What is the geometry of ch3
It is trigonal pyramidal
What is the geometry shape of CH3?
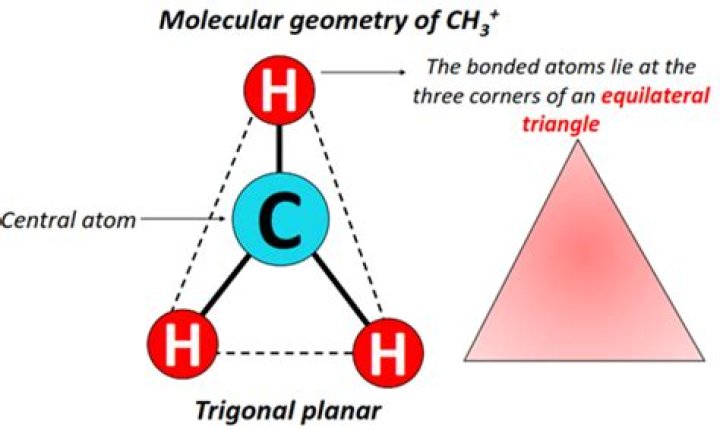
CH3 (methyl free radical) has a planar structure with sp2 hybridization of ‘C’ atom. The odd electron is present in an unhybridized 2pz orbital. (methyl carbonium ion) also has trigonal planar structure (sp2).
Is CH3+ trigonal pyramidal?
BF3 and CH3+ have Sp2 hybridisation and trigonal planer Geometry while CH3- have Sp3 hybridisation with 3 sigma and loan pair as negative charge , hence Geometry is trigonal pyramidal instead of tetrahedral.
What is the geometry of CH3 positive?
The shape of CH3^+ is (planar/bent).Does CH3+ have trigonal planar shape?
So, the correct answer is “Option A”.
Is CH3 a sp2?
Ch 2: sp3 hybridisation.
What is the shape and bond angle of CH3?
The carbon atom in the ion CH+3 is sp2 hybridized carbon and therefore, the geometry around this atom is trigonal planar. In this case, the H−C−H bond angle is 120∘ .
What is hybridization in CH3+?
The carbon atom in the ion CH3+ is sp2 hybridized carbon and therefore, the geometry around this atom is trigonal planar.Is CH3 polar?
Because there electronegativity difference is too less. So over all, CH3CH3 (Ethane) is a non polar compound. For the memorization of bonding nature following rules should be keep in mind.
What is the molecular geometry of ClO3?The shape of ClO3- ion is trigonal pyramidal. According to VSEPR (Valence shell electron pair) theory.
Article first time published onWhat is the molecular geometry of co32?
3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
Which is central atom in CH3?
The central atom in the CH3− ion is (C or H). This (C or H) atom has the following hybridized orbitals (sp, sp^2, or sp^3).
What shape does the methyl cation CH3 have?
shape of methyl cation will be of trigonal planar shaped as its hybridisation is SP2 and no lone pair is present.
Why is CH3 planar?
CH3• is planar because of H−H steric repulsion. This repulsion is much weaker for SiH3•, GeH3•, and SnH3• due to the larger H−H separation. Consequently, the geometry of the heavier homologs is determined by electronic effects, which always favor a pyramidal structure.
Does CH3 have tetrahedral bond angles?
Each CH 3 group is AX4; its electron-group arrangement is tetrahedral. … The CH3 groups should have about 109.5° bond angles. The C=O. double bond will compress C-C-C angle to less than the ideal 120°.
What is the bond angle for HCN?
HCN, hydrogen cyanide, is a volatile and poisnous compound with distinguished bitter odor. It is linear molecule with a triple bond between C and N atom and has bond angle of 180 degrees.
How do you find the molecular geometry?
- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
Is sp3 a tetrahedral?
For sp3 hybridized central atoms the only possible molecular geometry is tetrahedral. If all the bonds are in place the shape is also tetrahedral.
What is a sp3?
The term “sp3 hybridization” refers to the mixing character of one 2s-orbital and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be sp3 hybridized, it must have an s orbital and three p orbitals.
How do you know if its sp2 or sp3?
- If it’s 4, your atom is sp3.
- If it’s 3, your atom is sp2.
- If it’s 2, your atom is sp.
Is CH3 anion polar?
The [CH3-] group, a methyl anion, has covalent bonds between carbon and hydrogen. … Since there is a bond polarity to a S-O bond these are polar covalent bonds. The attraction of the Na+ cations to SO42- is ionic.
Why CH3 is non-polar?
When we take CH3 only, then we say that it is non-polar because the C and H have similar electronegativities. So, they cancel each other and thus CH3 is non-polar. But CH. While CH3+ is again non-polar because the structure is trigonal planar.
Is CH3 soluble in water?
You specifically asked about the solubility of methylamine (CH3NHCH3(. This is a polar molecule that doesn’t comntain any long carbon chains. The nitrogen-hydrogen group allows the formation of hydrogen bonds with water, so methylamine will dissolve quite well in water.
Why is CH3 sp2 hybridized?
In methyl free radical the hybridization is sp2 because it has 3 bond pairs and one unpaired electron which is very reactive so in hybridization it is not included and 3 bond pairs are present so one goes with s and other 2 with p.
Is CO32 a polar molecule?
In the carbonate ion, each of the carbon to oxygen bonds is polar due to the electronegativity difference between the two atoms. However because the three polar bonds are symmetrically arranged around the center of the structure, the carbonate ion is NON-polar.
Is asf3 trigonal planar?
Which of the following is the molecular geometry of AsF3? 3. A molecule is trigonal planar and has only single bonds to the central atom.
What is the shape of methyl?
Methyl radical Shape or Geometry: According to the valence shell electron pair repulsion theory, when three bond pair electrons are present around the central atom, it will result in the formation of trigonal planar shape or geometry of methyl radical.
Is CH3 3N trigonal planar?
(CH3)3 N is pyramidal but (SiH3)3 N is trigonal planar.



