What is the universal solvent
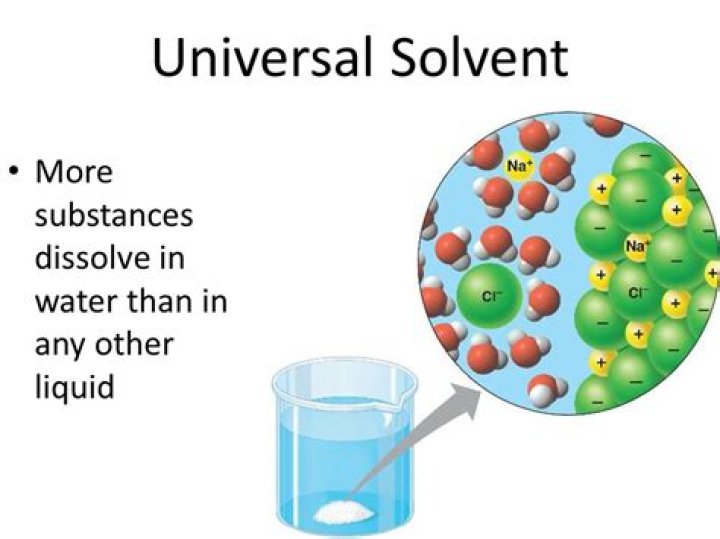
Water is called the “universal solvent” because it is capable of dissolving more substances than any other liquid. … Water molecules have a polar arrangement of oxygen and hydrogen atoms—one side (hydrogen) has a positive electrical charge and the other side (oxygen) had a negative charge.
What is universal solvent and example?
A universal solvent is a substance that dissolves most chemicals. Water is called the universal solvent because it dissolves more substances than any other solvent. However, no solvent, including water, dissolves every chemical. … Nonpolar solvents dissolve nonpolar molecules such as fats and other organic compounds.
Is a universal solvent *?
Water is a universal solvent.
What is the universal solvent of water?
Water is called the universal solvent because more substances dissolve in water than in any other chemical. This has to do with the polarity of each water molecule. The hydrogen side of each water (H2O) molecule carries a slight positive electric charge, while the oxygen side carries a slight negative electric charge.What are two universal solvents?
Water, described as the “universal solvent” for its ability to dissolve many substances. Alkahest, a hypothetical solvent able to dissolve every other substance. The Universal Solvent, a comic by Don Rosa.
Are there other universal solvents than water?
Perhaps the most common solvent in everyday life is water. Many other solvents are organic compounds, such as benzene, tetrachloroethylene, or turpentine.
Is acetone a universal solvent?
Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances, while other solvents can only dissolve one or the other. Acetone’s chemical makeup includes elements that are both polar and nonpolar which means acetone can be used with both organic and inorganic substances.
What is the formula of that universal solvent explain?
This is a big part of why water is the universal solvent. … Now, water has the molecular formula H2O, so every water molecule has two hydrogen atoms and one oxygen atom as you can see in the diagram below. The hydrogen atoms aren’t on opposite sides of each other, and they carry a partial positive charge.What is the formula of universal solvent?
The chemical composition and physical attributes of water make it an excellent solvent. The chemical formula of water is H2O. It is a neutral atom, in which one part has a partial positive charge and another part has a partial negative charge. Water is a polar molecule.
Why is water called a universal solvent Weegy?Water is called the “universal solvent” because it dissolves more substances than any other liquid.
Article first time published onWhy water is an excellent solvent?
Because of its polarity and ability to form hydrogen bonds, water makes an excellent solvent, meaning that it can dissolve many different kinds of molecules.
Which statement best explains why water is known as the universal solvent?
Water is called the “universal solvent” because it is capable of dissolving more substances than any other liquid. This is important to every living thing on earth. It means that wherever water goes, either through the air, the ground, or through our bodies, it takes along valuable chemicals, minerals, and nutrients.
Are made up of solutes and solvents?
A solution is a homogeneous mixture consisting of a solute dissolved into a solvent . The solute is the substance that is being dissolved, while the solvent is the dissolving medium. Solutions can be formed with many different types and forms of solutes and solvents.
