What is the energy of the particles in a liquid
The particles in a liquid have more kinetic energy than the particles in the corresponding solid. As a result, the particles in a liquid move faster in terms of vibration, rotation, and translation.
What energy do liquid particles have?
Liquids have more kinetic energy than solids. If you add heat energy to a liquid, the particles will move faster around each other as their kinetic energy increases. Some of these particles will have enough kinetic energy to break their liquid bonds and escape as a gas (evaporation).
What is the energy of particles in a gas?
Gas particles are in constant rapid motion in random directions . The fast motion of gas particles gives them a relatively large amount of kinetic energy. Recall that kinetic energy is the energy that an object possesses because of its motion.
What is the energy of particles in a solid?
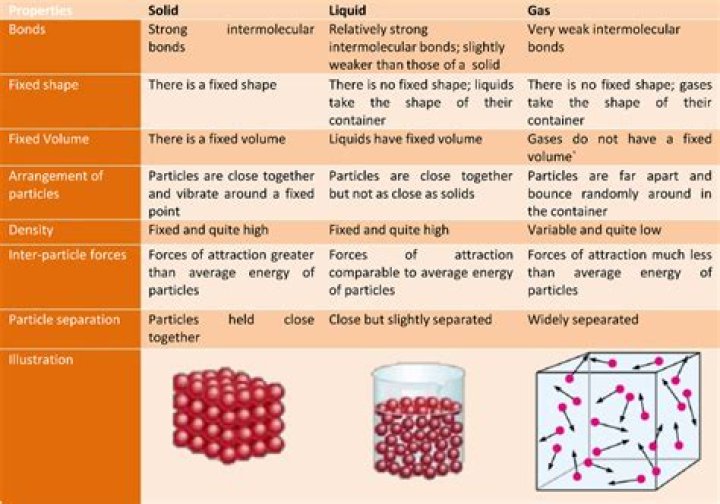
Solid � In a solid, the attractive forces keep the particles together tightly enough so that the particles do not move past each other. Their vibration is related to their kinetic energy. In the solid the particles vibrate in place.Is liquid high or low energy?
Molecules in a liquid have more energy than molecules in a solid. And if you heat it up even more, the molecules will speed up so much that they won’t be stuck together at all. The molecules in the gas have the most energy.
What is liquid in chemistry?
A liquid is made up of tiny vibrating particles of matter, such as atoms, held together by intermolecular bonds. Like a gas, a liquid is able to flow and take the shape of a container. Most liquids resist compression, although others can be compressed. … Water is, by far, the most common liquid on Earth.
What are the particles in a liquid?
The particles of a liquid are close together, always moving, and can slide past one another. compared to the particles in a solid or liquid, and are constantly moving. The particles don’t interact with one another but just hit and bounce off of each other when they collide.
What has more energy solid liquid or gas?
In terms of relative energy, gas particles have the most energy, solid particles have the least energy and liquid particles are somewhere in between.What are solid to liquid?
When a solid is heated above its melting point, it becomes liquid because the pressure is higher than the triple point of the substance. Intermolecular (or interatomic or interionic) forces are still important, but the molecules have enough energy to move around, which makes the structure mobile.
Are liquids and solids compressible?Solids are non-compressible and have constant volume and constant shape. Liquids are non-compressible and have constant volume but can change shape. … Gases do not have a constant volume or shape; they not only take the shape of the container they are in, they try to fill the entire container.
Article first time published onDoes liquid have potential energy?
Gases have zero potential energy (or close enough for real gases and zero for ideal gases). That means, that solids and liquids have negative potential energy. So, as heat is applied to a solid or liquid, potential energy increases, so the value of potential energy becomes less negative and gets closer to zero.
What is it called when a liquid turns into a gas?
Vaporization of a sample of liquid is a phase transition from the liquid phase to the gas phase. There are two types of vaporization: evaporation and boiling. Evaporation occurs at temperatures below the boiling point, and occurs on the liquid’s surface.
