What is the Lewis structure for ch3cn
Name of MoleculeAcetonitrile/methyl cyanideChemical formulaCH3CNMolecular geometry of CH3CNTetrahedral/linearElectron geometry of CH3CNTetrahedral/linearHybridizationSp³ and Sp
What is the Lewis structure for ch3cn? - Google Search
Name of MoleculeAcetonitrile/methyl cyanideChemical formulaCH3CNMolecular geometry of CH3CNTetrahedral/linearElectron geometry of CH3CNTetrahedral/linearHybridizationSp³ and Sp
How many atoms are bonded to C in CH3CN?
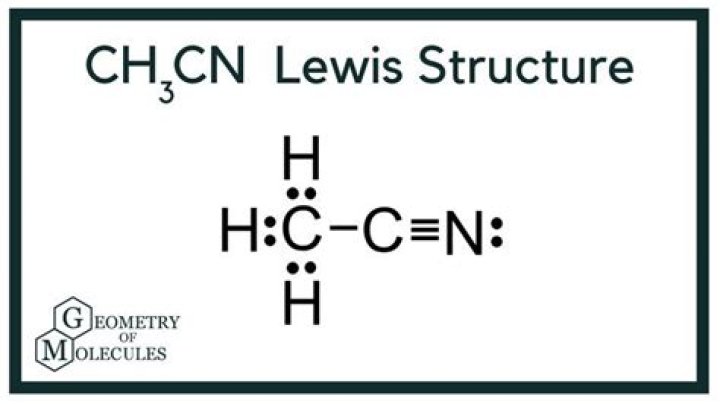
Explanation: Start by drawing the Lewis structure of acetonitrile, CH3CN . Now, the two carbon atoms will be bonded together via a single bond. One of the two carbon atoms will be bonded to the nitrogen atom via a triple bond and the other will be bonded to the three hydrogen atoms via single bonds.
How many electrons does CH3CN have?
For CH3CN we have 4 valence electrons for the Carbon plus 1 for the Hydrogen (we have 3 Hydrogens) plus 4 for the other Carbon and then 5 for that Nitrogen, giving us a total of 16 valence electrons. Carbon’s the least electronegative, so that’s going to go at the center.What type of hybridization is CH3CN?
Re: Hybridization of CH3CN The central C atom has a hybridization of sp3 for its four regions of electron density.
Is CH3CN symmetric?
Symmetric top molecules such as CF3CCH, CF3CN and CH3CN have been investigated in different vibrational excited states before.
What is the hybridization of CH3CN?
When we come to CH3CN, 1st carbon has 2sigma bonds and 2pi bonds,as from above I mentioned only sigma bonds and lone pairs are taken into consideration for hybridization.So 1st carbon hybridization is SP. And second carbon have 4sigma bonds so it is SP3 hybridized. The C atom of CH3 group is sp3 hybridised.
How many pi bonds are in CH3CN?
There are a total of 5 sigma bonds and 2 pi bonds in CH3CN.Is CH3CN ionic or covalent?
These measures show that the CuCN and CuNC bonding is dominantly ionic while the bonding for CH3CN and CH3NC is dominantly covalent. For the CH3 containing molecules, there is participation of the cyanide 2 pi orbital in the bond however, it is not involved in the ionic bond with Cu.
Is CH3CN polar or nonpolar?CH3CN is polar and thus has the strongest intermolecular forces and should have the highest boiling point.
Article first time published onHow do you find the shape of XeF5?
In $\text{XeF}_{\text{5}}^{-}$ , the 5 bond pairs and two lone pairs are arranged to give the basic shape of a bipyramidal pentagonal.
What is the shape of XeF5+?
Since the molecule \[XeF_5^ + \]has only five sigma bonds, its geometry is square pyramidal.
What is hybridization of XeF5?
Thus in XeF5+ steric no. = 1/2(8+5–1)=6. Hybridisation is sp3d2.
Is ch3cn a solvent?
It is an aliphatic nitrile and a volatile organic compound. Acetonitrile has many uses, including as a solvent, for spinning fibers, and in lithium batteries.
How do you find the hybridization of CH3Cl?
Answer: Hybridisation = number of sigma bonds + lone pair. CH3Cl = 3 sigma bonds between C & H and 1 between C and Cl There is no lone pair as carbon has 4 valence electrons and all of them have formed a bond (3 with hydrogen and 1 with Cl). Now, hybridisation = (3+1) + 0= 4 = sp3 (1 s & 3 p).
What is the type of hybridization in hconh2?
2) HCONH2 : In this compound carbon is sp2 hybridised.
How do you calculate hybridisation?
- If it’s 4, your atom is sp3.
- If it’s 3, your atom is sp2.
- If it’s 2, your atom is sp.
What is the Iupac name of CH3CN Class 11?
The IUPAC name of CH3CN is methyl cyanide.
Which of the following name is incorrect for CH3CN?
Methyl cyanide is the common name for ethane nitrile and therefore the above statement is false that methyl cyanide is not the IUPAC name of $ C{H_3}CN $ . This statement is false.
Is Mecn polar?
SolventRelative Polarityacetonitrile0.46nitromethane0.4812-propanol0.546benzyl alcohol0.608
Is c2h3n polar or nonpolar?
Acetonitrile is a strongly polar solvent in its solvability, comparable to alcohols. Like alcohols, it is not a donor of hydrogen bonds but is a strong acceptor of hydrogen bonds.
Is CH3CN an acid or base?
Acetonitrile (CH3CN) has a pKa of 25, making it more acidic than many other compounds having only…
What does CH3CN dissolve into?
NamesBoiling point81.3 to 82.1 °C; 178.2 to 179.7 °F; 354.4 to 355.2 KSolubility in waterMisciblelog P−0.334Vapor pressure9.71 kPa (at 20.0 °C)
Does CH3CN dissociate?
measured in bulk CH3CN solution. energies would certainly dissociate on the time scale from production to laser irradiation (several milliseconds).
Is CH3CN organic?
Acetonitrile, often abbreviated MeCN (methyl cyanide), is the chemical compound with the formula CH3CN. This colourless liquid is the simplest organic nitrile (hydrogen cyanide is a simpler nitrile, but the cyanide anion is not classed as organic). It is produced mainly as a byproduct of acrylonitrile manufacture.
How many sigma and pi bonds are in benzamide?
13 σ and 4 π-bonds.
How do you identify sigma and pi bonds in organic compounds?
Usually, all bonds between atoms in most organic compounds contain one sigma bond each. If it is a single bond, it contains only sigma bond. Double and Triple bonds, however, contains sigma and pi bonds. Double bonds have one each, and triple bonds have one sigma bond and two pi bonds.
What intermolecular force is NH2OH?
In this question, why is it that NH2OH has london forces, dipole-dipole, hydrogen bonding, but CBr4 only has London forces listed?
What is NH2OH called?
Hydroxylamine | NH2OH – PubChem.
What intermolecular force is CH3CH2CH3?
CH3CH2CH3 or CH3CH2CH2CH2CH2CH3. Dispersion forces are the only intermolecular forces in these compounds. The longer chain has more electrons (more bonds) and so it possesses the stronger dispersion forces.
What is the intermolecular force of CHCl3?
Dipole forces are the dominant intermolecular forces of attraction between CHCl3 molecules while the dominant intermolecular forces of attraction within CCl4 molecules are London forces.