What is short saturated solution?
A saturated solution is a solution that contains the maximum amount of solute that is capable of being dissolved. … When 30.0 g of NaCl is added to 100 ml of water, it all dissolves, forming an unsaturated solution. When 40.0 g is added, 36.0 g dissolves and 4.0 g remains undissolved, forming a saturated solution.
What is a saturated solution?
A solution in which the maximum amount of solvent has been dissolved. Any more solute added will sit as crystals on the bottom of the container.
What is a universal solvent question?
Question 4 Water dissolves most of the compound to form a solution. Water can dissolve all three states of matter solids, liquids, and gases. Hence water is called a universal solvent.
What is the strongest solvent?
As per the general information floating around in the web and the details given in some of the books water is the strongest solvent amongst others. It is also sometimes called the “universal solvent” as it can basically dissolve most of the substances than any other liquid. Water is a good solvent due to its polarity.
What are the types of solvent?
There are two types of solvents they are organic solvents and inorganic solvents. Inorganic solvents are those solvents which do not contain carbon such as water, ammonia whereas organic solvents are those solvents which contain carbon and oxygen in their composition such as alcohols, glycol ethers.
What type of solvent is ethanol?
Ernest Z. Water and ethanol are polar protic solvents. They both contain polar O-H bonds, so they are polar molecules. By definition, a polar solvent has a dipole moment greater than 1.6 D and a dielectric constant greater than 15.
Is salt solute or solvent?
In a NaCl solution (salt-water), the solvent is water. A solute is the component in a solution in the lesser amount. In a NaCl solution, the salt is the solute.
Is rubbing alcohol a solvent or solute?
From the above, you can see that isopropyl alcohol is the substance that is present in the largest quantity, therefore, it is the solvent. This would then make water and the medication the solutes.
Which is the better solvent?
Energy from hot water molecules makes solids more soluble. In hot water, molecules are moving around more, so there are more collisions between the water molecules and a solid. A solvent is a substance that can dissolve other substances – water is a good solvent.
Do all solutes dissolve in solvents?
Solutes and solvents may be any state of matter. Many solutes dissolve in water because water is a very polar compound. A general rule: like dissolves like. For example, polar solvents dissolve polar solutes, and nonpolar solvents dissolve nonpolar solutes.
Why is ice less dense than water?
Ice actually has a very different structure than liquid water, in that the molecules align themselves in a regular lattice rather than more randomly as in the liquid form. It happens that the lattice arrangement allows water molecules to be more spread out than in a liquid, and, thus, ice is less dense than water.
What makes a mixture different from other types of matter?
Matter can be broken down into two categories: pure substances and mixtures. … A chemical substance is composed of one type of atom or molecule. A mixture is composed of different types of atoms or molecules that are not chemically bonded.
What does the word competition mean in biology Weegy?
In biology, competition refers to the rivalry between or among living things for territory, resources, goods, mates, etc.
What does a pyramid biomass represent?
A biomass pyramid is a diagram that compares the biomass of different trophic levels within an ecosystem. It shows the mass of producers that are needed to support primary consumers, the mass of primary consumers required to support secondary consumers, and so on.
How is an organism related to a population?
Population – The population is defined as the group of organisms of the same species that live in the same area. Organism – An organism is a single individual plant, animal or other living thing.
What causes polarity?
Polarity results from the uneven partial charge distribution between various atoms in a compound. Atoms, such as nitrogen, oxygen, and halogens, that are more electronegative have a tendency to have partial negative charges. … A polar molecule results when a molecule contains polar bonds in an unsymmetrical arrangement.
Is Salt hydrophilic or hydrophobic?
The degree or extent to which a molecule or surface attracts water is known as the ‘hydrophilicity’ of that molecule. Some of the most common examples of hydrophilic substances are sugar, salt, starch, and cellulose. Hydrophilic substances are polar in nature.
What are the three states of matter of water?
Water is known to exist in three different states; as a solid, liquid or gas. Clouds, snow, and rain are all made of up of some form of water. A cloud is comprised of tiny water droplets and/or ice crystals, a snowflake is an aggregate of many ice crystals, and rain is just liquid water.