Do liquid particles vibrate?
liquid vibrate, move about, and slide past each other. solid vibrate (jiggle) but generally do not move from place to place.
What is the meaning of liquid energy?
Liquid fuels are combustible or energy-generating molecules that can be harnessed to create mechanical energy, usually producing kinetic energy; they also must take the shape of their container. … Many liquid fuels play a primary role in transportation and the economy.
How does the particles in a liquid move?
In liquids, particles are quite close together and move with random motion throughout the container. Particles move rapidly in all directions but collide with each other more frequently than in gases due to shorter distances between particles.
How particles are arranged in a liquid?
Particle Arrangement in Liquids In liquids, the particles are near each other, but not tightly packed. The particles in a liquid are able to slide past each other. … The looser arrangement of particles is what allows liquids to take on the shape of their container.
How are the molecules in a liquid?
The molecules (or atoms or ions) of a liquid, like those of a solid (and unlike those of a gas), are quite close together; however, while molecules in a solid are held in fixed positions by intermolecular forces, molecules in a liquid have too much thermal energy to be bound by these forces and move about freely within …
What is gasses in chemistry?
Gas is a state of matter that has no fixed shape and no fixed volume. Gases have lower density than other states of matter, such as solids and liquids. There is a great deal of empty space between particles, which have a lot of kinetic energy. … The particles exert more force on the interior volume of the container.
Are cats a liquid?
At the center of the definition of a liquid is an action: A material must be able to modify its form to fit within a container. … Cats are thus liquid if we give them the time to become liquid.
Why do liquids flow?
Liquids flow because the intermolecular forces between molecules are weak enough to allow the molecules to move around relative to one another. Intermolecular forces are the forces between neighboring molecules. … At a molecular level, liquids have some properties of gases and some of solids.
When you add energy to matter the particles move?
Energy added: particles move faster and further apart, and matter expands (state changes from solid to liquid to gas). Energy removed: particles move slower and closer together, and matter contracts (state changes from gas to liquid to solid).
How does adding energy to a solid affect the motion of the particles?
One change of state happens when you add energy to the substance. This change of state is called melting. By adding energy to the molecules in a solid the molecules begin to move quicker and can break away from the other molecules.
How do the particles in a liquid create surface tension?
- A molecule in the bulk liquid experiences cohesive forces with other molecules in all directions.
- A molecule at the surface of a liquid experiences only net inward cohesive forces.
What are the 3 states of energy?
- Chemical energy is energy stored in the bonds of atoms and molecules. …
- Mechanical energy is energy stored in objects by tension. …
- Nuclear energy is energy stored in the nucleus of an atom—the energy that holds the nucleus together.
Do atoms in a liquid have more energy than atoms in a solid?
The atoms in a liquid have more energy than the atoms in a solid. There is a special temperature for every substance called the melting point. … Liquid water freezes and becomes solid ice when the molecules lose energy.
Does gasoline have energy?
Gasoline also known as petrol is an energy-dense secondary fuel that can be thought of as an energy currency. It is used to power many heat engines, most importantly it acts as a fuel for a large proportion of cars. … Gasoline is essential to running most internal combustion engine cars.
Is gas considered a fluid?
Liquids and gases are called fluids because they can be made to flow, or move.
Are gasses compressible?
Gases are compressible because most of the volume of a gas is composed of the large amounts of empty space between the gas particles. At room temperature and standard pressure, the average distance between gas molecules is about ten times the diameter of the molecules themselves.
Do liquids have high compressibility?
The spaces between the molecules in liquids are large, so liquids have very high compressibility. Gases will compress more easily that solids or liquids because there is so much space between the gas molecules.
Does liquid have kinetic energy?
The particles in a liquid have more kinetic energy than the particles in the corresponding solid. As a result, the particles in a liquid move faster in terms of vibration, rotation, and translation. … Differences in kinetic energy alone cannot explain the relative densities of liquids and solids.
Which has the highest kinetic energy?
The particles of steam has highest kinetic energy because steam is in the form of gases in which the particles of gases are much apart from one another. The space between the particles of gases are very large.



