements of the periodic table. Consider the element helium. Its atomic number is 2, so it has two protons in its nucleus.What is the definition of atomic number and mass number?
The number of protons determines an element’s atomic number and is used to distinguish one element from another. … Together, the number of protons and the number of neutrons determine an element’s mass number.
What is the mass number of an isotope?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
What is the mass number of all elements?
ATOMIC NUMBERELEMENTATOMIC MASS1Hydrogen1.0082Helium4.00263Lithium6.944Beryllium9.0122
What is mass of nucleus?
The mass of the nucleus is associated with the atomic mass number, which is the total number of protons and neutrons in the nucleus of an atom. The mass number is different for each different isotope of a chemical element.Why is mass number always a whole number?
It is also called atomic mass number or nucleon number. Different isotopes of the same element have different mass numbers because their nuclei contain different numbers of neutrons. … The mass number is always a whole number; because it is a count of the particles.
What is class 11 mass number?What is Mass Number? The number of protons and neutrons combined to give us the mass number of an atom. It is represented using the letter ‘A. ‘ As both protons and neutrons are present in the nucleus of an atom, they are together called nucleons.
Article first time published onWhat is difference between mass number and atomic mass?
The mass number is the sum of the number of protons and neutrons in an atom. It is a whole number. The atomic mass is the average number of protons and neutrons for all natural isotopes of an element.
Where is the mass number located?
On the periodic table, the mass number is usually located below the element symbol. The mass number listed is the average mass of all of the element’s isotopes. Each isotope has a certain percentage abundance found in nature, and these are added and averaged to obtain the average mass number.
What is the mass number of the first 20 elements?
Atomic NumberElementAtomic Mass18Argon39.94819Potassium39.09820Calcium40.07821Scandium44.956
What is the defined mass of a proton?
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
What is the mass of 1 neutron?
ParticleSymbolMass (amu)protonp+1electrone−5.45 × 10−4neutronn01
How do you calculate mass number?
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
Why mass number is fraction?
Atomic Mass is calculated by taking the weighted mean of all isotopes according to the amounts of them present on earth. This creates fractions. Hence Atomic can be fractional but Mass number is always a whole number.
What is atomic number called?
The atomic number or proton number (symbol Z) of a chemical element is the number of protons found in the nucleus of every atom of that element. … It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons.
What is the mass of an atom?
Atomic mass is defined as the number of protons and neutrons in an atom, where each proton and neutron has a mass of approximately 1 amu (1.0073 and 1.0087, respectively). The electrons within an atom are so miniscule compared to protons and neutrons that their mass is negligible.
What is mass number in Chemistry for Class 9?
The sum of the total number of protons and neutrons present in the nucleus of an atom is called the mass number.
What is Class 9 mass number?
Mass number of an atom is defined as the sum of the number of protons and number of neutrons. Mass number is nearly equal to the atomic mass of an atom. Since, protons and neutrons reside in the nucleus, thus they are also known as nucleons. This means.
What is atom in science class 8?
An atom is defined as the structural and functional unit of a matter. The term “atom” comes from the Greek word which means indivisible, because atoms are the smallest things in the universe and could not be divided. atoms are made up of three subatomic particles like protons, neutrons and electrons.
What is the difference between the mass number of an isotope and its atomic number quizlet?
Atomic number is the amount of protons in the nucleus. The mass number is the total number of protons and neutrons.



